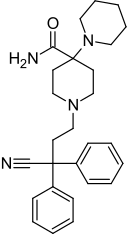
Piritramide
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Dipidolor |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral, IM, IV |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 95%[2] |
| Metabolism | Liver |
| Elimination half-life | 4-10 hours (acute dosing), 17.4 hours (chronic dosing) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Piritramide
Piritramide was developed and patented in Belgium, at Janssen, in 1960. It is part of an eponymous two-member class of opioids in clinical use with the other being
Not being in clinical use in the United States, it is a Schedule I Narcotic controlled substance with a DEA ACSCN of 9642 and manufacturing quota of zero.[7]
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- S2CID 195690966.
- ^ US Patent 3080366
- ^ a b Brayfield A, ed. (23 September 2011). "Piritramide". Martindale: The Complete Drug Reference. Pharmaceutical Press. Retrieved 22 April 2014.
- S2CID 17729725.
- ^ a b c "FACHINFORMATION (Zusammenfassung der Merkmale des Arzneimittels)" [PROFESSIONAL INFORMATION (Summary of Product Characteristics)] (PDF). Janssen. Janssen - Cilag Pharma GmbH. November 2013. Archived from the original (PDF) on 2 May 2014. Retrieved 9 April 2014.
- ^ "DEA Diversion Control Division". Archived from the original on 2017-05-14. Retrieved 2014-05-31.
