Photochemistry

Photochemistry is the branch of
In nature, photochemistry is of immense importance as it is the basis of photosynthesis, vision, and the formation of vitamin D with sunlight.[2] It is also responsible for the appearance of DNA mutations leading to skin cancers.[3]
Photochemical reactions proceed differently than temperature-driven reactions. Photochemical paths access high-energy intermediates that cannot be generated thermally, thereby overcoming large activation barriers in a short period of time, and allowing reactions otherwise inaccessible by thermal processes. Photochemistry can also be destructive, as illustrated by the photodegradation of plastics.
Concept
Grotthuss–Draper law and Stark–Einstein law
Fluorescence and phosphorescence
When a molecule or atom in the
Kasha's rule stipulates that higher singlet states would quickly relax by radiationless decay or internal conversion (IC) to S1. Thus, S1 is usually, but not always, the only relevant singlet excited state. This excited state S1 can further relax to S0 by IC, but also by an allowed radiative transition from S1 to S0 that emits a photon; this process is called fluorescence.
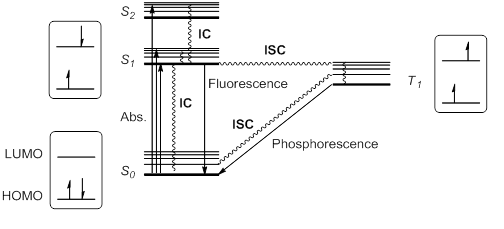
Alternatively, it is possible for the excited state S1 to undergo spin inversion and to generate a triplet excited state T1 having two unpaired electrons with the same spin. This violation of the spin selection rule is possible by intersystem crossing (ISC) of the vibrational and electronic levels of S1 and T1. According to Hund's rule of maximum multiplicity, this T1 state would be somewhat more stable than S1.
This triplet state can relax to the ground state S0 by radiationless IC or by a radiation pathway called phosphorescence. This process implies a change of electronic spin, which is forbidden by spin selection rules, making phosphorescence (from T1 to S0) much slower than fluorescence (from S1 to S0). Thus, triplet states generally have longer lifetimes than singlet states. These transitions are usually summarized in a state energy diagram or Jablonski diagram, the paradigm of molecular photochemistry.
These excited species, either S1 or T1, have a half-empty low-energy orbital, and are consequently more
Experimental setup

Photochemical reactions require a light source that emits wavelengths corresponding to an electronic transition in the reactant. In the early experiments (and in everyday life), sunlight was the light source, although it is polychromatic.
The emitted light must reach the targeted
Typically, the wavelength employed to induce a photochemical process is selected based on the absorption spectrum of the reactive species, most often the absorption maximum. Over the last years[when?], however, it has been demonstrated that, in the majority of bond-forming reactions, the absorption spectrum does not allow selecting the optimum wavelength to achieve the highest reaction yield based on absorptivity. This fundamental mismatch between absorptivity and reactivity has been elucidated with so-called photochemical action plots.[8][9]
Photochemistry in combination with flow chemistry
Continuous-flow photochemistry offers multiple advantages over batch photochemistry. Photochemical reactions are driven by the number of photons that are able to activate molecules causing the desired reaction. The large surface-area-to-volume ratio of a microreactor maximizes the illumination, and at the same time allows for efficient cooling, which decreases the thermal side products.[10]
Principles
In the case of photochemical reactions, light provides the activation energy. Simplistically, light is one mechanism for providing the activation energy required for many reactions. If laser light is employed, it is possible to selectively excite a molecule so as to produce a desired electronic and vibrational state.[11] Equally, the emission from a particular state may be selectively monitored, providing a measure of the population of that state. If the chemical system is at low pressure, this enables scientists to observe the energy distribution of the products of a chemical reaction before the differences in energy have been smeared out and averaged by repeated collisions.
The absorption of a photon by a reactant molecule may also permit a reaction to occur not just by bringing the molecule to the necessary activation energy, but also by changing the symmetry of the molecule's electronic configuration, enabling an otherwise-inaccessible reaction path, as described by the Woodward–Hoffmann selection rules. A [2+2] cycloaddition reaction is one example of a pericyclic reaction that can be analyzed using these rules or by the related frontier molecular orbital theory.
Some photochemical reactions are several orders of magnitude faster than thermal reactions; reactions as fast as 10−9 seconds and associated processes as fast as 10−15 seconds are often observed.
The photon can be absorbed directly by the reactant or by a photosensitizer, which absorbs the photon and transfers the energy to the reactant. The opposite process, when a photoexcited state is deactivated by a chemical reagent, is called quenching.
Most photochemical transformations occur through a series of simple steps known as primary photochemical processes. One common example of these processes is the excited state proton transfer.
Photochemical reactions
Examples of photochemical reactions
- Photosynthesis: Plants use solar energy to convert carbon dioxide and water into glucose and oxygen.
- Human formation of vitamin D by exposure to sunlight.
- Polymerizations started by photoinitiators, which decompose upon absorbing light to produce the free radicals for radical polymerization.
- Photodegradation of many substances, e.g. polyvinyl chloride and Fp. Medicine bottles are often made from darkened glass to protect the drugs from photodegradation.
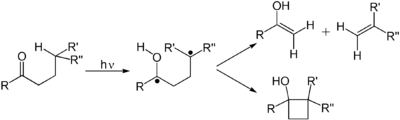
- Photochemical rearrangements, e.g.
- tumors by the action of singlet oxygen generated by photosensitized reactions of triplet oxygen. Typical photosensitizers include tetraphenylporphyrin and methylene blue. The resulting singlet oxygen is an aggressive oxidant, capable of converting C–H bonds into C–OH groups.
- Diazo printing process
- microelectroniccomponents.
- Vision is initiated by a photochemical reaction of rhodopsin.[15]
- Toray photochemical production of ε-caprolactame.[16]
- Photochemical production of artemisinin, an anti-malaria drug.[17][18]
- Photoalkylation, used for the light-induced addition of alkyl groups to molecules.
- DNA: photodimerization leading to cyclobutane pyrimidine dimers.[19]
Organic photochemistry
Examples of photochemical
In an industrial application, about 100,000 tonnes of benzyl chloride are prepared annually by the gas-phase photochemical reaction of toluene with chlorine.[22] The light is absorbed by chlorine molecules, the low energy of this transition being indicated by the yellowish color of the gas. The photon induces homolysis of the Cl-Cl bond, and the resulting chlorine radical converts toluene to the benzyl radical:
- Cl2 + hν → 2 Cl·
- C6H5CH3 + Cl· → C6H5CH2· + HCl
- C6H5CH2· + Cl· → C6H5CH2Cl
Inorganic and organometallic photochemistry
- Mo(CO)6 + THF → Mo(CO)5(THF) + CO
In a related reaction, photolysis of iron pentacarbonyl affords diiron nonacarbonyl (see figure):
- 2 Fe(CO)5 → Fe2(CO)9 + CO
Select photoreactive coordination complexes can undergo oxidation-reduction processes via single electron transfer. This electron transfer can occur within the inner or outer coordination sphere of the metal.[23]
Types of photochemical reactions
Here are some different types of photochemical reactions-
- Photo-dissociation: AB + hν → A* + B*
- Photo induced rearrangements, isomerization: A + hν → B
- Photo-addition: A + B + hν → AB + C
- Photo-substitution: A + BC + hν → AB + C
- Photo-redox reaction: A + B + hν → A− + B+
Historical
Although bleaching has long been practiced, the first photochemical reaction was described by Trommsdorff in 1834.[24] He observed that crystals of the compound α-santonin when exposed to sunlight turned yellow and burst. In a 2007 study the reaction was described as a succession of three steps taking place within a single crystal.[25]
The first step is a
(4). The bursting effect is attributed to a large change in crystal volume on dimerization.Specialized journals
- Journal of Photochemistry and Photobiology [1]
- ChemPhotoChem [2]
- Photochemistry and Photobiology [3]
- Photochemical & Photobiological Sciences [4]
- Photochemistry [5]
Learned Societies
- Inter-American Photochemical Society
- European Photochemistry Association
- Asian and Oceanian Photochemistry Association
International conferences
- IUPAC SYmposium on Photochemistry (biennial)
- International Conference on Photochemitry (biennial)
The organization of these conferences is facilitated by the International Foundation for Photochemistry.[26]
See also
- Photonic molecule
- Photoelectrochemical cell
- Photochemical logic gate
- Photosynthesis
- Light-dependent reactions
- List of photochemists
- Single photon sources
- Photogeochemistry
- Photoelectric effect
- Photolysis
- Blueprint
References
- PMID 27442273.
- ^ J. Cadet and T. Douki Photochem. & Photobiol. Sci. 2018 (17) pp 1816-1841 DOI: 10.1039/c7pp00395a
- ^ Calvert, J. G.; Pitts, J. N. Photochemistry. Wiley & Sons: New York, US, 1966. Congress Catalog number: 65-24288
- ^ Photochemistry, website of William Reusch (Michigan State University), accessed 26 June 2016
- ISBN 0-19-855886-4.
- ISSN 0365-9496.
- S2CID 244880552.
- PMID 37937391.
- PMID 21894087.
- PMID 29024596.
- ISBN 0444504079.
- S2CID 234209169, retrieved 2022-01-24
- S2CID 204117942.
- ISBN 9783527313044.
- S2CID 9323784.
- ^ Peplow, Mark (17 April 2013). "Sanofi launches malaria drug production". Chemistry World.
- PMID 23575629.
- ^ CYCLOBUTANE- TYPE PYRIMIDINE DIMERS IN POLYNUCLEOTIDES, R. B. Setlow, Science 1966 Vol. 153, p. 379, DOI: 10.1126/science.153.3734.379
- ISBN 978-1405190886.
- ISBN 978-1891389252.
- ISBN 3527306730.
- ISBN 9780120772506.
- .
- PMID 17645337.
- ^ IUPAC Symposia on Photochemistry. A Brief History, S. Braslavsky, Chemistry International, March–April 2014.
Further reading
- The Clarendon Press, 1942. 2nd edition, 1946.
- Photochemistry

