Polyethylene

| |

| |
| Names | |
|---|---|
| IUPAC name
Polyethene or poly(methylene)[1]
| |
| Other names
Polyethylene
Polythene | |
| Identifiers | |
| Abbreviations | PE |
| ChemSpider |
|
ECHA InfoCard
|
100.121.698 |
| KEGG | |
| MeSH | Polyethylene |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C2H4)n | |
| Density | 0.88–0.96 g/cm3[2] |
| Melting point | 115–135 °C (239–275 °F; 388–408 K)[2] |
| Not soluble | |
| log P | 1.02620[3] |
| −9.67×10−6 (HDPE, SI, 22 °C)[4] | |
| Thermochemistry | |
Std enthalpy of (ΔfH⦵298)formation |
−28 to −29 kJ/mole[5] |
| 650-651 kJ/mole, 46 MJ/kg[5] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

| Part of a series on |
| Fiber |
|---|
 |
| Natural fibers |
|
Human-made fibers |
|
Polyethylene or polythene (abbreviated PE;
Many kinds of polyethylene are known, with most having the chemical formula (C2H4)n. PE is usually a mixture of similar polymers of ethylene, with various values of n. It can be low-density or high-density and many variations thereof. Its properties can be modified further by crosslinking or copolymerization. All forms are nontoxic as well as chemically resilient. As a consequence of these factors, PE is used on an immense scale. Its widespread use is also problematic because it resists biodegradation. Like the hydrocarbon it is, PE is white or colorless and combustible.
History
Polyethylene was first synthesized by the German chemist Hans von Pechmann, who prepared it by accident in 1898 while investigating diazomethane.[10][a][11][b] When his colleagues Eugen Bamberger and Friedrich Tschirner characterized the white, waxy substance that he had created, they recognized that it contained long −CH2− chains and termed it polymethylene.[12]
The first industrially practical polyethylene synthesis (diazomethane is a notoriously unstable substance that is generally avoided in industrial syntheses) was again accidentally discovered in 1933 by Eric Fawcett and Reginald Gibson at the
The landmark breakthrough in the commercial production of polyethylene began with the development of
Properties
The properties of polyethylene depends strongly on type. The molecular weight, crosslinking, and presence of comonomers all strongly affect its properties. It is for this structure-property relation that intense effort has been invested into diverse kinds of PE.[7][17] LDPE is softer and more transparent than HDPE. For medium- and high-density polyethylene the melting point is typically in the range 120 to 130 °C (248 to 266 °F). The melting point for average commercial low-density polyethylene is typically 105 to 115 °C (221 to 239 °F). These temperatures vary strongly with the type of polyethylene, but the theoretical upper limit of melting of polyethylene is reported to be 144 to 146 °C (291 to 295 °F). Combustion typically occurs above 349 °C (660 °F).
Most
Polyethylene absorbs almost no
Polyethylene burns slowly with a blue flame having a yellow tip and gives off an odour of paraffin (similar to candle flame). The material continues burning on removal of the flame source and produces a drip.[18]
Polyethylene cannot be imprinted or bonded with adhesives without pretreatment. High-strength joints are readily achieved with plastic welding.
Electrical
Polyethylene is a good electrical insulator. It offers good electrical treeing resistance; however, it becomes easily electrostatically charged (which can be reduced by additions of graphite, carbon black or antistatic agents). When pure, the
Optical
Depending on thermal history and film thickness, PE can vary between almost clear (
Manufacturing process
Monomer
The ingredient or
2−) connected to each other. Typical specifications for PE purity are <5 ppm for water, oxygen, and other alkenes contents. Acceptable contaminants include N2, ethane (common precursor to ethylene), and methane. Ethylene is usually produced from petrochemical sources, but also is generated by dehydration of ethanol.[7]
Polymerization
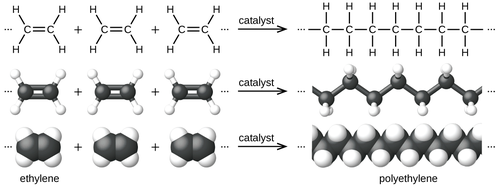
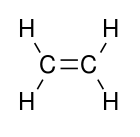
Polymerization of ethylene to polyethylene is described by the following chemical equation:
Ethylene is a stable molecule that polymerizes only upon contact with catalysts. The conversion is highly
Joining
Commonly used methods for joining polyethylene parts together include:[22]
- Welding
- Hot gas welding
- Infrared welding
- Laser welding
- Ultrasonic welding
- Heat sealing
- Heat fusion
- Fastening
- Adhesives[22]
- Pressure-sensitive adhesive (PSAs)
- Dispersion of solvent-type PSAs
- Polyurethanecontact adhesives
- Two-part polyurethane
- Epoxy adhesives
- Hot-melt adhesives
- nonpolarand has a high resistance to solvents.
- Pressure-sensitive adhesive (PSAs)
Pressure-sensitive adhesives (PSA) are feasible if the surface chemistry or charge is modified with plasma activation, flame treatment, or corona treatment.
Classification
Polyethylene is classified by its
- Ultra-high-molecular-weight polyethylene (UHMWPE)
- Ultra-low-molecular-weight polyethylene (ULMWPE or PE-WAX)
- High-molecular-weight polyethylene (HMWPE)
- High-density polyethylene (HDPE)
- High-density cross-linked polyethylene (HDXLPE)
- Cross-linked polyethylene (PEX or XLPE)
- Medium-density polyethylene (MDPE)
- Linear low-density polyethylene (LLDPE)
- Low-density polyethylene (LDPE)
- Very-low-density polyethylene (VLDPE)
- Chlorinated polyethylene (CPE)
With regard to sold volumes, the most important polyethylene grades are HDPE, LLDPE, and LDPE.
Ultra-high-molecular-weight (UHMWPE)
UHMWPE is polyethylene with a molecular weight numbering in the millions, usually between 3.5 and 7.5 million
High-density (HDPE)

HDPE is defined by a density of greater or equal to 0.941 g/cm3. HDPE has a low degree of branching. The mostly linear molecules pack together well, so intermolecular forces are stronger than in highly branched polymers. HDPE can be produced by
HDPE has high tensile strength. It is used in products and packaging such as milk jugs, detergent bottles, butter tubs, garbage containers, and water pipes.
Cross-linked (PEX or XLPE)
PEX is a medium- to high-density polyethylene containing
Medium-density (MDPE)
MDPE is defined by a density range of 0.926–0.940 g/cm3. MDPE can be produced by chromium/silica catalysts, Ziegler–Natta catalysts, or metallocene catalysts. MDPE has good shock and drop resistance properties. It also is less notch-sensitive than HDPE; stress-cracking resistance is better than HDPE. MDPE is typically used in gas pipes and fittings, sacks, shrink film, packaging film, carrier bags, and screw closures.
Linear low-density (LLDPE)
LLDPE is defined by a density range of 0.915–0.925 g/cm3. LLDPE is a substantially linear polymer with significant numbers of short branches, commonly made by
Low-density (LDPE)
LDPE is defined by a density range of 0.910–0.940 g/cm3. LDPE has a high degree of short- and long-chain branching, which means that the chains do not pack into the
The radical polymerization process used to make LDPE does not include a catalyst that "supervises" the radical sites on the growing PE chains. (In HDPE synthesis, the radical sites are at the ends of the PE chains, because the catalyst stabilizes their formation at the ends.) Secondary
Very-low-density (VLDPE)
VLDPE is defined by a density range of 0.880–0.915 g/cm3. VLDPE is a substantially linear polymer with high levels of short-chain branches, commonly made by copolymerization of ethylene with short-chain alpha-olefins (for example, 1-butene, 1-hexene and 1-octene). VLDPE is most commonly produced using metallocene catalysts due to the greater co-monomer incorporation exhibited by these catalysts. VLDPEs are used for hose and tubing, ice and frozen food bags, food packaging and stretch wrap as well as impact modifiers when blended with other polymers.
Much research activity has focused on the nature and distribution of long chain branches in polyethylene. In HDPE, a relatively small number of these branches, perhaps one in 100 or 1,000 branches per backbone carbon, can significantly affect the rheological properties of the polymer.
Copolymers
In addition to
Types of polyethylenes
The particular material properties of "polyethylene" depend on its molecular structure. Molecular weight and crystallinity are the most significant factors; crystallinity in turn depends on molecular weight and degree of branching. The less the polymer chains are branched, and the lower the molecular weight, the higher the crystallinity of polyethylene. Crystallinity ranges from 35% (PE-LD/PE-LLD) to 80% (PE-HD). Polyethylene has a density of 1.0 g/cm3 in crystalline regions and 0.86 g/cm3 in amorphous regions. An almost linear relationship exists between density and crystallinity.[17]
The degree of branching of the different types of polyethylene can be schematically represented as follows:[17]
| PE-HD | |
| PE-LLD | |
| PE-LD | 
|
The figure shows polyethylene backbones, short-chain branches and side-chain branches. The polymer chains are represented linearly.
Chain branches
The properties of polyethylene are highly dependent on type and number of chain branches. The chain branches in turn depend on the process used: either the high-pressure process (only PE-LD) or the low-pressure process (all other PE grades). Low-density polyethylene is produced by the high-pressure process by radical polymerization, thereby numerous short chain branches as well as long chain branches are formed. Short chain branches are formed by intramolecular chain transfer reactions, they are always butyl or ethyl chain branches because the reaction proceeds after the following mechanism:
Environmental issues


The widespread usage of polyethylene poses potential difficulties for waste management because it is not readily biodegradable.
In Japan, getting rid of plastics in an environmentally friendly way was the major problem discussed until the
In 2010, a Japanese researcher, Akinori Ito, released the prototype of a machine which creates oil from polyethylene using a small, self-contained vapor distillation process.[25]
Biodegradability
Polyethylene, like other synthetic plastics, is not readily biodegradable, and thus accumulates in landfills. However, there are a number of species of bacteria and animals that are able to degrade polyethylene.
In May 2008, Daniel Burd, a 16-year-old Canadian, won the Canada-Wide Science Fair in Ottawa after discovering that Pseudomonas fluorescens, with the help of Sphingomonas, can degrade over 40% of the weight of plastic bags within six weeks. He later guessed that it would be gone after six more weeks.[26]
The thermophilic bacterium Brevibacillus borstelensis (strain 707) was isolated from a soil sample and found to use low-density polyethylene as a sole carbon source when incubated together at 50 °C. Biodegradation increased with time exposed to ultraviolet radiation.[27]
Acinetobacter sp. 351 can degrade lower molecular-weight PE oligomers. When PE is subjected to thermo- and photo-oxidization, products including alkanes, alkenes, ketones, aldehydes, alcohols, carboxylic acid, keto-acids, dicarboxylic acids, lactones, and esters are released.[28]
In 2014, a Chinese researcher named Jun Yang discovered that
In 2017, researchers reported that the caterpillar of Galleria mellonella eats plastic garbage such as polyethylene.[31][32] The caterpillar is able to digest polyethylene due to a combination of its gut microbiota[33] and its saliva containing enzymes that oxidise and depolymerise the plastic.[34]
Climate change
When exposed to ambient solar radiation the plastic produces two greenhouse gases, methane and ethylene. The plastic type which releases gases at the highest rate is low-density polyethylene (LDPE). Due to its low density it breaks down more easily over time, leading to higher surface areas. The production of these trace gases from virgin LDPE increase over time. When incubated in air, LDPE emits gases at rates ~2 times and ~76 times higher in comparison to incubation in water for methane and ethylene, respectively. However, based on the rates measured in the study methane production by plastics is presently an insignificant component of the global methane budget.[35]
Chemically modified polyethylene
Polyethylene may either be modified in the polymerization by
Non-polar ethylene copolymers
α-olefins
In the low pressure process
Metallocene polyethylene (PE-MC)
Metallocene polyethylene (PE-M) is prepared by means of
Polyethylene with multimodal molecular weight distribution
Polyethylene with multimodal molecular weight distribution consists of several polymer fractions, which are homogeneously mixed. Such polyethylene types offer extremely high stiffness, toughness, strength, stress crack resistance and an increased crack propagation resistance. They consist of equal proportions higher and lower molecular polymer fractions. The lower molecular weight units crystallize easier and relax faster. The higher molecular weight fractions form linking molecules between crystallites, thereby increasing toughness and stress crack resistance. Polyethylene with multimodal molecular weight distribution can be prepared either in two-stage reactors, by catalysts with two active centers on a carrier or by blending in extruders.[17]: 238
Cyclic olefin copolymers (COC)
Polar ethylene copolymers
The basic compounds used as polar comonomers are vinyl alcohol (
Ethylene copolymers with unsaturated alcohols
Ethylene/vinyl alcohol copolymer (EVOH) is (formally) a copolymer of PE and vinyl alcohol (ethenol), which is prepared by (partial) hydrolysis of ethylene-vinyl acetate copolymer (as vinyl alcohol itself is not stable). However, typically EVOH has a higher comonomer content than the VAC commonly used.[37]: 239
EVOH is used in multilayer films for packaging as a barrier layer (barrier plastic). As EVOH is hygroscopic (water-attracting), it absorbs water from the environment, whereby it loses its barrier effect. Therefore, it must be used as a core layer surrounded by other plastics (like LDPE, PP, PA or PET). EVOH is also used as a coating agent against corrosion at street lights, traffic light poles and noise protection walls.[37]: 239
Ethylene/acrylic acid copolymers (EAA)
Copolymer of ethylene and unsaturated carboxylic acids (such as acrylic acid) are characterized by good adhesion to diverse materials, by resistance to stress cracking and high flexibility.[38] However, they are more sensitive to heat and oxidation than ethylene homopolymers. Ethylene/acrylic acid copolymers are used as adhesion promoters.[17]
If salts of an unsaturated carboxylic acid are present in the polymer, thermo-reversible ion networks are formed, they are called ionomers. Ionomers are highly transparent thermoplastics which are characterized by high adhesion to metals, high abrasion resistance and high water absorption.[17]
Ethylene copolymers with unsaturated esters
If unsaturated esters are copolymerized with ethylene, either the alcohol moiety may be in the polymer backbone (as it is the case in ethylene-vinyl acetate copolymer) or of the acid moiety (e. g. in ethylene-ethyl acrylate copolymer). Ethylene-vinyl acetate copolymers are prepared similarly to LD-PE by high pressure polymerization. The proportion of comonomer has a decisive influence on the behaviour of the polymer.
The density decreases up to a comonomer share of 10% because of the disturbed crystal formation. With higher proportions it approaches to the one of polyvinyl acetate (1.17 g/cm3).[37]: 235 Due to decreasing crystallinity ethylene vinyl acetate copolymers are getting softer with increasing comonomer content. The polar side groups change the chemical properties significantly (compared to polyethylene):[17]: 224 weather resistance, adhesiveness and weldability rise with comonomer content, while the chemical resistance decreases. Also mechanical properties are changed: stress cracking resistance and toughness in the cold rise, whereas yield stress and heat resistance decrease. With a very high proportion of comonomers (about 50%) rubbery thermoplastics are produced (thermoplastic elastomers).[37]: 235
Ethylene-ethyl acrylate copolymers behave similarly to ethylene-vinyl acetate copolymers.[17]: 240
Crosslinking
A basic distinction is made between peroxide crosslinking (PE-Xa), silane crosslinking (PE-Xb), electron beam crosslinking (PE-Xc) and azo crosslinking (PE-Xd).[39]
Shown are the peroxide, the silane and irradiation crosslinking. In each method, a radical is generated in the polyethylene chain (top center), either by radiation (h·ν) or by peroxides (R-O-O-R). Then, two radical chains can either directly crosslink (bottom left) or indirectly by silane compounds (bottom right).
- Peroxide crosslinking (PE-Xa): The crosslinking of polyethylene using peroxides (e. g. dicumyl or di-tert-butyl peroxide) is still of major importance. In the so-called Engel process, a mixture of HDPE and 2%[40] peroxide is at first mixed at low temperatures in an extruder and then crosslinked at high temperatures (between 200 and 250 °C).[39] The peroxide decomposes to peroxide radicals (RO•), which abstract (remove) hydrogen atoms from the polymer chain, leading to radicals. When these combine, a crosslinked network is formed.[41] The resulting polymer network is uniform, of low tension and high flexibility, whereby it is softer and tougher than (the irradiated) PE-Xc.[39]
- Silane crosslinking (PE-Xb): In the presence of Catalysts such as dibutyltin dilaurate may accelerate the reaction.[40]
- Irradiation crosslinking (PE-Xc): The crosslinking of polyethylene is also possible by a downstream radiation source (usually an β-radiation possesses a penetration depth of 10 mm, ɣ-radiation 100 mm. Thereby the interior or specific areas can be excluded from the crosslinking.[39] However, due to high capital and operating costs radiation crosslinking plays only a minor role compared with the peroxide crosslinking.[37] In contrast to peroxide crosslinking, the process is carried out in the solid state. Thereby, the cross-linking takes place primarily in the amorphous regions, while the crystallinity remains largely intact.[40]
- Azo crosslinking (PE-Xd): In the so-called Lubonyl process polyethylene is crosslinked preadded azo compounds after extrusion in a hot salt bath.[37][39]
Chlorination and sulfochlorination
Chlorosulfonated PE (CSM) is used as starting material for ozone-resistant synthetic rubber.[42]
Bio-based polyethylene
Polyethylene can also be made from other feedstocks, including
Nomenclature and general description of the process
The name polyethylene comes from the ingredient and not the resulting chemical compound, which contains no double bonds. The scientific name polyethene is systematically derived from the scientific name of the monomer.
Footnotes
- ^ Erwähnt sei noch, dass aus einer ätherischen Diazomethanlösung sich beim Stehen manchmal minimale Quantitäten eines weissen, flockigen, aus Chloroform krystallisirenden Körpers abscheiden; ... [It should be mentioned that from an ether solution of diazomethane, upon standing, sometimes small quantities of a white, flakey substance precipitate, which can be crystallized with chloroform; ...].[10]: 2643
- ^ Die Abscheidung weisser Flocken aus Diazomethanlösungen erwähnt auch v. Pechmann (diese Berichte 31, 2643);[10] er hat sie aber wegen Substanzmangel nicht untersucht. Ich hatte übrigens Hrn. v. Pechmann schon einige Zeit vor Erscheinen seiner Publication mitgetheilt, dass aus Diazomethan ein fester, weisser Körper entstehe, der sich bei der Analyse als (CH2)x erwiesen habe, worauf mir Hr. v. Pechmann schrieb, dass er den weissen Körper ebensfalls beobachtet, aber nicht untersucht habe. Zuerst erwähnt ist derselbe in der Dissertation meines Schülers. (Hindermann, Zürich (1897), S. 120)[11]: footnote 3 on page 956 [Von Pechmann (these Reports, 31, 2643)[10] also mentioned the precipitation of white flakes from diazomethane solutions; however, due to a scarcity of the material, he didn't investigate it. Incidentally, some time before the appearance of his publication, I had communicated to Mr. von Pechmann that a solid, white substance arose from diazomethane, which on analysis proved to be (CH2)x, whereupon Mr. von Pechmann wrote me that he had likewise observed the white substance, but not investigated it. It is first mentioned in the dissertation of my student. (Hindermann, Zürich (1897), p. 120)].
References
- ^ Compendium of Polymer Terminology and Nomenclature – IUPAC Recommendations 2008 (PDF). Retrieved 28 August 2018.
- ^ a b Batra, Kamal (2014). Role of Additives in Linear Low Density Polyethylene (LLDPE) Films. p. 9. Retrieved 16 September 2014.
- ^ "poly(ethylene)". ChemSrc.
- S2CID 11545416.
- ^ a b Paul L. Splitstone and Walter H. Johnson (20 May 1974). "The Enthalpies of Combustion and Formation of Linear Polyethylene" (PDF). Journal of Research of the National Bureau Af Standards.
- ISBN 978-1-80455-987-1.
- ^ .
- PMID 28776036.
- ^ "Plastics: The Facts" (PDF). Plastics Europe. Archived from the original (PDF) on 4 February 2018. Retrieved 29 August 2018.
- ^ a b c d von Pechmann, H. (1898). "Ueber Diazomethan und Nitrosoacylamine". Berichte der Deutschen Chemischen Gesellschaft zu Berlin. 31: 2640–2646.
- ^ .
- .
[page 956]: Eine theilweise – übrigens immer nur minimale – Umwandlung des Diazomethans in Stickstoff und Polymethylen vollzieht sich auch bei ganz andersartigen Reactionen; ... [A partial – incidentally, always only minimal – conversion of diazomethane into nitrogen and polymethylene takes place also during quite different reactions; ...]
- ^ "Winnington history in the making". This is Cheshire. 23 August 2006. Archived from the original on 21 January 2010. Retrieved 20 February 2014.
- ^ "Poly – the all-star plastic". Popular Mechanics. Vol. 91, no. 1. Hearst Magazines. July 1949. pp. 125–129. Retrieved 20 February 2014 – via Google Books.
- ^ A History of Union Carbide Corporation (PDF). p. 69.
- ISBN 978-0-470-13798-7.
- ^ ISBN 978-3-446-43047-1.
- ^ "How to Identify Plastic Materials Using The Burn Test". Boedeker Plastics. Retrieved 8 May 2012.
- ^ "Electrical Properties of Plastic Materials" (PDF). professionalplastics.com. Professional Plastics. Archived (PDF) from the original on 20 September 2022. Retrieved 11 October 2022.
- ^ Chung, C. I. (2010) Extrusion of Polymers: Theory and Practice. 2nd ed.. Hanser: Munich.
- ^ Victor Ostrovskii et al. Ethylene Polymerization Heat (abstract) in Doklady Chemistry 184(1):103–104. January 1969.
- ^ ISBN 1-884207-17-0.
- ISBN 9780323354356.
- ^ Prideaux, Eric (3 November 2007). "Plastic incineration rise draws ire". The Japan Times. Archived from the original on 22 November 2012. Retrieved 8 May 2012.
- ^ Nguyen, Tuan (17 February 2011). "New invention turns plastic bags into oil". smartplanet.com. Retrieved 20 February 2014.
- ^ "CanadaWorld – WCI student isolates microbe that lunches on plastic bags". The Record.com. Archived from the original on 18 July 2011. Retrieved 20 February 2014.
- S2CID 2977246.
- PMID 19865515.
- ^ Balster, Lori (27 January 2015). "Discovery of plastic-eating bacteria may speed waste reduction". fondriest.com.
- PMID 25384056.
- ^ "Forscherin entdeckt zufällig Plastik-fressende Raupe". Der Spiegel (in German). 24 April 2017. Retrieved 24 April 2017.
- ^ Briggs, Helen. "Plastic-eating caterpillar could munch waste, scientists say". BBC News. Retrieved 24 April 2017.
- PMID 32126962.
- PMID 36195604.
- PMID 30067755.
- ^ ISBN 978-1859574935.
- ^ ISBN 978-3-642-16173-5.
- ISBN 978-3-642-16173-5.
- ^
- ^ ISBN 978-3527306732.
- ISBN 978-3-642-34773-3.
- ^ Chlorsulfoniertes Polyethylen (CSM). ChemgaPedia.de
- ^ "Braskem & Toyota Tsusho start joint marketing activities for green polyethylene from sugar cane" (Press release). yourindustrynews.com. 26 September 2008. Archived from the original on 21 May 2013. Retrieved 20 February 2014.
- ^ ISBN 978-0632037025. Retrieved 20 February 2014.
- ^ .
- ^ "IUPAC Provisional Recommendations on the Nomenclature of Organic Chemistry". International Union of Pure and Applied Chemistry. 27 October 2004. Retrieved 20 February 2014.
Bibliography
- Piringer, Otto G.; Baner, Albert Lawrence (2008). Plastic Packaging: Interactions with Food and Pharmaceuticals (2nd ed.). Wiley-VCH. ISBN 978-3-527-31455-3. Retrieved 20 February 2014.
- Plastics Design Library (1997). Handbook of Plastics Joining: A Practical Guide (illustrated ed.). William Andrew. ISBN 978-1-884207-17-4. Retrieved 20 February 2014.
External links
- Polythene's story: The accidental birth of plastic bags
- Polythene Technical Properties & Applications
- Article describing the discovery of Sphingomonas as a biodegrader of plastic bags Archived 5 November 2012 at the Wayback Machine Kawawada, Karen, Waterloo Region Record (22 May 2008).