Photon
Bosonic | ||
|---|---|---|
| Family | Gauge boson | |
| Interactions | Electromagnetic, weak (and gravity) | |
| Symbol | γ | |
| Theorized | Albert Einstein (1905) The name "photon" is generally attributed to Gilbert N. Lewis (1926) | |
| Mass | 0 (theoretical value) < 1×10−18 Mean lifetime Stable[1] | |
| Electric charge | 0
< 1×10−35 |
A photon (from
As with other elementary particles, photons are best explained by quantum mechanics and exhibit wave–particle duality, their behavior featuring properties of both waves and particles.[2] The modern photon concept originated during the first two decades of the 20th century with the work of Albert Einstein, who built upon the research of Max Planck. While Planck was trying to explain how matter and electromagnetic radiation could be in thermal equilibrium with one another, he proposed that the energy stored within a material object should be regarded as composed of an integer number of discrete, equal-sized parts. To explain the photoelectric effect, Einstein introduced the idea that light itself is made of discrete units of energy. In 1926, Gilbert N. Lewis popularized the term photon for these energy units.[3][4][5] Subsequently, many other experiments validated Einstein's approach.[6][7][8]
In the
Nomenclature
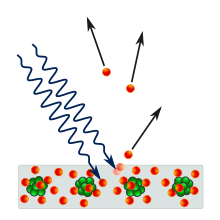

The word quanta (singular quantum, Latin for how much) was used before 1900 to mean particles or amounts of different quantities, including electricity. In 1900, the German physicist Max Planck was studying black-body radiation, and he suggested that the experimental observations, specifically at shorter wavelengths, would be explained if the energy stored within a molecule was a "discrete quantity composed of an integral number of finite equal parts", which he called "energy elements".[9] In 1905, Albert Einstein published a paper in which he proposed that many light-related phenomena—including black-body radiation and the photoelectric effect—would be better explained by modelling electromagnetic waves as consisting of spatially localized, discrete wave-packets.[10] He called such a wave-packet a light quantum (German: ein Lichtquant).[a]
The name photon derives from the Greek word for light, φῶς (transliterated phôs). Arthur Compton used photon in 1928, referring to Gilbert N. Lewis, who coined the term in a letter to Nature on 18 December 1926.[3][11] The same name was used earlier but was never widely adopted before Lewis: in 1916 by the American physicist and psychologist Leonard T. Troland, in 1921 by the Irish physicist John Joly, in 1924 by the French physiologist René Wurmser (1890–1993), and in 1926 by the French physicist Frithiof Wolfers (1891–1971).[5] The name was suggested initially as a unit related to the illumination of the eye and the resulting sensation of light and was used later in a physiological context. Although Wolfers's and Lewis's theories were contradicted by many experiments and never accepted, the new name was adopted by most physicists very soon after Compton used it.[5][b]
In physics, a photon is usually denoted by the symbol γ (the Greek letter gamma). This symbol for the photon probably derives from gamma rays, which were discovered in 1900 by Paul Villard,[13][14] named by Ernest Rutherford in 1903, and shown to be a form of electromagnetic radiation in 1914 by Rutherford and Edward Andrade.[15] In chemistry and optical engineering, photons are usually symbolized by hν, which is the photon energy, where h is the Planck constant and the Greek letter ν (nu) is the photon's frequency.[16]
Physical properties
The photon has no
In a vacuum, a photon has two possible
Photons are emitted in many natural processes. For example, when a charge is accelerated it emits synchrotron radiation. During a molecular, atomic or nuclear transition to a lower energy level, photons of various energy will be emitted, ranging from radio waves to gamma rays. Photons can also be emitted when a particle and its corresponding antiparticle are annihilated (for example, electron–positron annihilation).[26]: 572, 1114, 1172
Relativistic energy and momentum
In empty space, the photon moves at c (the speed of light) and its energy and momentum are related by E = pc, where p is the magnitude of the momentum vector p. This derives from the following relativistic relation, with m = 0:[27]
The energy and momentum of a photon depend only on its frequency () or inversely, its wavelength (λ):
where k is the wave vector, where
- k ≡ |k| = 2π /λ is the wave number, and
- ω ≡ 2 πν is the angular frequency, and
- ħ ≡ h/ 2π is the reduced Planck constant.[28]
Since points in the direction of the photon's propagation, the magnitude of its momentum is
Polarization and spin angular momentum
The photon also carries spin angular momentum, which is related to photon polarization. (Beams of light also exhibit properties described as orbital angular momentum of light).
The angular momentum of the photon has two possible values, either +ħ or −ħ. These two possible values correspond to the two possible pure states of circular polarization. Collections of photons in a light beam may have mixtures of these two values; a linearly polarized light beam will act as if it were composed of equal numbers of the two possible angular momenta.[29]: 325
The spin angular momentum of light does not depend on its frequency, and was experimentally verified by C. V. Raman and S. Bhagavantam in 1931.[30]
Antiparticle annihilation
The collision of a particle with its antiparticle can create photons. In free space at least two photons must be created since, in the
Seen another way, the photon can be considered as its own antiparticle (thus an "antiphoton" is simply a normal photon with opposite momentum, equal polarization, and 180° out of phase). The reverse process, pair production, is the dominant mechanism by which high-energy photons such as gamma rays lose energy while passing through matter.[32] That process is the reverse of "annihilation to one photon" allowed in the electric field of an atomic nucleus.
The classical formulae for the energy and momentum of electromagnetic radiation can be re-expressed in terms of photon events. For example, the pressure of electromagnetic radiation on an object derives from the transfer of photon momentum per unit time and unit area to that object, since pressure is force per unit area and force is the change in momentum per unit time.[33]
Experimental checks on photon mass
Current commonly accepted physical theories imply or assume the photon to be strictly massless. If photons were not purely massless, their speeds would vary with frequency, with lower-energy (redder) photons moving slightly slower than higher-energy photons. Relativity would be unaffected by this; the so-called speed of light, c, would then not be the actual speed at which light moves, but a constant of nature which is the
If a photon did have non-zero mass, there would be other effects as well.
Sharper upper limits on the mass of light have been obtained in experiments designed to detect effects caused by the galactic vector potential. Although the galactic vector potential is large because the galactic magnetic field exists on great length scales, only the magnetic field would be observable if the photon is massless. In the case that the photon has mass, the mass term 1/2m2AμAμ would affect the galactic plasma. The fact that no such effects are seen implies an upper bound on the photon mass of m < 3×10−27 eV/c2.[37] The galactic vector potential can also be probed directly by measuring the torque exerted on a magnetized ring.[38] Such methods were used to obtain the sharper upper limit of 1.07×10−27 eV/c2 (the equivalent of 10−36 daltons) given by the Particle Data Group.[39]
These sharp limits from the non-observation of the effects caused by the galactic vector potential have been shown to be model-dependent.[40] If the photon mass is generated via the Higgs mechanism then the upper limit of m ≲ 10−14 eV/c2 from the test of Coulomb's law is valid.
Historical development

In most theories up to the eighteenth century, light was pictured as being made of particles. Since

The Maxwell wave theory, however, does not account for all properties of light. The Maxwell theory predicts that the energy of a light wave depends only on its intensity, not on its frequency; nevertheless, several independent types of experiments show that the energy imparted by light to atoms depends only on the light's frequency, not on its intensity. For example, some chemical reactions are provoked only by light of frequency higher than a certain threshold; light of frequency lower than the threshold, no matter how intense, does not initiate the reaction. Similarly, electrons can be ejected from a metal plate by shining light of sufficiently high frequency on it (the photoelectric effect); the energy of the ejected electron is related only to the light's frequency, not to its intensity.[48][d]
At the same time, investigations of black-body radiation carried out over four decades (1860–1900) by various researchers[50] culminated in Max Planck's hypothesis[51][52] that the energy of any system that absorbs or emits electromagnetic radiation of frequency ν is an integer multiple of an energy quantum E = hν . As shown by Albert Einstein,[10][53] some form of energy quantization must be assumed to account for the thermal equilibrium observed between matter and electromagnetic radiation; for this explanation of the photoelectric effect, Einstein received the 1921 Nobel Prize in physics.[54]
Since the Maxwell theory of light allows for all possible energies of electromagnetic radiation, most physicists assumed initially that the energy quantization resulted from some unknown constraint on the matter that absorbs or emits the radiation. In 1905, Einstein was the first to propose that energy quantization was a property of electromagnetic radiation itself.[10] Although he accepted the validity of Maxwell's theory, Einstein pointed out that many anomalous experiments could be explained if the energy of a Maxwellian light wave were localized into point-like quanta that move independently of one another, even if the wave itself is spread continuously over space.[10] In 1909[53] and 1916,[55] Einstein showed that, if Planck's law regarding black-body radiation is accepted, the energy quanta must also carry momentum p = h / λ , making them full-fledged particles. This photon momentum was observed experimentally by Arthur Compton,[56] for which he received the Nobel Prize in 1927. The pivotal question then, was how to unify Maxwell's wave theory of light with its experimentally observed particle nature. The answer to this question occupied Albert Einstein for the rest of his life,[57] and was solved in quantum electrodynamics and its successor, the Standard Model. (See § Quantum field theory and § As a gauge boson, below.)

Einstein's 1905 predictions were verified experimentally in several ways in the first two decades of the 20th century, as recounted in
Even after Compton's experiment,
A few physicists persisted[63] in developing semiclassical models in which electromagnetic radiation is not quantized, but matter appears to obey the laws of quantum mechanics. Although the evidence from chemical and physical experiments for the existence of photons was overwhelming by the 1970s, this evidence could not be considered as absolutely definitive; since it relied on the interaction of light with matter, and a sufficiently complete theory of matter could in principle account for the evidence. Nevertheless, all semiclassical theories were refuted definitively in the 1970s and 1980s by photon-correlation experiments.[e] Hence, Einstein's hypothesis that quantization is a property of light itself is considered to be proven.
Wave–particle duality and uncertainty principles
Photons obey the laws of quantum mechanics, and so their behavior has both wave-like and particle-like aspects. When a photon is detected by a measuring instrument, it is registered as a single, particulate unit. However, the probability of detecting a photon is calculated by equations that describe waves. This combination of aspects is known as
While many introductory texts treat photons using the mathematical techniques of non-relativistic quantum mechanics, this is in some ways an awkward oversimplification, as photons are by nature intrinsically relativistic. Because photons have zero
Another difficulty is finding the proper analogue for the uncertainty principle, an idea frequently attributed to Heisenberg, who introduced the concept in analyzing a thought experiment involving an electron and a high-energy photon. However, Heisenberg did not give precise mathematical definitions of what the "uncertainty" in these measurements meant. The precise mathematical statement of the position–momentum uncertainty principle is due to Kennard, Pauli, and Weyl.[73][74] The uncertainty principle applies to situations where an experimenter has a choice of measuring either one of two "canonically conjugate" quantities, like the position and the momentum of a particle. According to the uncertainty principle, no matter how the particle is prepared, it is not possible to make a precise prediction for both of the two alternative measurements: if the outcome of the position measurement is made more certain, the outcome of the momentum measurement becomes less so, and vice versa.[75] A coherent state minimizes the overall uncertainty as far as quantum mechanics allows.[72] Quantum optics makes use of coherent states for modes of the electromagnetic field. There is a tradeoff, reminiscent of the position–momentum uncertainty relation, between measurements of an electromagnetic wave's amplitude and its phase.[72] This is sometimes informally expressed in terms of the uncertainty in the number of photons present in the electromagnetic wave, , and the uncertainty in the phase of the wave, . However, this cannot be an uncertainty relation of the Kennard–Pauli–Weyl type, since unlike position and momentum, the phase cannot be represented by a
Bose–Einstein model of a photon gas
In 1924,
The modern view on this is that photons are, by virtue of their integer spin, bosons (as opposed to
Stimulated and spontaneous emission

In 1916, Albert Einstein showed that Planck's radiation law could be derived from a semi-classical, statistical treatment of photons and atoms, which implies a link between the rates at which atoms emit and absorb photons. The condition follows from the assumption that functions of the emission and absorption of radiation by the atoms are independent of each other, and that thermal equilibrium is made by way of the radiation's interaction with the atoms. Consider a cavity in thermal equilibrium with all parts of itself and filled with electromagnetic radiation and that the atoms can emit and absorb that radiation. Thermal equilibrium requires that the energy density of photons with frequency (which is proportional to their number density) is, on average, constant in time; hence, the rate at which photons of any particular frequency are emitted must equal the rate at which they are absorbed.[84]
Einstein began by postulating simple proportionality relations for the different reaction rates involved. In his model, the rate for a system to absorb a photon of frequency and transition from a lower energy to a higher energy is proportional to the number of atoms with energy and to the energy density of ambient photons of that frequency,
where is the
where is the rate constant for emitting a photon spontaneously, and is the rate constant for emissions in response to ambient photons (induced or stimulated emission). In thermodynamic equilibrium, the number of atoms in state and those in state must, on average, be constant; hence, the rates and must be equal. Also, by arguments analogous to the derivation of
and
The and are collectively known as the Einstein coefficients.[85]
Einstein could not fully justify his rate equations, but claimed that it should be possible to calculate the coefficients , and once physicists had obtained "mechanics and electrodynamics modified to accommodate the quantum hypothesis".[86] Not long thereafter, in 1926, Paul Dirac derived the rate constants by using a semiclassical approach,[87] and, in 1927, succeeded in deriving all the rate constants from first principles within the framework of quantum theory.[88][89] Dirac's work was the foundation of quantum electrodynamics, i.e., the quantization of the electromagnetic field itself. Dirac's approach is also called second quantization or quantum field theory;[90][91][92] earlier quantum mechanical treatments only treat material particles as quantum mechanical, not the electromagnetic field.
Einstein was troubled by the fact that his theory seemed incomplete, since it did not determine the direction of a spontaneously emitted photon. A probabilistic nature of light-particle motion was first considered by Newton in his treatment of birefringence and, more generally, of the splitting of light beams at interfaces into a transmitted beam and a reflected beam. Newton hypothesized that hidden variables in the light particle determined which of the two paths a single photon would take.[44] Similarly, Einstein hoped for a more complete theory that would leave nothing to chance, beginning his separation[57] from quantum mechanics. Ironically, Max Born's probabilistic interpretation of the wave function[93][94] was inspired by Einstein's later work searching for a more complete theory.[95]
Quantum field theory
Quantization of the electromagnetic field

In 1910,
In 1925,

Dirac took this one step further.[88][89] He treated the interaction between a charge and an electromagnetic field as a small perturbation that induces transitions in the photon states, changing the numbers of photons in the modes, while conserving energy and momentum overall. Dirac was able to derive Einstein's and coefficients from first principles, and showed that the Bose–Einstein statistics of photons is a natural consequence of quantizing the electromagnetic field correctly (Bose's reasoning went in the opposite direction; he derived
Dirac's second-order perturbation theory can involve virtual photons, transient intermediate states of the electromagnetic field; the static electric and magnetic interactions are mediated by such virtual photons. In such quantum field theories, the probability amplitude of observable events is calculated by summing over all possible intermediate steps, even ones that are unphysical; hence, virtual photons are not constrained to satisfy , and may have extra polarization states; depending on the gauge used, virtual photons may have three or four polarization states, instead of the two states of real photons. Although these transient virtual photons can never be observed, they contribute measurably to the probabilities of observable events.[98]
Indeed, such second-order and higher-order perturbation calculations can give apparently infinite contributions to the sum. Such unphysical results are corrected for using the technique of renormalization.[99]
Other virtual particles may contribute to the summation as well; for example, two photons may interact indirectly through virtual electron–positron pairs.[100] Such photon–photon scattering (see two-photon physics), as well as electron–photon scattering, is meant to be one of the modes of operations of the planned particle accelerator, the International Linear Collider.[101]
In modern physics notation, the quantum state of the electromagnetic field is written as a Fock state, a tensor product of the states for each electromagnetic mode
where represents the state in which photons are in the mode . In this notation, the creation of a new photon in mode (e.g., emitted from an atomic transition) is written as . This notation merely expresses the concept of Born, Heisenberg and Jordan described above, and does not add any physics.
As a gauge boson
The electromagnetic field can be understood as a
The quanta of an
must be . These two spin components correspond to the classical concepts of right-handed and left-handed circularly polarized light. However, the transient virtual photons of quantum electrodynamics may also adopt unphysical polarization states.[102]In the prevailing
Hadronic properties
Measurements of the interaction between energetic photons and
Contributions to the mass of a system
The energy of a system that emits a photon is decreased by the energy of the photon as measured in the rest frame of the emitting system, which may result in a reduction in mass in the amount . Similarly, the mass of a system that absorbs a photon is increased by a corresponding amount. As an application, the energy balance of nuclear reactions involving photons is commonly written in terms of the masses of the nuclei involved, and terms of the form for the gamma photons (and for other relevant energies, such as the recoil energy of nuclei).[112]
This concept is applied in key predictions of quantum electrodynamics (QED, see above). In that theory, the mass of electrons (or, more generally, leptons) is modified by including the mass contributions of virtual photons, in a technique known as renormalization. Such "radiative corrections" contribute to a number of predictions of QED, such as the magnetic dipole moment of leptons, the Lamb shift, and the hyperfine structure of bound lepton pairs, such as muonium and positronium.[113]
Since photons contribute to the stress–energy tensor, they exert a gravitational attraction on other objects, according to the theory of general relativity. Conversely, photons are themselves affected by gravity; their normally straight trajectories may be bent by warped spacetime, as in gravitational lensing, and their frequencies may be lowered by moving to a higher gravitational potential, as in the Pound–Rebka experiment. However, these effects are not specific to photons; exactly the same effects would be predicted for classical electromagnetic waves.[114]
In matter
Light that travels through transparent matter does so at a lower speed than c, the speed of light in vacuum. The factor by which the speed is decreased is called the
Photons can be scattered by matter. For example, photons engage in so many collisions on the way from the core of the Sun that radiant energy can take about a million years to reach the surface;[116] however, once in open space, a photon takes only 8.3 minutes to reach Earth.[117]
Photons can also be
Technological applications
Photons have many applications in technology. These examples are chosen to illustrate applications of photons per se, rather than general optical devices such as lenses, etc. that could operate under a classical theory of light. The laser is an important application and is discussed above under stimulated emission.
Individual photons can be detected by several methods. The classic
Planck's energy formula is often used by engineers and chemists in design, both to compute the change in energy resulting from a photon absorption and to determine the frequency of the light emitted from a given photon emission. For example, the emission spectrum of a gas-discharge lamp can be altered by filling it with (mixtures of) gases with different electronic energy level configurations.[121]
Under some conditions, an energy transition can be excited by "two" photons that individually would be insufficient. This allows for higher resolution microscopy, because the sample absorbs energy only in the spectrum where two beams of different colors overlap significantly, which can be made much smaller than the excitation volume of a single beam (see two-photon excitation microscopy). Moreover, these photons cause less damage to the sample, since they are of lower energy.[122]
In some cases, two energy transitions can be coupled so that, as one system absorbs a photon, another nearby system "steals" its energy and re-emits a photon of a different frequency. This is the basis of
Several different kinds of
Quantum optics and computation
Much research has been devoted to applications of photons in the field of
Two-photon physics studies interactions between photons, which are rare. In 2018, Massachusetts Institute of Technology researchers announced the discovery of bound photon triplets, which may involve polaritons.[127][128]
See also
- Advanced Photon Source at Argonne National Laboratory
- Ballistic photon
- Dirac equation
- Doppler effect
- EPR paradox
- High energy X-ray imaging technology
- Luminiferous aether
- Medipix
- Phonon
- Photography
- Photon counting
- Photon epoch
- Photonic molecule
- Photonics
- Single-photon source
- Static forces and virtual-particle exchange
- Variable speed of light
Notes
- ^ Although the 1967 Elsevier translation of Planck's Nobel Lecture interprets Planck's Lichtquant as "photon", the more literal 1922 translation by Hans Thacher Clarke and Ludwik Silberstein Planck, Max (1922). "via Google Books". The Origin and Development of the Quantum Theory. Clarendon Press – via Internet Archive (archive.org, 2007-03-01). uses "light-quantum". No evidence is known that Planck himself had used the term "photon" as of 1926 (see also).
- ^ Asimov[12] credits Arthur Compton with defining quanta of energy as photons in 1923.[12]
- ^ However, it is possible if the system interacts with a third particle or field for the annihilation to produce one photon, since the third particle or field can absorb momentum equal and opposite to the single photon, providing dynamic balance. An example is when a positron annihilates with a bound atomic electron; in that case, it is possible for only one photon to be emitted, as the nuclear Coulomb field breaks translational symmetry.
- ^ The phrase "no matter how intense" refers to intensities below approximately 1013 W/cm2 at which point perturbation theory begins to break down. In contrast, in the intense regime, which for visible light is above approximately 1014 W/cm2, the classical wave description correctly predicts the energy acquired by electrons, called ponderomotive energy.[49] By comparison, sunlight is only about 0.1 W/cm2.
- ^ These experiments produce results that cannot be explained by any classical theory of light, since they involve anticorrelations that result from the quantum measurement process. In 1974, the first such experiment was carried out by Clauser, who reported a violation of a classical Cauchy–Schwarz inequality. In 1977, Kimble et al. demonstrated an analogous anti-bunching effect of photons interacting with a beam splitter; this approach was simplified and sources of error eliminated in the photon-anticorrelation experiment of Grangier, Roger, & Aspect (1986);[64] This work is reviewed and simplified further in Thorn, Neel, et al. (2004).[65]
- Lorentz boosts do not commute, and so simultaneously assigning low uncertainties to all coordinates of a relativistic particle's position becomes problematic.[71]
References
- ^ a b c d e f
Amsler, C.; et al. (S2CID 227119789.
- ^ Joos, George (1951). Theoretical Physics. London and Glasgow: Blackie and Son Limited. p. 679.
- ^ a b "December 18, 1926: Gilbert Lewis coins "photon" in letter to Nature". www.aps.org. Retrieved 2019-03-09.
- ^ "Gilbert N. Lewis". Atomic Heritage Foundation. Retrieved 2019-03-09.
- ^ arXiv:1401.0293 [physics.hist-ph].
- ^ Compton, Arthur H. (1965) [12 Dec 1927]. "X-rays as a branch of optics" (PDF). From Nobel Lectures, Physics 1922–1941. Amsterdam: Elsevier Publishing Company.
- .
- S2CID 250837011.
- .
- ^ .
- S2CID 4110026. see also
"Discordances entre l'expérience et la théorie électromagnétique du rayonnement". Written at Bruxelles, Belgium. Electrons et photons: Rapports et discussions du cinquième Conseil de Physique tenu à Bruxelles du 24 au 29 octobre 1927 sous les auspices de l'Institut International de Physique Solvay. Cinquième Conseil de Physique (in French). l'Institut International de Physique Solvay (host institution). Paris, France: Gauthier-Villars et Cie (published 1928). 24–29 October 1927. pp. 55–85.{{cite conference}}: CS1 maint: others (link) - ^ LCCN 66022515.
- Comptes Rendus des Séances de l'Académie des Sciences(in French). 130: 1010–1012.
- Comptes Rendus des Séances de l'Académie des Sciences(in French). 130: 1178–1179.
- .
- ISBN 978-1-118-69025-3.
- ^ Frisch, David H.; Thorndike, Alan M. (1964). Elementary Particles. Princeton, New Jersey: David Van Nostrand. p. 22.
- S2CID 119409823.
- Baez, John. "What is the mass of a photon?" (pers. academic site). U.C. Riverside.
- ISSN 0034-4885.
- ISSN 0034-6861.
- ISSN 0031-9007.
- ISBN 978-1-107-03473-0.
- ISBN 978-0-85274-328-7.
- S2CID 227119789.
- ^ ISBN 978-0-471-23231-5.
- ^ See Alonso & Finn 1968, Section 1.6.
- ^ Soper, Davison E. "Electromagnetic radiation is made of photons". Institute of Theoretical Science. University of Oregon.
- ISBN 978-0-201-83887-9.
- S2CID 4064852. Archived from the original(PDF) on 2016-06-03. Retrieved 2008-12-28.
- ISBN 978-3-527-40601-2.
- ^ Alonso & Finn 1968, Section 9.3.
- ISBN 978-0-486-65984-8.
- doi:10.1119/1.13917.
- .
- .
- .
- .
- S2CID 227119789. Summary Table
- S2CID 31249827.
- ISBN 978-0-268-00870-3.
- ISBN 978-0-486-49564-4.
- Traité de la lumière (in French).. An English translation is available from Project Gutenberg
- ^ ISBN 978-0-486-60205-9.
- )
- S2CID 186207827. This article followed a presentation by Maxwell on 8 December 1864 to the Royal Society.
- ^ Hertz, Heinrich (1888). "Über Strahlen elektrischer Kraft". Sitzungsberichte der Preussischen Akademie der Wissenschaften (in German). 1888. Berlin, Deutschland: 1297–1307.
- ^ "Frequency-dependence of luminiscence" pp. 276ff., §1.4 "photoelectric effect" in Alonso & Finn 1968.
- doi:10.1063/1.50410.
- ^ a b Wien, W. (1911). "Wilhelm Wien Nobel Lecture". nobelprize.org.
- ^
- ^ a b Planck, Max (1920). "Max Planck's Nobel Lecture". nobelprize.org.
- ^ a b c Einstein, Albert (1909). "Über die Entwicklung unserer Anschauungen über das Wesen und die Konstitution der Strahlung" (PDF). Physikalische Zeitschrift (in German). 10: 817–825. An English translation is available from Wikisource.
- ^ Presentation speech by Svante Arrhenius for the 1921 Nobel Prize in Physics, December 10, 1922. Online text from [nobelprize.org], The Nobel Foundation 2008. Access date 2008-12-05.
- ^ Einstein, Albert (1916). "Zur Quantentheorie der Strahlung". Mitteilungen der Physikalischen Gesellschaft zu Zürich (in German). 16: 47. Also Physikalische Zeitschrift (in German), 18, 121–128 (1917).
- ^ .
- ^ ISBN 978-0-19-853907-0.
- ^ Millikan, Robert A. (1924). "Robert A. Millikan's Nobel Lecture".
- .
- (in German), 24, p. 69 (1924).
- S2CID 9454552.
- ^ Heisenberg, Werner (1933). "Heisenberg Nobel lecture".
- ^ )
- S2CID 250837011.
- .
- Taylor, Geoffrey Ingram(1909). Interference fringes with feeble light. Cambridge Philosophical Society. Proceedings of the Cambridge Philosophical Society. Vol. 15. pp. 114–115.
- ISBN 978-0-471-35832-9.
- .
- .
- PMID 9912446.
- )
- ^ ISBN 978-0-521-43595-6.
- S2CID 24507489.
- .
- OCLC 2284121.
- .
- S2CID 186235974.
- ^ Einstein, Albert (1924). "Quantentheorie des einatomigen idealen Gases". Sitzungsberichte der Preussischen Akademie der Wissenschaften (Berlin), Physikalisch-mathematische Klasse (in German). 1924: 261–267.
- )
- S2CID 540834.
- ^ Cuneo, Michael (1999-02-18). "Physicists Slow Speed of Light". Harvard Gazette. Retrieved 2023-12-07.
- ^ "Light Changed to Matter, Then Stopped and Moved". www.photonics.com. Retrieved 2023-12-07.
- ISBN 978-0-201-09410-7.
- Bibcode:1916DPhyG..18..318E.
- ISBN 978-0-13-523705-2.
- Bibcode:1916DPhyG..18..318E.
p. 322: Die Konstanten and würden sich direkt berechnen lassen, wenn wir im Besitz einer im Sinne der Quantenhypothese modifizierten Elektrodynamik und Mechanik wären."
- .
- ^ .
- ^ a b .
- ^
S2CID 121928597.
- ^
S2CID 186219228.
- .
- ^
S2CID 119896026.
- S2CID 126244962.
- ISBN 978-0-19-851997-3. Specifically, Born claimed to have been inspired by Einstein's never-published attempts to develop a "ghost-field" theory, in which point-like photons are guided probabilistically by ghost fields that follow Maxwell's equations.
- .
- S2CID 186237037.
- PMID 33266857.
- OCLC 50479292.
- ISBN 978-0-07-032071-0.
- S2CID 118517359.
- ^ ISBN 978-0-521-47814-4.
- ^ Sheldon Glashow Nobel lecture, delivered 8 December 1979.
- ^ Abdus Salam Nobel lecture, delivered 8 December 1979.
- ^ Steven Weinberg Nobel lecture, delivered 8 December 1979.
- ISBN 978-0-521-26092-3.
- .
- .
- .
- .
- S2CID 119437227.
- ISBN 978-0-534-39294-9.
- ISBN 978-0-07-032071-0.
- ISBN 978-0-521-37941-0.
- ISBN 978-3-540-24115-7.
- ^
Naeye, R. (1998). Through the Eyes of Hubble: Birth, Life and Violent Death of Stars. CRC Press. OCLC 40180195.
- ISBN 9780763743871.
- ISBN 978-0-07-050115-7.
- ^ Nobel lecture given by G. Wald on December 12, 1967, online at nobelprize.org: The Molecular Basis of Visual Excitation.
- ISBN 978-1-4200-8243-2.
- ISBN 978-0-262-23048-3.
- S2CID 2414593.
- ISBN 978-0-387-31278-1.
- S2CID 13118587.
- .
- ^
Introductory-level material on the various sub-fields of quantum optics can be found in Fox, M. (2006). Quantum Optics: An introduction. Oxford University Press. ISBN 978-0-19-856673-1– via Google Books.
- ^ Hignett, Katherine (16 February 2018). "Physics creates new form of light that could drive the quantum computing revolution". Newsweek. Retrieved 17 February 2018.
- PMID 29449489.
Further reading
- By date of publication
- Alonso, M.; Finn, E. J. (1968). Fundamental University Physics. Vol. III: Quantum and Statistical Physics. Addison-Wesley. ISBN 978-0-201-00262-1.
- Clauser, J. F. (1974). "Experimental distinction between the quantum and classical field-theoretic predictions for the photoelectric effect". S2CID 118320287.
- Subtle is the Lord: The Science and the Life of Albert Einstein. Oxford University Press.
- ISBN 978-0-691-12575-6.
- Grangier, P.; Roger, G.; Aspect, A. (1986). "Experimental evidence for a photon anticorrelation effect on a beam splitter: A new light on single-photon interferences". S2CID 250837011.
- S2CID 263785760.
- "Special supplemental issue" (PDF). Optics and Photonics News. Vol. 14. October 2003. Archived from the original (PDF) on June 5, 2022.
- Roychoudhuri, C.; Rajarshi, R. (2003). "The nature of light: What is a photon?". Optics and Photonics News. Vol. 14. pp. S1 (Supplement).
- Zajonc, A. (2003). "Light reconsidered". Optics and Photonics News. Vol. 14. pp. S2–S5 (Supplement).
- Loudon, R. (2003). "What is a photon?". Optics and Photonics News. Vol. 14. pp. S6–S11 (Supplement).
- Finkelstein, D. (2003). "What is a photon?". Optics and Photonics News. Vol. 14. pp. S12–S17 (Supplement).
- Muthukrishnan, A.; Scully, M. O.; Zubairy, M. S. (2003). "The concept of the photon – revisited". Optics and Photonics News. Vol. 14. pp. S18–S27 (Supplement).
- Mack, H.; Schleich, Wolfgang P. (2003). "A photon viewed from Wigner phase space". Optics and Photonics News. Vol. 14. pp. S28–S35 (Supplement).
- Glauber, R. (2005). "One Hundred Years of Light Quanta" (PDF). Nobel Prize. Physics Lecture. Archived from the original (PDF) on 2008-07-23. Retrieved 2009-06-29.
- Hentschel, K. (2007). "Light quanta: The maturing of a concept by the stepwise accretion of meaning". Physics and Philosophy. 1 (2): 1–20.
- Education with single photons
- Thorn, J. J.; Neel, M. S.; Donato, V. W.; Bergreen, G. S.; Davies, R. E.; Beck, M. (2004). "Observing the quantum behavior of light in an undergraduate laboratory" (PDF). .
- Bronner, P.; Strunz, Andreas; Silberhorn, Christine; Meyn, Jan-Peter (2009). "Interactive screen experiments with single photons". S2CID 38626417.













































