Cell potency
Cell potency is a cell's ability to differentiate into other cell types.[1][2] The more cell types a cell can differentiate into, the greater its potency. Potency is also described as the gene activation potential within a cell, which like a continuum, begins with
Totipotency
Totipotency (Latin: totipotentia, lit. 'ability for all [things]') is the ability of a single cell to divide and produce all of the differentiated cells in an organism. Spores and zygotes are examples of totipotent cells.[3] In the spectrum of cell potency, totipotency represents the cell with the greatest differentiation potential, being able to differentiate into any embryonic cell, as well as any extraembryonic tissue cell. In contrast, pluripotent cells can only differentiate into embryonic cells.[4][5]
A fully differentiated cell can return to a state of totipotency.
The human development model can be used to describe how totipotent cells arise.
Research on
Primordial germ cells
In mouse primordial
Pluripotency
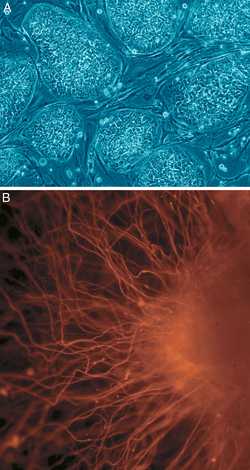
B: Nerve cells
In cell biology, pluripotency (Latin: pluripotentia, lit. 'ability for many [things]') However, cell pluripotency is a continuum, ranging from the completely pluripotent cell that can form every cell of the embryo proper, e.g., embryonic stem cells and iPSCs, to the incompletely or partially pluripotent cell that can form cells of all three germ layers but that may not exhibit all the characteristics of completely pluripotent cells.
Induced pluripotency
Induced pluripotent stem cells, commonly abbreviated as iPS cells or iPSCs, are a type of pluripotent
Due to their great similarity to ESCs, the medical and research communities are interested iPSCs. iPSCs could potentially have the same therapeutic implications and applications as ESCs but without the controversial use of embryos in the process, a topic of great bioethical debate. The induced pluripotency of somatic cells into
Somatic expression of combined transcription factors can directly induce other defined somatic cell fates (transdifferentiation); researchers identified three neural-lineage-specific transcription factors that could directly convert mouse fibroblasts (connective tissue cells) into fully functional neurons.[27] This result challenges the terminal nature of cellular differentiation and the integrity of lineage commitment; and implies that with the proper tools, all cells are totipotent and may form all kinds of tissue.
Some of the possible medical and therapeutic uses for iPSCs derived from patients include their use in cell and tissue transplants without the risk of rejection that is commonly encountered. iPSCs can potentially replace animal models unsuitable as well as in vitro models used for disease research.[28]

Naive vs. primed pluripotency states
Findings with respect to epiblasts before and after implantation have produced proposals for classifying pluripotency into two states: "naive" and "primed", representing pre- and post-implnatation epiblast, respectively.[29] Naive-to-primed continuum is controlled by reduction of Sox2/Oct4 dimerization on SoxOct DNA elements controlling naive pluripotency.[30] Primed pluripotent stem cells from different species could be reset to naive state using a cocktail containing Klf4 and Sox2 or "super-Sox" − a chimeric transcription factor with enhanced capacity to dimerize with Oct4.[30]
The baseline stem cells commonly used in science that are referred as embryonic stem cells (ESCs) are derived from a pre-implantation epiblast; such epiblast is able to generate the entire fetus, and one epiblast cell is able to contribute to all cell lineages if injected into another blastocyst. On the other hand, several marked differences can be observed between the pre- and post-implantation epiblasts, such as their difference in morphology, in which the epiblast after implantation changes its morphology into a cup-like shape called the "egg cylinder" as well as chromosomal alteration in which one of the X-chromosomes under random inactivation in the early stage of the egg cylinder, known as
Another major difference is that post-implantation epiblast stem cells are unable to contribute to blastocyst
Native pluripotency in plants
Un-induced pluripotency has been observed in
Multipotency

Multipotency is when
Research related to multipotent cells suggests that multipotent cells may be capable of conversion into unrelated cell types. In another case, human umbilical cord blood stem cells were converted into human neurons.[38] There is also research on converting multipotent cells into pluripotent cells.[39]
Multipotent cells are found in many, but not all human cell types. Multipotent cells have been found in
MSCs may prove to be a valuable source for stem cells from molars at 8–10 years of age, before adult dental calcification. MSCs can differentiate into osteoblasts, chondrocytes, and adipocytes.[44]
Oligopotency
In biology, oligopotency is the ability of progenitor cells to differentiate into a few
Unipotency
In
See also
References
- ^ Cell totipotency was discovered by Habertland and the term was coined by Thomas Hund Morgan.
Mahla RS (2016). "Stem Cells Applications in Regenerative Medicine and Disease Therapeutics". International Journal of Cell Biology. 2016 (7): 6940283. PMID 27516776.
- ^ ISBN 978-0-7546-5755-2.
- PMID 19343304.
- ISBN 978-1319067748.
- ^ "What is the difference between totipotent, pluripotent, and multipotent?".
- PMID 19412894.
- PMID 21236679.
- PMID 22722858.
- PMID 23746443.
- S2CID 837230.
- S2CID 16988032.
- PMID 8583008.
- S2CID 130017.
- PMID 18414021.
- PMID 20595612.
- PMID 23223451.
- ^ "Biology Online". Biology-Online.org. Retrieved 25 April 2013.
- ISBN 978-3540237358.
- ^ .
- PMID 20952534.
- S2CID 1565219.
- ^ "The Nobel Prize in Physiology or Medicine 2012". Nobelprize.org. Nobel Media AB 2013. Web. 28 Nov 2013.
- S2CID 8531539.
- ^ PMID 23247625.
- PMID 32402162.
- ^ Choi, Charles. "Cell-Off: Induced Pluripotent Stem Cells Fall Short of Potential Found in Embryonic Version". Scientific American. Retrieved 25 April 2013.
- PMID 20107439.
- S2CID 13321484.
- PMID 19497275.
- ^ .
- PMID 15145348.
- S2CID 16264083.
- S2CID 17685207.
- S2CID 14128314.
- S2CID 4365390.
- PMID 22669820.
- S2CID 73498853.
- PMID 22814375.
- S2CID 4350560.
- ^
Zhao Y, Mazzone T (December 2010). "Human cord blood stem cells and the journey to a cure for type 1 diabetes". Autoimmunity Reviews. 10 (2): 103–107. PMID 20728583.
- S2CID 36604144.
- S2CID 15588806.
- PMID 22242308.
- S2CID 3347616.
- ^ Ibelgaufts, Horst. "Cytokines & Cells Online Pathfinder Encyclopedia". Retrieved 25 April 2013.
- ISBN 978-1-947172-04-3.
- ^ "hepatoblast differentiation". GONUTS. Archived from the original on 2016-03-03. Retrieved 2012-08-31.
