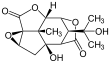
Picrotoxin
Picrotoxinin (left) and picrotin (right) | |||
| Clinical data | |||
|---|---|---|---|
JSmol) | |||
| |||
| |||
| | |||
Picrotoxin, also known as cocculin, is a poisonous crystalline plant compound. It was first isolated by the French pharmacist and chemist Pierre François Guillaume Boullay (1777–1869) in 1812.[1] The name "picrotoxin" is a combination of the Greek words "picros" (bitter) and "toxicon" (poison).[2] A mixture of two different compounds, picrotoxin occurs naturally in the fruit of the Anamirta cocculus plant, although it can also be synthesized chemically.
Due to its interactions with the inhibitory neurotransmitter GABA, picrotoxin acts as a stimulant and convulsant. It mainly impacts the central nervous system, causing seizures and respiratory paralysis in high enough doses.
Chemical structure and synthesis
Picrotoxin is an equimolar mixture of two compounds, picrotoxinin (C15H16O6; CAS# 17617-45-7) and picrotin (C15H18O7; CAS# 21416-53-5).[3] Of the two compounds, picrotin is less active.[4]
Picrotoxin occurs naturally in the fruit of the Anamirta cocculus, a climbing plant from India and other parts of Southeast Asia. The plant is known for its large stems of white wood and sweetly-scented flowers. It produces small stone fruits, Cocculus indicus, which are typically dried.[citation needed]
Currently, there are as many as five total syntheses of picrotoxinin — one of which was published as recently as June 2020.[5] Most syntheses use carvone as a stereochemical template.
![Begin with methyl (1S,4S,5R,7R,8S,9R,10R,11R)-10-(acetyloxy)-7-hydroxy-11-methyl-3-oxo-9-(prop-1-en-2-yl)-4,5-bis[(trimethylsilyl)oxy]-2-oxatricyclo[5.3.1.04,11]undecane-8-carboxylate. (1) Intramolecular transesterification, releasing methyl acetate; then (2) deprotection of a trimethylsilyl-protected vicinal diol, followed by (3) reductive dehydration to an olefin, and (4) stereospecific epoxidation to a glycidic ester](http://upload.wikimedia.org/wikipedia/commons/thumb/9/96/Picrotoxinin_Synthesis.png/500px-Picrotoxinin_Synthesis.png)
In 1988, researchers from Tohoku University in Japan completed a total stereoselective synthesis of both (‑)‑picrotoxinin and (-)-picrotin beginning with (+)‑5β‑hydroxycarvone. In this synthesis, eight asymmetric centers were stereoselectively prepared on a cis-fused hydrindane ring system using several different reactions: a Claisen rearrangement to introduce the quaternary center, an organoselenium-mediated reduction of an epoxy ketone, and a stereospecific construction of a glycidic ester.[7]
The June 2020 synthesis instead employed the quick formation of the polycyclic core, followed by the manipulation of oxidation states of key carbon atoms in order to produce the target molecule.[5]
Some research suggests that picritoxin can be made by the cyclofunctionalization of cycloalkenyl systems. Under kinetically controlled conditions, this process generally results in exo cyclization and forms bridged ring systems like those found in picrotoxin.[8]
Several techniques have been developed to isolate picrotoxinin and picrotin individually. Reaction with the nearby cis alcohol is the key obstruction, and can be inhibited by pretreatment (protection) with trifluoroacetic anhydride in pyridine:[9]

Picrotoxin has also been used as a starting material in several synthetic processes, including the creation of dl-picrotoxadiene, which retains certain features of the picrotoxin skeleton.[10]
Mechanism of action
Some crustacean muscle fibers have excitatory and inhibitory innervation. Picrotoxin blocks inhibition.
Other research suggests that the toxin acts instead as a
Toxicity
Picrotoxin acts as a central nervous system and respiratory stimulant. It is extremely toxic to fish and humans, as well as rodents and other mammals. According to the Register of Toxic Effects of Chemical Substances, the LDLo, or lowest reported lethal dose, is 0.357 mg/kg. Symptoms of picrotoxin poisoning include coughing, difficulty breathing, headache, dizziness, confusion, gastro-intestinal distress, nausea or vomiting, and changes in heart rate and blood pressure. Although especially dangerous if swallowed, systemic effects can also result from inhalation or absorption into the blood stream through lesions in the skin.[16] Picrotoxin also acts as a convulsant. In larger doses, it has been found to induce clonic seizures or cardiac dysrhythmias, with especially high doses ultimately proving fatal, typically due to respiratory paralysis.[17]
Clinical applications and other uses
Due to its toxicity, picrotoxin is now most commonly used as a research tool. However, due to its antagonist effect on GABA receptors, it has been used as a central nervous system stimulant. It was also previously used as an antidote for poisoning by CNS depressants, especially barbiturates.[18]
Although not commonly used, picrotoxin is effective as both a pesticide and a
Despite its potential toxicity to mammals in large enough doses, picrotoxin is also sometimes used as a performance enhancer in horses. It is classified as an illegal "Class I substance" by the American Quarter Horse Association. Substances that are classified as “Class I” are likely to affect performance and have no therapeutic use in equine medicine.[21] In 2010, quarter horse trainer Robert Dimitt was suspended after his horse, Stoli Signature, tested positive for the substance. As with humans, it is used to counteract barbiturate poisoning.[22]
See also
References
- ^ Boullay PF (1812). "Analyse chimique de la Coque du Levant, Menispermum cocculus". Bulletin de Pharmacie (in French). 4: 5–34.
Menispermum cocculus" has been renamed "Anamirta cocculus"
- ^ (Boullay, 1812), p. 31.
- ^ Law V, Knox C, Djoumbou Y, Jewison T, Guo AC, Liu Y, et al. "Picrotoxin". DrugBank. Retrieved April 26, 2017.
- ^ Gammill R, Tulinsky J (1994). "The Chemistry and Pharmacology of GABAA and GABAB Ligands". Current Medicinal Chemistry. 1 (3): 242. Retrieved April 26, 2017.
- ^ PMID 32573211.
- . Retrieved May 7, 2017.
- .
- ISBN 9780080405957. Retrieved May 7, 2017.
- .
- .
- PMID 13529017.
- PMID 8961191.
- ^ Law V, Knox C, Djoumbou Y, Jewison T, Guo AC, Liu Y, et al. "Picrotoxin". DrugBank. Retrieved April 26, 2017.
- PMID 16606858.
- PMID 1317428.
- ^ "Picrotoxin" (PDF). Santa Cruz Biotechnology. Retrieved April 26, 2017.
- ^ "Picrotoxin". Toxnet. U.S. National Laboratory of Medicine. Retrieved April 26, 2017.
- PMID 15432128.
- ISBN 978-3-319-99545-8.
- ^ Bell J (1869). Report of the Committee on the Relations of Alcohol to Medicine. United States: Collins. p. 32.
- ^ "Uniform Classification Guidelines for Foreign Substances and Recommended Penalties and Model Rule" (PDF). Association of Racing Commissioners International, Inc. Retrieved April 26, 2017.
- ^ Lemoreaux P (September 2, 2017). "Two Quarter Horse trainers suspended for drug violations at Prairie Meadows". Daily Racing Form. Daily Racing Form. Retrieved April 26, 2017.
Further reading
- Ehrenberger K, Benkoe E, Felix D (1982). "Suppressive action of picrotoxin, a GABA antagonist, on labyrinthine spontaneous nystagmus and vertigo in man". Acta Oto-Laryngologica. 93 (1–6): 269–73. PMID 7064710.
- Dupont L, Dideberg O, Lamotte-Brasseur J, Angenot L (1976). "Structure cristalline et moléculaire de la picrotoxine, C15H16O6·C15H18O7". Acta Crystallographica B (in French). 32 (11): 2987–2993. hdl:2268/31560.
- Olsen RW, DeLorey TM (1999). "GABA Receptor Physiology and Pharmacology". In Siegel GJ, Agranoff BW, Albers RW, et al. (eds.). Basic Neurochemistry: Molecular, Cellular and Medical Aspects (6th ed.). Philadelphia, PA, USA: Lippincott-Raven.