Podophyllotoxin
 | |
| Clinical data | |
|---|---|
| Trade names | Condylox,[1] Wartec, others |
| Other names | (5R,5aR,8aR,9R)-9-hydroxy-5-(3,4,5-trimethoxyphenyl)-5,8,8a,9-tetrahydrofuro[3',4':6,7]naphtho[2,3-d] [1,3]dioxol-6(5aH)-one |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a684055 |
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 1.0 to 4.5 hours. |
| Identifiers | |
| |
JSmol) | |
| Melting point | 183.3 to 184 °C (361.9 to 363.2 °F) |
| |
| |
| | |
Podophyllotoxin (PPT) is the active ingredient in Podofilox, which is a medical cream that is used to treat
It is a non-alkaloid toxin lignin extracted from the roots and rhizomes of Podophyllum species.[3] A less refined form known as podophyllum resin is also available, but has greater side effects.[4][5]
Podophyllotoxin was first isolated in pure form in 1880 by Valerian Podwyssotzki (1818 – 28 January 1892), a Polish-Russian privatdozent at the University of Dorpat (now: Tartu, Estonia) and assistant at the Pharmacological Institute there.[6][7][8]
It is on the World Health Organization's List of Essential Medicines.[9]
Medical uses
Podophyllotoxin possesses a large number of medical applications, as it is able to stop replication of both cellular and viral DNA by binding necessary enzymes. It can additionally destabilize microtubules and prevent cell division. Because of these interactions it is considered an antimitotic drug, although modern medicine instead use less orally toxic derivatives when such effect is wanted.[10]
Podophyllotoxin cream is commonly prescribed as a potent topical antiviral.[11] It is used for the treatment of HPV infections with external warts as well as molluscum contagisum infections.[11] 0.5% PPT cream is prescribed for twice daily applications for 3 days followed by 4 days with no application, this weekly cycle is repeated for 4 weeks.[12] It can also be prescribed as a gel, as opposed to cream. PPT is also sold under the names condyline and warticon.[13]
Adverse effects
The most common side effects of podophyllotoxin cream are typically limited to irritation of tissue surrounding the application site, including burning, redness, pain, itching, swelling.[14] Application can be immediately followed by burning or itching. Small sores, itching and peeling skin can also follow, for these reasons it is recommended that application be done in a way that limits contact with surrounding, uninfected tissue[15]
Neither podophyllin resin nor podophyllotoxin lotions or gels are used during pregnancy because these medications have been shown to be embryotoxic in both mice and rats. Additionally, antimitotic agents are not typically recommended during pregnancy.[16] Additionally, it has not been determined if podophyllotoxin can pass into breast milk from topical applications and therefore it is not recommended for breastfeeding women.[17]
Podophyllotoxin cream is safe for topical use; however, it can cause CNS depression as well as enteritis if ingested. The podophyllum resin from which podophyllotoxin is derived has the same effect.[18]
Mechanism of action
This section may require cleanup to meet Wikipedia's quality standards. The specific problem is: Poorly organized. Seems to self-contradict with regard to Topo II inhibition. (August 2022) |
Podophyllotoxin destabilizes microtubules by binding tubulin and thus preventing cell division.
Furthermore, podophyllotoxin is classified as an arytetralin lignan for[dubious ] its ability to bind and deactivate DNA.[28] It and its derivates bind Topo II and prevent its ability to catalyze rejoining of DNA that has been broken for replication. Lastly, experimental evidence has shown that these arytetralin lignans can interact with cellular factors to create chemical DNA adducts, thus further deactivating DNA.[28]
Chemistry
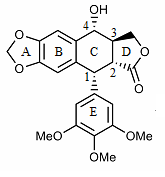
Structural characteristic
The structure of podophyllotoxin was first elucidated in the 1930s.
Derivatives of podophyllotoxin are synthesized as properties of the rings and carbon 1 through 4 are diversified. For example, ring A is not essential to antimitotic activity. Aromatization of ring C leads to loss of activity, possibly from ring E no longer being placed on the axial position. In addition, the stereochemistry at C-2 and C-3 configures a trans-lactone, which has more activity than the cis counterpart. Chirality at C-1 is also important as it implies an axial position for ring E.[30]
Biosynthesis
The biosynthetic route of podophyllotoxin was not completely eludicidated for many years; however, in September 2015, the identity of the six missing enzymes in podophyllotoxin biosynthesis were reported for the first time.
A sequence of enzymes involved has been reported to be
Chemical synthesis
Podophyllotoxin has been successfully synthesized in a laboratory; however, synthesis mechanisms require many steps, resulting in a low overall yield. It therefore remains more efficient to obtain podophyllotoxin from natural sources.[32]
Four routes have been used to synthesize podophyllotoxin with varying success: an oxo ester route,[33] lactonization of a dihydroxy acid,[34] cyclization of a conjugate addition product,[35] and a Diels-Alder reaction.[36]
Derivatives
Podophyllotoxin and its derivatives are used as
Natural abundance
Podophyllotoxin is present at concentrations of 0.3% to 1.0% by mass in the
It is
References
- ^ "What is Condylox?" (in German). Retrieved 2018-08-15.
- ^ a b c "Podofilox". The American Society of Health-System Pharmacists. Retrieved 8 December 2016.
- PMID 19149581.
- ^ "Podophyllum Resin". The American Society of Health-System Pharmacists. Retrieved 8 December 2016.
- ISBN 9789241547659.
- ^ See:
- Podwyssotzki V (1880). "Pharmakologische Studien über Podophyllum peltatum" [Pharmacological studies of Podophyllum peltatum]. Archiv für experimentelle Pathologie und Pharmakologie (in German). 13 (1–2): 29–52. S2CID 33803976. Podwyssotzki named podophyllotoxin on p. 30: " … ich nenne sie bis auf weiteres Podophyllotoxin." ( … for the time being I call it "podophyllotoxin".)
- Podwyssotzki V (10 September 1881). "The active constituents of podophyllotoxin". Pharmaceutical Journal and Transactions. 3rd series. 12: 217–219.
- Podwyssotzki V (March 1882). "On the active constituents of podophyllin". American Journal of Pharmacy. 54: 102–115.
- Podwyssotzki V (1882). "Über die wirksamen Bestandtheile des Podophyllins" [On the active components of podophyllin]. Berichte der Deutschen Chemischen Gesellschaft (in German). 15: 377–378.
- Obituary: Kobert R (February 1893). "Valerian Podwyssotzki". Bulletin of Pharmacy. 7 (2): 49–52.
- Podwyssotzki V (1880). "Pharmakologische Studien über Podophyllum peltatum" [Pharmacological studies of Podophyllum peltatum]. Archiv für experimentelle Pathologie und Pharmakologie (in German). 13 (1–2): 29–52.
- ISBN 9781439813836.
- PMID 33921719.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ PMID 15302526.
- ^ PMID 28884079.
- ^ "Podofilox Monograph for Professionals - Drugs.com". Drugs.com. Retrieved 2018-05-06.
- ^ "Podophyllotoxin for anogenital warts; Podophyllotoxin info". patient.info. Retrieved 2018-05-06.
- PMID 11350195.
- ^ "PRODUCT INFORMATION WARTEC® SOLUTION" (PDF). GlaxoSmithKline Australia Pty Ltd. Retrieved 6 January 2013.
- PMID 2742385.
- ^ "Podophyllotoxin | DermNet New Zealand". www.dermnetnz.org. Retrieved 2018-05-06.
- PMID 458391.
- S2CID 2231369.
- PMID 11102564.
- ^ PMID 10872202.
- PMID 7054166.
- ^ PMID 6831403.
- PMID 6848174.
- PMID 2568357.
- PMID 2568584.
- ISBN 978-0-07-178923-3.
- ^ PMID 11562272.
- .
- ^ PMID 15892669.
- ^ PMID 26359402.
- ^ PMID 10872202.
- .
- .
- .
- .
- S2CID 247493530.
- PMID 24827545.
- .
Further reading
- Kelly M, Hartwell JL (February 1954). "The biological effects and the chemical composition of podophyllin: a review". Journal of the National Cancer Institute. 14 (4): 967–1010. PMID 13233838.
- Hartwell JL, Schrecker AW (1951). "Components of Podophyllin. V. The Constitution of Podophyllotoxin". .