Platinum(IV) chloride

| |

| |
| Names | |
|---|---|
| IUPAC name
Platinum tetrachloride
| |
| Other names
Platinum(IV) chloride
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.033.300 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| PtCl4 | |
| Molar mass | 336.89 g/mol |
| Appearance | brown-red powder |
| Density | 4.303 g/cm3 (anhydrous) 2.43 g/cm3 (pentahydrate) |
| Melting point | 370 °C (698 °F; 643 K) (decomposes) |
| 58.7 g/100 mL (anhydrous) very soluble (pentahydrate) | |
| Solubility | anhydrous soluble in acetone slightly soluble in ethanol insoluble in ether pentahydrate soluble in alcohol, ether |
| −93.0·10−6 cm3/mol | |
| Structure | |
| Square planar | |
| Hazards | |
| GHS labelling:[1] | |
   
| |
| Danger | |
| H290, H301, H314, H317, H334 | |
| P234, P260, P261, P264, P270, P272, P280, P285, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P304+P341, P305+P351+P338, P310, P321, P330, P333+P313, P342+P311, P363, P390, P404, P405, P501 | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
276 mg/kg (rat, oral) |
| Related compounds | |
Other anions
|
Platinum(IV) sulfide
|
Other cations
|
Iridium(IV) chloride
|
Related compounds
|
Platinum(VI) fluoride
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Platinum(IV) chloride is the inorganic compound of platinum and chlorine with the empirical formula PtCl4. This brown solid features platinum in the 4+ oxidation state.
Structure
Typical of Pt(IV), the metal centers adopt an
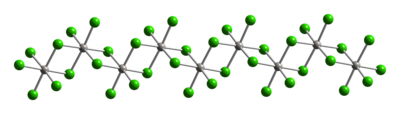
|
| Part of a (PtCl4)∞ chain from the crystal structure of platinum(IV) chloride |
Formation and reactions
PtCl4 is mainly encountered in the handling of chloroplatinic acid, obtained by dissolving of Pt metal in aqua regia. Heating H2PtCl6 to 220 °C gives impure PtCl4:[3]
- H2PtCl6 → PtCl4 + 2 HCl
A purer product can be produced by heating under chlorine gas at 250 °C.[4]
If excess acids are removed, PtCl4 crystallizes from aqueous solutions in large red crystals of pentahydrate PtCl4·5(H2O),[5] which can be dehydrated by heating to about 300 °C in a current of dry chlorine. The pentahydrate is stable and is used as the commercial form of PtCl4.
Treatment of PtCl4 with aqueous base gives the [Pt(OH)6]2− ion. With methyl Grignard reagents followed by partial hydrolysis, PtCl4 converts to the cuboidal cluster [Pt(CH3)3(OH)]4.[6] Upon heating PtCl4 evolves chlorine to give PtCl2:
- PtCl4 → PtCl2 + Cl2
The heavier halides, PtBr4 and PtI4, are also known.
References
- Cotton, S. A. Chemistry of Precious Metals, Chapman and Hall (London): 1997. ISBN 0-7514-0413-6.
- ^ "Platinum tetrachloride". pubchem.ncbi.nlm.nih.gov. Retrieved 27 December 2021.
- .
- .
- ISBN 978-3-432-02328-1.
- ^ George Samuel Newth (1920). A text-book of inorganic chemistry. Longmans, Green, and co. p. 694.
- ISBN 0-7506-3365-4.
