Plant disease resistance

Plant disease resistance protects plants from pathogens in two ways: by pre-formed structures and chemicals, and by infection-induced responses of the immune system. Relative to a susceptible plant, disease resistance is the reduction of pathogen growth on or in the plant (and hence a reduction of disease), while the term disease tolerance describes plants that exhibit little disease damage despite substantial pathogen levels. Disease outcome is determined by the three-way interaction of the pathogen, the plant and the environmental conditions (an interaction known as the disease triangle).
Defense-activating compounds can move cell-to-cell and systematically through the plant's vascular system. However, plants do not have circulating immune cells, so most cell types exhibit a broad suite of antimicrobial defenses. Although obvious qualitative differences in disease resistance can be observed when multiple specimens are compared (allowing classification as "resistant" or "susceptible" after infection by the same pathogen strain at similar inoculum levels in similar environments), a gradation of quantitative differences in disease resistance is more typically observed between plant strains or genotypes. Plants consistently resist certain pathogens but succumb to others; resistance is usually specific to certain pathogen species or pathogen strains.
Background
Plant disease resistance is crucial to the reliable production of food, and it provides significant reductions in agricultural use of land, water, fuel and other inputs. Plants in both natural and cultivated populations carry inherent disease resistance, but this has not always protected them.
The late blight
Plant pathogens can spread rapidly over great distances, vectored by water, wind, insects, and humans. Across large regions and many crop species, it is estimated that diseases typically reduce plant yields by 10% every year in more developed nations or agricultural systems, but yield loss to diseases often exceeds 20% in less developed settings.[1]
However, disease control is reasonably successful for most crops. Disease control is achieved by use of plants that have been bred for good resistance to many diseases, and by plant cultivation approaches such as crop rotation, pathogen-free seed, appropriate planting date and plant density, control of field moisture, and pesticide use.
Common disease resistance mechanisms
Pre-formed structures and compounds

- Plant cuticle/surface
- Plant cell walls
- )
- Antimicrobial peptides
- Enzyme inhibitors
- Detoxifying enzymes that break down pathogen-derived toxins
- Receptors that perceive pathogen presence and activate inducible plant defences[3]
Inducible post-infection plant defenses
- Cell wall reinforcement (cellulose, lignin, suberin, callose, cell wall proteins)[4]
- Antimicrobial chemicals, including reactive oxygen species such as phytoalexins such as genisteinor camalexin
- Antimicrobial proteins such as defensins, thionins, or PR-1
- Antimicrobial enzymes such as chitinases, beta-glucanases, or peroxidases[4]
- Hypersensitive response – a rapid host cell death response associated with defence induction.
Immune system
The plant immune system carries two interconnected tiers of receptors, one most frequently sensing molecules outside the cell and the other most frequently sensing molecules inside the cell. Both systems sense the intruder and respond by activating antimicrobial defenses in the infected cell and neighboring cells. In some cases, defense-activating signals spread to the rest of the plant or even to neighboring plants. The two systems detect different types of pathogen molecules and classes of plant receptor proteins.[5][6]
The first tier is primarily governed by
The second tier, primarily governed by R gene products, is often termed effector-triggered immunity (ETI). ETI is typically activated by the presence of specific pathogen "effectors" and then triggers strong antimicrobial responses (see R gene section below).
In addition to PTI and ETI, plant defenses can be activated by the sensing of damage-associated compounds (DAMP), such as portions of the plant cell wall released during pathogenic infection.[9]
Responses activated by PTI and ETI receptors include
Plant immune systems show some mechanistic similarities with the
The term quantitative resistance (discussed below) refers to plant disease resistance that is controlled by multiple genes and multiple molecular mechanisms that each have small effects on the overall resistance trait. Quantitative resistance is often contrasted to ETI resistance mediated by single major-effect R genes.
Pattern-triggered immunity
Effector triggered immunity
Effector triggered immunity (ETI) is activated by the presence of pathogen effectors. The ETI response is reliant on
R genes and R proteins
Plants have evolved
Studied R genes usually confer specificity for particular strains of a pathogen species (those that express the recognized effector). As first noted by Harold Flor in his mid-20th century formulation of the gene-for-gene relationship, a plant R gene has specificity for a pathogen avirulence gene (Avr gene). Avirulence genes are now known to encode effectors. The pathogen Avr gene must have matched specificity with the R gene for that R gene to confer resistance, suggesting a receptor/ligand interaction for Avr and R genes.[10] Alternatively, an effector can modify its host cellular target (or a molecular decoy of that target), and the R gene product (NLR protein) activates defenses when it detects the modified form of the host target or decoy.[6][12]
Effector biology
Effectors are central to the pathogenic or symbiotic potential of microbes and microscopic plant-colonizing animals such as nematodes.[13][14][15] Effectors typically are proteins that are delivered outside the microbe and into the host cell. These colonist-derived effectors manipulate the host's cell physiology and development. As such, effectors offer examples of co-evolution (example: a fungal protein that functions outside of the fungus but inside of plant cells has evolved to take on plant-specific functions). Pathogen host range is determined, among other things, by the presence of appropriate effectors that allow colonization of a particular host.[5] Pathogen-derived effectors are a powerful tool to identify plant functions that play key roles in disease and in disease resistance. Apparently most effectors function to manipulate host physiology to allow disease to occur. Well-studied bacterial plant pathogens typically express a few dozen effectors, often delivered into the host by a Type III secretion apparatus.[13] Fungal, oomycete and nematode plant pathogens apparently express a few hundred effectors.[14][15]
So-called "core" effectors are defined operationally by their wide distribution across the population of a particular pathogen and their substantial contribution to pathogen virulence. Genomics can be used to identify core effectors, which can then be used to discover new R gene alleles, which can be used in plant breeding for disease resistance.
Small RNAs and RNA interference
Plant
Transport of sRNAs during infection is likely facilitated by extracellular vesicles (EVs) and multivesicular bodies (MVBs).
Species-level resistance
In a small number of cases, plant genes are effective against an entire pathogen species, even though that species is pathogenic on other genotypes of that host species. Examples include
Signaling mechanisms
Perception of pathogen presence
Plant defense signaling is activated by the pathogen-detecting receptors that are described in an above section.
Transcription factors and the hormone response
Numerous genes and/or proteins as well as other molecules have been identified that mediate plant defense signal transduction.
Mechanisms of transcription factors and hormones
Plant immune system activity is regulated in part by signaling hormones such as:[34][35]
There can be substantial cross-talk among these pathways.[34]
Regulation by degradation
As with many signal transduction pathways, plant gene expression during immune responses can be regulated by degradation. This often occurs when hormone binding to hormone receptors stimulates ubiquitin-associated degradation of repressor proteins that block expression of certain genes. The net result is hormone-activated gene expression. Examples:[36]
- Auxin: binds to receptors that then recruit and degrade repressors of transcriptional activators that stimulate auxin-specific gene expression.
- Jasmonic acid: similar to auxin, except with jasmonate receptors impacting jasmonate-response signaling mediators such as JAZ proteins.
- Gibberellic acid: Gibberellin causes receptor conformational changes and binding and degradation of Della proteins.
- Ethylene: Inhibitory phosphorylation of the EIN2 ethylene response activator is blocked by ethylene binding. When this phosphorylation is reduced, EIN2 protein is cleaved and a portion of the protein moves to the nucleus to activate ethylene-response gene expression.
Ubiquitin and E3 signaling
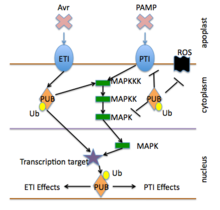
E3 signaling
The E3 ubiquitin ligase enzyme is a main component that provides specificity in protein degradation pathways, including immune signaling pathways.[36] The E3 enzyme components can be grouped by which domains they contain and include several types.[38]
These include the Ring and U-box single subunit, HECT, and CRLs.[39][40] Plant signaling pathways including immune responses are controlled by several feedback pathways, which often include negative feedback; and they can be regulated by De-ubiquitination enzymes, degradation of transcription factors and the degradation of negative regulators of transcription.[36][41]
Quantitative resistance
Differences in plant disease resistance are often incremental or quantitative rather than qualitative. The term quantitative resistance (QR) refers to plant disease resistance that is controlled by multiple genes and multiple molecular mechanisms that each have small or minor effects on the overall resistance trait.[42] QR is important in plant breeding because the resulting resistance is often more durable (effective for more years), and more likely to be effective against most or all strains of a particular pathogen. QR is typically effective against one pathogen species or a group of closely related species, rather than being broadly effective against multiple pathogens.[42] QR is often obtained through plant breeding without knowledge of the causal genetic loci or molecular mechanisms. QR is likely to depend on many of the plant immune system components discussed in this article, as well as traits that are unique to certain plant-pathogen pairings (such as sensitivity to certain pathogen effectors), as well as general plant traits such as leaf surface characteristics or root system or plant canopy architecture. The term QR is synonymous with minor gene resistance.[43]
Adult plant resistance and seedling resistance
Adult plant resistance (APR) is a specialist term referring to quantitative resistance that is not effective in the seedling stage but is effective throughout many remaining plant growth stages.
Plant breeding for disease resistance
Plant breeders emphasize selection and development of disease-resistant plant lines. Plant diseases can also be partially controlled by use of
Breeding for resistance typically includes:
- Identification of plants that may be less desirable in other ways, but which carry a useful disease resistance trait, including wild plant lines that often express enhanced resistance.
- Crossing of a desirable but disease-susceptible variety to a plant that is a source of resistance.
- Growth of breeding candidates in a disease-conducive setting, possibly including pathogen inoculation. Attention must be paid to the specific pathogen isolates, to address variability within a single pathogen species.
- Selection of disease-resistant individuals that retain other desirable traits such as yield, quality and including other disease resistance traits.[46]
Resistance is termed durable if it continues to be effective over multiple years of widespread use as pathogen populations evolve. "Vertical resistance" is specific to certain races or strains of a pathogen species, is often controlled by single R genes and can be less durable. Horizontal or broad-spectrum resistance against an entire pathogen species is often only incompletely effective, but more durable, and is often controlled by many genes that segregate in breeding populations.[2] Durability of resistance is important even when future improved varieties are expected to be on the way: The average time from human recognition of a new fungal disease threat to the release of a resistant crop for that pathogen is at least twelve years.[47][48]
Crops such as potato, apple, banana and sugarcane are often propagated by
Scientific breeding for disease resistance originated with Sir Rowland Biffen, who identified a single recessive gene for resistance to wheat yellow rust. Nearly every crop was then bred to include disease resistance (R) genes, many by introgression from compatible wild relatives.[1]
GM or transgenic engineered disease resistance
The term GM (
A similar strategy was deployed to combat papaya ringspot virus, which by 1994 threatened to destroy Hawaii's papaya industry. Field trials demonstrated excellent efficacy and high fruit quality. By 1998 the first transgenic virus-resistant papaya was approved for sale. Disease resistance has been durable for over 15 years. Transgenic papaya accounts for ~85% of Hawaiian production. The fruit is approved for sale in the U.S., Canada and Japan.
Potato lines expressing viral replicase sequences that confer resistance to potato leafroll virus were sold under the trade names NewLeaf Y and NewLeaf Plus, and were widely accepted in commercial production in 1999–2001, until McDonald's Corp. decided not to purchase GM potatoes and Monsanto decided to close their NatureMark potato business.[51] NewLeaf Y and NewLeaf Plus potatoes carried two GM traits, as they also expressed Bt-mediated resistance to Colorado potato beetle.
No other crop with engineered disease resistance against microbial pathogens had reached the market by 2013, although more than a dozen were in some state of development and testing.
| Publication year | Crop | Disease resistance | Mechanism | Development status |
|---|---|---|---|---|
| 2012 | Tomato | Bacterial spot | R gene from pepper | 8 years of field trials |
| 2012 | Rice | Bacterial blight and bacterial streak |
Engineered E gene | Laboratory |
| 2012 | Wheat | Powdery mildew | Overexpressed R gene from wheat | 2 years of field trials at time of publication |
| 2011 | Apple |
Apple scab fungus | Thionin gene from barley | 4 years of field trials at time of publication |
| 2011 | Potato | Potato virus Y | Pathogen-derived resistance | 1 year of field trial at time of publication |
| 2010 | Apple | Fire blight | Antibacterial protein from moth | 12 years of field trials at time of publication |
| 2010 | Tomato | Multibacterial resistance | PRR from Arabidopsis | Laboratory scale |
| 2010 | Banana | Xanthomonas wilt | Novel gene from pepper | Now in field trial |
| 2009 | Potato | Late blight |
R genes from wild relatives | 3 years of field trials |
| 2009 | Potato | Late blight | R gene from wild relative | 2 years of field trials at time of publication |
| 2008 | Potato | Late blight | R gene from wild relative | 2 years of field trials at time of publication |
| 2008 | Plum | Plum pox virus |
Pathogen-derived resistance | Regulatory approvals, no commercial sales |
| 2005 | Rice | Bacterial streak | R gene from maize | Laboratory |
| 2002 | Barley | Stem rust | Resting lymphocyte kinase (RLK) gene from resistant barley cultivar | Laboratory |
| 1997 | Papaya | Ring spot virus | Pathogen-derived resistance | Approved and commercially sold since 1998, sold into Japan since 2012 |
| 1995 | Squash | Three mosaic viruses | Pathogen-derived resistance | Approved and commercially sold since 1994 |
| 1993 | Potato | Potato virus X | Mammalian interferon-induced enzyme | 3 years of field trials at time of publication |
PRR transfer
Research aimed at engineered resistance follows multiple strategies. One is to transfer useful PRRs into species that lack them. Identification of functional PRRs and their transfer to a recipient species that lacks an orthologous receptor could provide a general pathway to additional broadened PRR repertoires. For example, the
Stacking
The second strategy attempts to deploy multiple NLR genes simultaneously, a breeding strategy known as stacking. Cultivars generated by either DNA-assisted
The avrBs2 effector gene from Xanthomona perforans is the causal agent of bacterial spot disease of pepper and tomato. The first “effector-rationalized” search for a potentially durable R gene followed the finding that avrBs2 is found in most disease-causing Xanthomonas species and is required for pathogen fitness. The Bs2 NLR gene from the wild pepper, Capsicum chacoense, was moved into tomato, where it inhibited pathogen growth. Field trials demonstrated robust resistance without bactericidal chemicals. However, rare strains of Xanthomonas overcame Bs2-mediated resistance in pepper by acquisition of avrBs2 mutations that avoid recognition but retain virulence. Stacking R genes that each recognize a different core effector could delay or prevent adaptation.[1]
More than 50 loci in wheat strains confer disease resistance against wheat stem, leaf and yellow stripe rust pathogens. The Stem rust 35 (Sr35) NLR gene, cloned from a diploid relative of cultivated wheat,
Executor genes
Another class of plant disease resistance genes opens a “trap door” that quickly kills invaded cells, stopping pathogen proliferation. Xanthomonas and
Engineered executor genes were demonstrated by successfully redesigning the pepper Bs3 promoter to contain two additional binding sites for TAL effectors from disparate pathogen strains. Subsequently, an engineered executor gene was deployed in rice by adding five TAL effector binding sites to the Xa27 promoter. The synthetic Xa27 construct conferred resistance against Xanthomonas bacterial blight and bacterial leaf streak species.[1]
Host susceptibility alleles
Most plant pathogens reprogram host gene expression patterns to directly benefit the pathogen. Reprogrammed genes required for pathogen survival and proliferation can be thought of as “disease-susceptibility genes.” Recessive resistance genes are disease-susceptibility candidates. For example, a mutation disabled an Arabidopsis gene encoding
Lr34 is a gene that provides partial resistance to leaf and yellow rusts and powdery mildew in wheat. Lr34 encodes an adenosine triphosphate (ATP)–binding cassette (ABC) transporter. The dominant allele that provides disease resistance was recently found in cultivated wheat (not in wild strains) and, like MLO provides broad-spectrum resistance in barley.[1]
Natural alleles of host translation elongation initiation factors eif4e and eif4g are also recessive viral-resistance genes. Some have been deployed to control potyviruses in barley, rice, tomato, pepper, pea, lettuce and melon. The discovery prompted a successful mutant screen for chemically induced eif4e alleles in tomato.[1]
Natural promoter variation can lead to the evolution of recessive disease-resistance alleles. For example, the recessive resistance gene xa13 in rice is an allele of Os-8N3. Os-8N3 is transcriptionally activated byXanthomonas oryzae pv. oryzae strains that express the TAL effector PthXo1. The xa13 gene has a mutated effector-binding element in its promoter that eliminates PthXo1 binding and renders these lines resistant to strains that rely on PthXo1. This finding also demonstrated that Os-8N3 is required for susceptibility.[1]
Xa13/Os-8N3 is required for pollen development, showing that such mutant alleles can be problematic should the disease-susceptibility phenotype alter function in other processes. However, mutations in the Os11N3 (OsSWEET14) TAL effector–binding element were made by fusing TAL effectors to nucleases (
Gene silencing
Host range
Among the thousands of species of plant pathogenic microorganisms, only a small minority have the capacity to infect a broad range of plant species. Most pathogens instead exhibit a high degree of host-specificity. Non-host plant species are often said to express non-host resistance. The term host resistance is used when a pathogen species can be pathogenic on the host species but certain strains of that plant species resist certain strains of the pathogen species. The causes of host resistance and non-host resistance can overlap. Pathogen host range is determined, among other things, by the presence of appropriate effectors that allow colonization of a particular host.[5] Pathogen host range can change quite suddenly if, for example, the pathogen's capacity to synthesize a host-specific toxin or effector is gained by gene shuffling/mutation, or by horizontal gene transfer.[54] [55]
Epidemics and population biology
Native populations are often characterized by substantial genotype diversity and dispersed populations (growth in a mixture with many other plant species). They also have undergone of plant-pathogen
Monocrop agricultural systems provide an ideal environment for pathogen evolution, because they offer a high density of target specimens with similar/identical genotypes.[56] The rise in mobility stemming from modern transportation systems provides pathogens with access to more potential targets.[56] Climate change can alter the viable geographic range of pathogen species and cause some diseases to become a problem in areas where the disease was previously less important.[56]
These factors make modern agriculture more prone to disease epidemics. Common solutions include constant breeding for disease resistance, use of pesticides, use of border inspections and plant import restrictions, maintenance of significant genetic diversity within the crop gene pool (see crop diversity), and constant surveillance to accelerate initiation of appropriate responses. Some pathogen species have much greater capacity to overcome plant disease resistance than others, often because of their ability to evolve rapidly and to disperse broadly.[56]
Case Study of American Chestnut Blight
Chestnut blight was first noticed in American Chestnut trees that were growing in what is now known as the Bronx Zoo in the year 1904. For years following this incident, it was argued as to what the identity of the pathogen was, as well as the appropriate approach to its control. The earliest attempts to fix the problem on the chestnut involved chemical solutions or physical ones. They attempted to use fungicides, cut limbs off of trees to stop the infection, and completely remove infected trees from habitations to not allow them to infect the others. All of these strategies ended up unsuccessful. Even quarantine measures were put into place which were helped by the passage of Plant Quarantine Act. Chestnut blight still proved to be a huge problem as it rapidly moved through the densely populated forests of chestnut trees. In 1914, the idea was considered to induce blight resistance to the trees through various different means and breeding mechanisms.[57]
See also
- Gene-for-gene relationship
- Induced systemic resistance
- Plant defense against herbivory
- Plant pathology
- Plant use of endophytic fungi in defense
- Systemic acquired resistance
References
- ^ PMID 23950531.
- ^ ISBN 978-0120445653.
- ^ Lutz, Diana (2012). Key part of plants' rapid response system revealed. Washington University in St. Louis.
- ^ PMID 25688916.
- ^ S2CID 8989912.
- ^ PMID 17108957.
- S2CID 21610374.
- ^ PMID 27173932.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 33509925.
- ^ S2CID 32547933.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 21278123.
- S2CID 3810233.
- ^ PMID 22341410.
- ^ PMID 22658704.
- ^ PMID 22809272.
- PMID 18619960.
- PMID 19608454.
- PMID 21153682.
- PMID 24453975.
- PMID 21751852.
- PMID 20573268.
- PMID 22558219.
- PMID 17635765.
- PMID 21995769.
- S2CID 39002035.
- PMID 17905731.
- PMID 15610353.
- S2CID 3377305.
- ISSN 0885-5765.
- PMID 20884801.
- PMID 24092744.
- PMID 12732319.
- PMID 24095918.
- ^ PMID 21841124.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 31415749.
- ^ PMID 22897362.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 20471305.
- PMID 19276192.
- PMID 15579807.
- PMID 20471305.
- S2CID 36185464.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ S2CID 190533925.
- ^ PMID 26047566.
- ^ Bhavani, Sridhar (2021-06-18). "Adult plant resistance (APR): the strategy to beat persistent pathogens". CIMMYT (International Maize and Wheat Improvement Center). Retrieved 2021-07-06.
- S2CID 4603818.
- ^ ISBN 9780123741653.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - S2CID 55486617.
- S2CID 52096158.
- S2CID 205278530.
- S2CID 20940279.
- ^ Kaniewski, Wojciech K.; Thomas, Peter E. (2004). "The Potato Story". AgBioForum. 7 (1&2): 41–46.
- S2CID 7260214– via www.nature.com.
- S2CID 85060938.
- PMID 25566270.
- PMID 24906125.
- ^ S2CID 23726106.
- ^ Barnes, J.C. and Delborne, J.A., 2021. The politics of genetic technoscience for conservation: The case of blight-resistant American chestnut. Environment and Planning E: Nature and Space. 5(3): 1518-1540.
Further reading
- Lucas, J.A., "Plant Defence." Chapter 9 in Plant Pathology and Plant Pathogens, 3rd ed. 1998 Blackwell Science. ISBN 0-632-03046-1
- Hammond-Kosack, K. and Jones, J.D.G. "Responses to plant pathogens." In: Buchanan, Gruissem and Jones, eds. Biochemistry and Molecular Biology of Plants, Second Edition. 2015. Wiley-Blackwell, Hoboken, NJ. ISBN 9780470714218
- Dodds, P.; Rathjen, J. (2010). "Plant immunity: towards an integrated view of plant–pathogen interactions". Nature Reviews Genetics. 11 (8): 539–548. S2CID 8989912.
- Michelmore, Richard W.; Christopoulou, Marilena; Caldwell, Katherine S. (2013-08-04). "Impacts of Resistance Gene Genetics, Function, and Evolution on a Durable Future". S2CID 22234708.
- Schumann, G. Plant Diseases: Their Biology and Social Impact. 1991 APS Press, St. Paul, MN. ISBN 0890541167
