Polyketide
In
History
Naturally produced polyketides by various plants and organisms have been used by humans since before studies on them began in the 19th and 20th century. In 1893, J. Norman Collie synthesized detectable amounts of orcinol by heating dehydracetic acid with barium hydroxide causing the pyrone ring to open into a triketide.[2] Further studies in 1903 by Collie on the triketone polyketide intermediate noted the condensation occurring amongst compounds with multiple keten groups coining the term polyketides.[3]
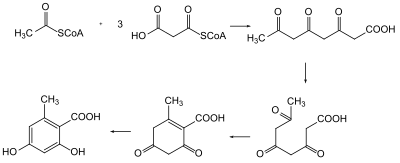
It wasn't until 1955 that the biosynthesis of polyketides were understood.
Discovery
Polyketides can be produced in bacteria, fungi, plants, and certain marine organisms.[6] Earlier discovery of naturally occurring polyketides involved the isolation of the compounds being produced by the specific organism using organic chemistry purification methods based on bioactivity screens.[7] Later technology allowed for the isolation of the genes and heterologous expression of the genes to understand the biosynthesis.[8] In addition, further advancements in biotechnology have allowed for the use of metagenomics and genome mining to find new polyketides using similar enzymes to known polyketides.[9]
Biosynthesis
Polyketides are synthesized by multienzyme polypeptides that resemble eukaryotic fatty acid synthase but are often much larger.[4] They include acyl-carrier domains plus an assortment of enzymatic units that can function in an iterative fashion, repeating the same elongation/modification steps (as in fatty acid synthesis), or in a sequential fashion so as to generate more heterogeneous types of polyketides.[10]

Polyketide synthase
Polyketides are produced by
PKSs are multi-domain enzymes or enzyme complex consisting of various domains. The polyketide chains produced by a minimal
Termination of the polyketide scaffold biosynthesis can also vary. It is sometimes accompanied by a thioesterase that releases the polyketide via hydrating the thioester linkage (as in fatty acid synthesis) creating a linear polyketide scaffold. However, if water is not able to reach the active site, the hydrating reaction will not occur and an intramolecular reaction is more probable creating a macrocyclic polyketide. Another possibility is spontaneous hydrolysis without the aid of a thioesterase.[15]
Post-tailoring enzymes
Further possible modifications to the polyketide scaffolds can be made. This can include glycosylation via a glucosyltransferase or oxidation via a monooxygenase.[16] Similarly, cyclization and aromatization can be introduced via a cyclase, sometimes proceeded by the enol tautomers of the polyketide.[17] These enzymes are not part of the domains of the polyketide synthase. Instead, they are found in gene clusters in the genome close to the polyketide synthase genes.[18]
Classification
Polyketides are a structurally diverse family.
Polyketide synthases are also broadly divided into three classes: Type I PKSs (multimodular megasynthases that are non-iterative, often producing macrocodes, polyethers, and polyenes), Type II PKSs (dissociated
In addition to these subclasses, there also exist polyketides that are hybridized with nonribosomal peptides (Hybrid NRP-PK and PK-NRP). Since nonribosomal peptide assembly lines use carrier proteins similar to those use in polyketide synthases, convergence of the two systems evolved to form hybrids, resulting in polypeptides with nitrogen in the skeletal structure and complex function groups similar to those found in amino acids.[21]
Applications
Polyketide
are in commercial use.Medicinal
There are more than 10,000 known polyketides, 1% of which are known to have potential for drug activity.[27] Polyketides comprise 20% of the top-selling pharmaceuticals with combined worldwide revenues of over USD 18 billion per year.[28]
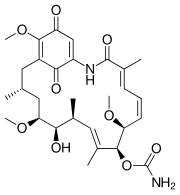
|
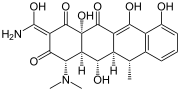
|

|
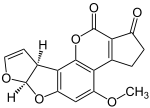
|
| Geldanamycin, an antibiotic. | Doxycycline, an antibiotic. | Erythromycin, an antibiotic. | Aflatoxin B1 known carcinogenic compound. |
Examples
- Macrolides
- Pikromycin, the first isolated macrolide (1951[29])
- The antibiotics erythromycin A, clarithromycin, and azithromycin
- The antihelminthics ivermectin
- Ansamycins
- The antitumor agents geldanamycin and macbecin,
- The antibiotic rifamycin
- The
- Polyenes
- The pimaricin
- The
- Polyethers
- The antibiotic monensin
- Tetracyclines
- The antibiotic agent doxycycline
- Acetogenins
- Others
- The inhibitor)
- HSP90inhibitors)
- The cholesterol lowering agent lovastatin
- Discodermolide
- Aflatoxin
- Usnic acid
- Anthracimycin
- Anthramycin
- Olivetolic acid (intermediate in cannabinoid pathways)[30]
Agricultural
Polyketides can be used for crop protection as pesticides.[31]
Examples
- Pesticides
- spinosad or spinosyn (an insecticide)
- avermectin
- polynactins
- tetramycin
Industrial
Polyketides can be used for industrial purposes, such as pigmentation[32] and dietary flavonoids.[33]
Examples
- Pigments
- azaphilones
- hydroxyanthraquinones
- naphthoquinones
- Flavonoids
Biotechnology
Protein engineering has opened avenues for creating polyketides not found in nature. For example, the modular nature of PKSs allows for domains to be replaced, added or deleted. Introducing diversity in assembly lines enables the discovery of new polyketides with increased bioactivity or new bioactivity.[21]
Furthermore, the use of genome mining allows for discovery of new natural polyketides and their assembly lines.[9]
See also
References
- ISSN 0368-1645.
- ISSN 0368-1645.
- ^ PMID 17898897.
- ISSN 1445-0038.
- PMID 21170424.
- ISSN 2040-2511.
- PMID 11238987.
- ^ PMID 24688489.
- ^ ISBN 9780470547847.
- PMID 11548049.
- PMID 11902441.
- PMID 32448179.
- PMID 30787330.
- ^ OCLC 985609285.
- PMID 31064143.
- PMID 1678529.
- PMID 27388157.
- PMID 11851471.
- PMID 12714063.
- ^ PMID 31838842.
- ^ "5.13E: Polyketide Antibiotics". Biology LibreTexts. 2017-05-09. Retrieved 2021-07-05.
- ^ PMID 25250879.
- PMID 30013965.
- PMID 19374124.
- PMID 25287333.
- ISBN 978-0-8412-3978-4.
- S2CID 205496204.
- .
- PMID 22802619.
- S2CID 235378697.
- ISBN 978-3-319-19456-1.
- PMID 32847100.
