Photoheterotroph
Photoheterotrophs (
Research
Studies have shown that mammalian
Furthermore, inoculation experiments suggest that mixotrophic Ochromonas danica (i.e., Golden algae)—and comparable eukaryotes—favor photoheterotrophy in oligotrophic (i.e., nutrient-limited) aquatic habitats.[5] This preference may increase energy-use efficiency and growth by reducing investment in inorganic carbon fixation (e.g., production of autotrophic machineries such as RuBisCo and PSII).
Metabolism
Photoheterotrophs generate
Other organisms, including
as it needs reduced carbon compounds to live and cannot subsist on only light and CO2. It cannot carry out reactions in the form of- n CO2 + 2n H2D + photons → (CH2O)n+ 2n D + n H2O,
where H2D may be water, H2S or another compound/compounds providing the reducing electrons and protons; the 2D + H2O pair represents an oxidized form.
However, it can fix carbon in reactions like:
- CO2 + malate+ ADP +Pi
where malate or other useful molecules are otherwise obtained by breaking down other compounds by
- carbohydrate + O2 → malate + CO2 + energy.
This method of carbon fixation is useful when reduced carbon compounds are scarce and cannot be wasted as CO2 during interconversions, but energy is plentiful in the form of sunlight.
Ecology

Distribution and Niche Partitioning
Photoheterotrophs—either 1) cyanobacteria (i.e. facultative heterotrophs in nutrient-limited environments like Synechococcus and Prochlorococcus), 2) aerobic anoxygenic photoheterotrophic bacteria (AAP; employing bacteriochlorophyll-based reaction centers), 3) proteorhodopsin (PR)-containing bacteria and archaea, and 4) heliobacteria (i.e., the only phototroph with bacteriochlorophyll g pigments, or Gram-positive membrane) are found in various aquatic habitats including oceans, stratified lakes, rice fields, and environmental extremes.[10][11][12][13]
In oceans' photic zones, up to 10% of bacterial cells are capable of AAP, whereas greater than 50% of net marine microorganisms house PR—reaching up to 90% in coastal biomes.
In stratified (i.e., euxinic) lakes, photoheterotrophs—alongside other anoxygenic phototrophs (e.g., purple/green sulfur bacteria fixing carbon dioxide via electron donors such as ferrous iron, sulfide, and hydrogen gas)—often occupy the chemocline in the water column and/or sediments.[19] In this zone, dissolved oxygen is reduced, light is limited to long wavelengths (e.g., red and infrared) left-over by oxygenic phototrophs (e.g., cyanobacteria), and anaerobic metabolisms (i.e., those occurring in the absence of oxygen) begin introducing sulfide and bioavailable nutrients (e.g., organic carbon, phosphate, and ammonia) through upward diffusion.[20]
Heliobacteria are obligate anaerobes primarily located in rice fields, where low sulfide concentrations prevent competitive exclusion of purple/green sulfur bacteria.[21] These waterlogged environments may facilitate symbiotic relationships between heliobacteria and rice plants as fixed nitrogen—from the former—is exchanged for carbon-rich root exudates.
Observation studies have characterized photoheterotrophs (e.g., Green non-sulfur bacteria such as
Biogeochemical Cycling
Photoheterotrophs influence global carbon cycling by assimilating dissolved organic carbon (DOC).[23][20] Therefore, when harvesting light-energy, carbon is maintained in the microbial loop without corresponding respiration (i.e., carbon dioxide release to the atmosphere as DOC is oxidized to fuel energy production). This disconnect, the discovery of facultative photoheterotrophs (e.g., AAPs with flexible energy sources), and previous measurements taken in the dark (i.e., to avoid skewed oxygen consumption values due to photooxidation, UV light, and oxygenic photosynthesis) lead to overestimated aquatic CO2 emissions. For example, a 15.2% decrease in community respiration was observed in Cep Lake, Czechia—alongside preferential glucose and pyruvate uptake—is attributed to facultative photoheterotrophs preferring light-energy during the daytime, given fitness benefits mentioned previously.[23]
Flowchart
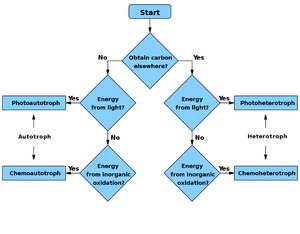
Energy source Carbon source |
Chemotroph | Phototroph |
|---|---|---|
| Autotroph | Chemoautotroph |
Photoautotroph |
| Heterotroph | Chemoheterotroph |
Photoheterotroph |
See also
References
- PMID 16997562.
- PMID 22900140.
- PMID 24198392.
- PMID 27731322.
- PMID 25138174.
- PMID 16997562.
- ISSN 0142-7873.
- ^ PMID 18552178.
- PMID 20436956.
- ISSN 0142-7873.
- ISBN 978-0-12-373944-5, retrieved 2023-04-15
- ^ PMID 31690593.
- ISSN 1726-4189.
- S2CID 220435352.
- PMID 25138174.
- ISBN 9780124158368, retrieved 2023-04-15
- PMID 19502441.
- S2CID 256555570.
- S2CID 11132075.
- ^ S2CID 98430905.
- ISBN 0-7923-3681-X, retrieved 2023-04-16
- ^ PMID 32198176.
- ^ PMID 34802055.
Sources
"Microbiology Online" (textbook). University of Wisconsin, Madison.
