Piperidine
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Piperidine
| |||
| Preferred IUPAC name
Piperidine[2] | |||
| Other names
Hexahydropyridine
Azacyclohexane Pentamethyleneamine Azinane | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.003.467 | ||
| EC Number |
| ||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 2401 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C5H11N | |||
| Molar mass | 85.150 g·mol−1 | ||
| Appearance | Colorless liquid | ||
| Odor | Semen-like,[3] fishy-ammoniacal, pungent | ||
| Density | 0.862 g/mL | ||
| Melting point | −7 °C (19 °F; 266 K) | ||
| Boiling point | 106 °C (223 °F; 379 K) | ||
| Miscible | |||
| Acidity (pKa) | 11.22 (protonated)[4] | ||
| -64.2·10−6 cm3/mol | |||
| Viscosity | 1.573 cP at 25 °C | ||
| Hazards | |||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H225, H311, H314, H331 | |||
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P271, P280, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P311, P312, P321, P322, P361, P363, P370+P378, P403+P233, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | MSDS1 | ||
| Legal status | |||
| Related compounds | |||
Related compounds
|
Pyridine Pyrrolidine Piperazine Phosphorinane Arsinane | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Piperidine is an
Production
Piperidine was first reported in 1850 by the Scottish chemist Thomas Anderson and again, independently, in 1852 by the French chemist Auguste Cahours, who named it.[9][10][11] Both of them obtained piperidine by reacting piperine with nitric acid.
Industrially, piperidine is produced by the hydrogenation of pyridine, usually over a molybdenum disulfide catalyst:[12]
- C5H5N + 3 H2 → C5H10NH
Pyridine can also be reduced to piperidine via a modified Birch reduction using sodium in ethanol.[13]
Natural occurrence of piperidine and derivatives
Piperidine itself has been obtained from black pepper,[14][15] from Psilocaulon absimile (Aizoaceae),[16] and in Petrosimonia monandra.[17]
The piperidine structural motif is present in numerous natural
Conformation
Piperidine prefers a
 |

|
| axial conformation | equatorial conformation |
Reactions
Piperidine is widely used to convert ketones to enamines.[23] Enamines derived from piperidine are substrates in the Stork enamine alkylation reaction.[24]
Upon treatment with
NMR chemical control
- 13C NMR: (CDCl3, ppm) 47, 27.2, 25.2[citation needed]
- 1H NMR: (CDCl3, ppm) 2.79, 2.19, 1.51[citation needed]
Uses
Piperidine is used as a
A significant industrial application of piperidine is for the production of dipiperidinyl dithiuram tetrasulfide, which is used as an accelerator of the sulfur vulcanization of rubber.[12]
List of piperidine medications
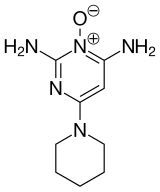
Piperidine and its derivatives are ubiquitous building blocks in pharmaceuticals[26] and fine chemicals. The piperidine structure is found in, for example:
- Icaridin (Insect repellent)
- SSRIs (selective serotonin reuptake inhibitors)
- Stimulants and nootropics:
- Histamine 1 (H1) receptor antagonists/inverse agonists:
- Histamine 3 (H3) receptor antagonists/inverse agonists:
- SERM (selective estrogen receptor modulators)
- Vasodilators
- Antipsychotic medications:
- Opioids:
- Dipipanone
- Fentanyl and analogs
- Loperamide
- Pethidine (meperidine)
- Prodine
- Arylcyclohexylamines:
- PCP and analogs
- chemical weapons
- Ditran
- N-Methyl-3-piperidyl benzilate(JB-336, BZ)
Piperidine is also commonly used in chemical degradation reactions, such as the sequencing of
Piperidine is listed as a Table II precursor under the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances due to its use (peaking in the 1970s) in the clandestine manufacture of phencyclidine.[27]
References
- ^ "International Chemical Safety Card 0317".
- ISBN 978-0-85404-182-4.
- S2CID 19318345.
- .
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-15.
- ^ Frank Johnson Welcher (1947). Organic Analytical Reagents. D. Van Nostrand. p. 149.
- ISBN 978-0-444-52239-9.
- .
- ^ Warnhoff, Edgar W. (1998). "When piperidine was a structural problem" (PDF). Bulletin for the History of Chemistry. 22: 29–34.

- ^ Cahours, Auguste (1852). "Recherches sur un nouvel alcali dérivé de la pipérine" [Investigations of a new alkali derived from piperine]. Comptes Rendus. 34: 481–484.
L'alcali nouveau dérivé de la pipérine, que je désignerai sous le nom de 'pipéridine',… (The new alkali derived from piperine, which I will designate by the name of 'piperidine',…

- ^ ISBN 978-3527306732.
- .
- .
- .
- .
- ^ Juraschewski; Stepanov (1939). J. Gen. Chem. USSR. 9: 1687.
{{cite journal}}: Missing or empty|title=(help) - PMID 16990598.
- ^ Thomas Anderson Henry (1949). The Plant Alkaloids (4th ed.). The Blakiston Company.
- ^ S2CID 98028598.
- .
- .
- ^ Kane, Vinayak V.; Jones, Maitland Jr. (1990). "Spiro[5.7]trideca-1,4-dien-3-one". Organic Syntheses; Collected Volumes, vol. 7, p. 473.
- ISBN 978-0-471-58589-3.
- .
- PMID 25255204.
- ^ "List of Precursors and Chemicals Frequently Used in the Illicit Manufacture of Narcotic Drugs and Psychotropic Substances Under International Control" (PDF). International Narcotics Control Board. Archived from the original (PDF) on 2008-02-27.
External links
 Media related to Piperidine at Wikimedia Commons
Media related to Piperidine at Wikimedia Commons



