Herd immunity

Herd immunity (also called herd effect, community immunity, population immunity, or mass immunity) is a form of indirect protection that applies only to
Once the herd immunity has been reached, disease gradually disappears from a population and may result in eradication or permanent reduction of infections to zero if achieved worldwide.[5][6] Herd immunity created via vaccination has contributed to the reduction of many diseases.[7]
Effects
Protection of those without immunity

Some individuals either cannot develop immunity after vaccination or for medical reasons cannot be vaccinated.[8][9][10] Newborn infants are too young to receive many vaccines, either for safety reasons or because passive immunity renders the vaccine ineffective.[11] Individuals who are immunodeficient due to HIV/AIDS, lymphoma, leukemia, bone marrow cancer, an impaired spleen, chemotherapy, or radiotherapy may have lost any immunity that they previously had and vaccines may not be of any use for them because of their immunodeficiency.[9][10][11][12]
A portion of those vaccinated may not develop long-term immunity.
High levels of immunity in one age group can create herd immunity for other age groups.
For sexually transmitted infections (STIs), high levels of immunity in heterosexuals of one sex induces herd immunity for heterosexuals of both sexes.[22][23][24] Vaccines against STIs that are targeted at heterosexuals of one sex result in significant declines in STIs in heterosexuals of both sexes if vaccine uptake in the target sex is high.[23][24][25] Herd immunity from female vaccination does not, however, extend to males who have sex with males.[24] High-risk behaviors make eliminating STIs difficult because, even though most infections occur among individuals with moderate risk, the majority of transmissions occur because of individuals who engage in high-risk behaviors.[22] For this reason, in certain populations it may be necessary to immunize high-risk individuals regardless of sex.[22][24]
Evolutionary pressure and serotype replacement
Herd immunity itself acts as an evolutionary pressure on pathogens, influencing viral evolution by encouraging the production of novel strains, referred to as escape mutants, that are able to evade herd immunity and infect previously immune individuals.[26][27] The evolution of new strains is known as serotype replacement, or serotype shifting, as the prevalence of a specific serotype declines due to high levels of immunity, allowing other serotypes to replace it.[28][29]
At the molecular level, viruses escape from herd immunity through antigenic drift, which is when mutations accumulate in the portion of the viral genome that encodes for the virus's surface antigen, typically a protein of the virus capsid, producing a change in the viral epitope.[30][31] Alternatively, the reassortment of separate viral genome segments, or antigenic shift, which is more common when there are more strains in circulation, can also produce new serotypes.[26][32] When either of these occur, memory T cells no longer recognize the virus, so people are not immune to the dominant circulating strain.[31][32] For both influenza and norovirus, epidemics temporarily induce herd immunity until a new dominant strain emerges, causing successive waves of epidemics.[30][32] As this evolution poses a challenge to herd immunity, broadly neutralizing antibodies and "universal" vaccines that can provide protection beyond a specific serotype are in development.[27][33][34]
Initial vaccines against Streptococcus pneumoniae significantly reduced nasopharyngeal carriage of vaccine serotypes (VTs), including antibiotic-resistant types,[18][35] only to be entirely offset by increased carriage of non-vaccine serotypes (NVTs).[18][28][29] This did not result in a proportionate increase in disease incidence though, since NVTs were less invasive than VTs.[28] Since then, pneumococcal vaccines that provide protection from the emerging serotypes have been introduced and have successfully countered their emergence.[18] The possibility of future shifting remains, so further strategies to deal with this include expansion of VT coverage and the development of vaccines that use either killed whole-cells, which have more surface antigens, or proteins present in multiple serotypes.[18][36]
Eradication of diseases
If herd immunity has been established and maintained in a population for a sufficient time, the disease is inevitably eliminated – no more endemic transmissions occur.[5] If elimination is achieved worldwide and the number of cases is permanently reduced to zero, then a disease can be declared eradicated.[6] Eradication can thus be considered the final effect or end-result of public health initiatives to control the spread of contagious disease.[6][7] In cases in which herd immunity is compromised, on the contrary, disease outbreaks among the unvaccinated population are likely to occur.[37]
The benefits of eradication include ending all
Free riding
Herd immunity is vulnerable to the
Mechanism
Individuals who are immune to a disease act as a barrier in the spread of disease, slowing or preventing the transmission of disease to others.[51] An individual's immunity can be acquired via a natural infection or through artificial means, such as vaccination.[51] When a critical proportion of the population becomes immune, called the herd immunity threshold (HIT) or herd immunity level (HIL), the disease may no longer persist in the population, ceasing to be endemic.[5][26]
The theoretical basis for herd immunity generally assumes that vaccines induce solid immunity, that populations mix at random, that the pathogen does not evolve to evade the immune response, and that there is no non-human vector for the disease.[2]
Theoretical basis

The critical value, or threshold, in a given population, is the point where the disease reaches an
S can be rewritten as (1 − p), where p is the proportion of the population that is immune so that p + S equals one. Then, the equation can be rearranged to place p by itself as follows:[citation needed]
With p being by itself on the left side of the equation, it can be renamed as pc, representing the critical proportion of the population needed to be immune to stop the transmission of disease, which is the same as the "herd immunity threshold" HIT.[22] R0 functions as a measure of contagiousness, so low R0 values are associated with lower HITs, whereas higher R0s result in higher HITs.[26][40] For example, the HIT for a disease with an R0 of 2 is theoretically only 50%, whereas a disease with an R0 of 10 the theoretical HIT is 90%.[26]
When the effective reproduction number Re of a contagious disease is reduced to and sustained below 1 new individual per infection, the number of cases occurring in the population gradually decreases until the disease has been eliminated.[22][26][52] If a population is immune to a disease in excess of that disease's HIT, the number of cases reduces at a faster rate, outbreaks are even less likely to happen, and outbreaks that occur are smaller than they would be otherwise.[2][22] If the effective reproduction number increases to above 1, then the disease is neither in a steady state nor decreasing in incidence, but is actively spreading through the population and infecting a larger number of people than usual.[41][52]
An assumption in these calculations is that populations are homogeneous, or well-mixed, meaning that every individual is equally likely to come into contact with any other individual, when in reality populations are better described as social networks as individuals tend to cluster together, remaining in relatively close contact with a limited number of other individuals. In these networks, transmission only occurs between those who are geographically or physically close to one another.[2][40][41] The shape and size of a network is likely to alter a disease's HIT, making incidence either more or less common.[26][40] Mathematical models can use contact matrices to estimate the likelihood of encounters and thus transmission.[53]
In heterogeneous populations, R0 is considered to be a measure of the number of cases generated by a "typical" contagious person, which depends on how individuals within a network interact with each other.[2] Interactions within networks are more common than between networks, in which case the most highly connected networks transmit disease more easily, resulting in a higher R0 and a higher HIT than would be required in a less connected network.[2][41] In networks that either opt not to become immune or are not immunized sufficiently, diseases may persist despite not existing in better-immunized networks.[41]
| Disease | Transmission |
R0 | HIT[a] |
|---|---|---|---|
| Measles | Aerosol | 12–18[54][55] | 92–94% |
| Chickenpox (varicella) | Aerosol | 10–12[56] | 90–92% |
| Mumps | Respiratory droplets | 10–12[57] | 90–92% |
| COVID-19 (see values for specific strains below) | Respiratory droplets and aerosol | 2.9-9.5[58] | 65–89% |
| Rubella | Respiratory droplets | 6–7[b] | 83–86% |
| Polio | Fecal–oral route | 5–7[b] | 80–86% |
| Pertussis | Respiratory droplets | 5.5[63] | 82% |
| Smallpox | Respiratory droplets | 3.5–6.0[64] | 71–83% |
| HIV/AIDS | Body fluids | 2–5[65] | 50–80% |
SARS |
Respiratory droplets | 2–4[66] | 50–75% |
| Diphtheria | Saliva | 2.6 (1.7–4.3)[67] | 62% (41–77%) |
| Common cold (e.g., rhinovirus) | Respiratory droplets | 2–3[68][medical citation needed] | 50–67% |
| Mpox | Physical contact, body fluids, respiratory droplets, sexual (MSM) | 2.1 (1.1–2.7)[69][70] | 53% (22–63%) |
2014 outbreak ) |
Body fluids | 1.8 (1.4–1.8)[71] | 44% (31–44%) |
| Influenza (seasonal strains) | Respiratory droplets | 1.3 (1.2–1.4)[72] | 23% (17–29%) |
| Andes hantavirus | Respiratory droplets and body fluids | 1.2 (0.8–1.6)[73] | 16% (0–36%)[c] |
| Nipah virus | Body fluids | 0.5[74] | 0%[c] |
| MERS | Respiratory droplets | 0.5 (0.3–0.8)[75] | 0%[c] |
Overshoot
The cumulative proportion of individuals who get infected during the course of a disease outbreak can exceed the HIT. This is because the HIT does not represent the point at which the disease stops spreading, but rather the point at which each infected person infects fewer than one additional person on average. When the HIT is reached, the number of additional infections does not immediately drop to zero. The excess of the cumulative proportion of infected individuals over the theoretical HIT is known as the overshoot.[76][77][78]
Boosts
Vaccination
The primary way to boost levels of immunity in a population is through vaccination.[2][79] Vaccination is originally based on the observation that milkmaids exposed to cowpox were immune to smallpox, so the practice of inoculating people with the cowpox virus began as a way to prevent smallpox.[39] Well-developed vaccines provide protection in a far safer way than natural infections, as vaccines generally do not cause the diseases they protect against and severe adverse effects are significantly less common than complications from natural infections.[80][81]
The
Assuming a vaccine is 100% effective, then the equation used for calculating the herd immunity threshold can be used for calculating the vaccination level needed to eliminate a disease, written as Vc.[2] Vaccines are usually imperfect however, so the effectiveness, E, of a vaccine must be accounted for:
From this equation, it can be observed that if E is less than (1 − 1/R0), then it is impossible to eliminate a disease, even if the entire population is vaccinated.[2] Similarly, waning vaccine-induced immunity, as occurs with acellular pertussis vaccines, requires higher levels of booster vaccination to sustain herd immunity.[2][16] If a disease has ceased to be endemic to a population, then natural infections no longer contribute to a reduction in the fraction of the population that is susceptible. Only vaccination contributes to this reduction.[22] The relation between vaccine coverage and effectiveness and disease incidence can be shown by subtracting the product of the effectiveness of a vaccine and the proportion of the population that is vaccinated, pv, from the herd immunity threshold equation as follows:
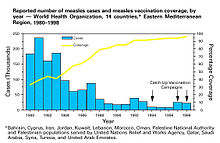
It can be observed from this equation that, all other things being equal ("ceteris paribus"), any increase in either vaccine coverage or vaccine effectiveness, including any increase in excess of a disease's HIT, further reduces the number of cases of a disease.[22] The rate of decline in cases depends on a disease's R0, with diseases with lower R0 values experiencing sharper declines.[22]
Vaccines usually have at least one contraindication for a specific population for medical reasons, but if both effectiveness and coverage are high enough then herd immunity can protect these individuals.[8][12][15] Vaccine effectiveness is often, but not always, adversely affected by passive immunity,[86][87] so additional doses are recommended for some vaccines while others are not administered until after an individual has lost his or her passive immunity.[11][15]
Passive immunity
Individual immunity can also be gained passively, when antibodies to a pathogen are transferred from one individual to another. This can occur naturally, whereby maternal antibodies, primarily immunoglobulin G antibodies, are transferred across the placenta and in colostrum to fetuses and newborns.[88][89] Passive immunity can also be gained artificially, when a susceptible person is injected with antibodies from the serum or plasma of an immune person.[82][90]
Protection generated from passive immunity is immediate, but wanes over the course of weeks to months, so any contribution to herd immunity is temporary.[5][82][91] For diseases that are especially severe among fetuses and newborns, such as influenza and tetanus, pregnant women may be immunized in order to transfer antibodies to the child.[8][92][93] In the same way, high-risk groups that are either more likely to experience infection, or are more likely to develop complications from infection, may receive antibody preparations to prevent these infections or to reduce the severity of symptoms.[90]
Cost–benefit analysis
Herd immunity is often accounted for when conducting cost–benefit analyses of vaccination programs. It is regarded as a positive externality of high levels of immunity, producing an additional benefit of disease reduction that would not occur had no herd immunity been generated in the population.[94][95] Therefore, herd immunity's inclusion in cost–benefit analyses results both in more favorable cost-effectiveness or cost–benefit ratios, and an increase in the number of disease cases averted by vaccination.[95] Study designs done to estimate herd immunity's benefit include recording disease incidence in households with a vaccinated member, randomizing a population in a single geographic area to be vaccinated or not, and observing the incidence of disease before and after beginning a vaccination program.[96] From these, it can be observed that disease incidence may decrease to a level beyond what can be predicted from direct protection alone, indicating that herd immunity contributed to the reduction.[96] When serotype replacement is accounted for, it reduces the predicted benefits of vaccination.[95]
History
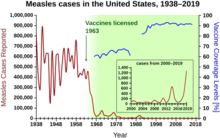
Herd immunity was recognized as a naturally occurring phenomenon in the 1930s when it was observed that after a significant number of children had become immune to measles, the number of new infections temporarily decreased.[97] Mass vaccination to induce herd immunity has since become common and proved successful in preventing the spread of many contagious diseases.[22] Opposition to vaccination has posed a challenge to herd immunity, allowing preventable diseases to persist in or return to populations with inadequate vaccination rates.[45][46][47]
The exact herd immunity threshold (HIT) varies depending on the basic reproduction number of the disease. An example of a disease with a high threshold was the measles, with a HIT exceeding 95%.[98]
The term "herd immunity" was first used in 1894 by American veterinary scientist and then Chief of the Bureau of Animal Industry of the US Department of Agriculture Daniel Elmer Salmon to describe the healthy vitality and resistance to disease of well-fed herds of hogs. In 1916 veterinary scientists inside the same Bureau of Animal Industry used the term to refer to the immunity arising following recovery in cattle infected with brucellosis, also known as "contagious abortion." By 1923 it was being used by British bacteriologists to describe experimental epidemics with mice, experiments undertaken as part of efforts to model human epidemic disease. By the end of the 1920s the concept was used extensively - particularly among British scientists - to describe the build up of immunity in populations to diseases such as diphtheria, scarlet fever, and influenza.[99] Herd immunity was recognized as a naturally occurring phenomenon in the 1930s when A. W. Hedrich published research on the epidemiology of measles in Baltimore, and took notice that after many children had become immune to measles, the number of new infections temporarily decreased, including among susceptible children.[100][97] In spite of this knowledge, efforts to control and eliminate measles were unsuccessful until mass vaccination using the measles vaccine began in the 1960s.[97] Mass vaccination, discussions of disease eradication, and cost–benefit analyses of vaccination subsequently prompted more widespread use of the term herd immunity.[2] In the 1970s, the theorem used to calculate a disease's herd immunity threshold was developed.[2] During the smallpox eradication campaign in the 1960s and 1970s, the practice of ring vaccination, to which herd immunity is integral, began as a way to immunize every person in a "ring" around an infected individual to prevent outbreaks from spreading.[101]
Since the adoption of mass and ring vaccination, complexities and challenges to herd immunity have arisen.[2][79] Modeling of the spread of contagious disease originally made a number of assumptions, namely that entire populations are susceptible and well-mixed, which is not the case in reality, so more precise equations have been developed.[2] In recent decades, it has been recognized that the dominant strain of a microorganism in circulation may change due to herd immunity, either because of herd immunity acting as an evolutionary pressure or because herd immunity against one strain allowed another already-existing strain to spread.[30][29] Emerging or ongoing fears and controversies about vaccination have reduced or eliminated herd immunity in certain communities, allowing preventable diseases to persist in or return to these communities.[45][46][47]
See also
- Premunity
- Social distancing
Notes
References
- ^ "Herd immunity | immunology". Encyclopedia Britannica. Archived from the original on 1 April 2023. Retrieved 13 June 2021.
- ^ PMID 21427399.
- ISBN 978-1455742516. Archivedfrom the original on 1 July 2023. Retrieved 29 March 2015.
- ^ "Cold-Causing Coronaviruses Don't Seem to Confer Lasting Immunity". The Scientist Magazine®. Archived from the original on 7 January 2021. Retrieved 26 January 2021.
- ^ ISBN 978-1118308646. Archivedfrom the original on 1 May 2021. Retrieved 29 March 2015.
- ^ ISBN 978-0199596614. Archivedfrom the original on 1 May 2021. Retrieved 29 March 2015.
- ^ PMID 21604922.
- ^ PMID 23910028.
- ^ a b c "Herd Immunity". Oxford Vaccine Group, University of Oxford. Archived from the original on 2 August 2019. Retrieved 12 December 2017.
- ^ PMID 24868544.
- ^ PMID 21293327.
- ^ PMID 22773718.
- PMID 22862749.
- ^ ISBN 978-0323313087. Archivedfrom the original on 1 May 2021. Retrieved 30 March 2015.
- ^ ISBN 978-0124157675. Archivedfrom the original on 1 May 2021. Retrieved 30 March 2015.
- ^ S2CID 8273985. Archived from the original(PDF) on 3 March 2019.
- PMID 21600850.
- ^ PMID 22862432.
- S2CID 5405583.
- from the original on 16 October 2019. Retrieved 5 July 2019.
- ^ PMID 25003085.
- ^ PMID 15627236.
- ^ PMID 22219162.
- ^ PMID 23391351.
- PMID 21962468.
- ^ PMID 24175217.
- ^ PMID 23330954.
- ^ PMID 21492929.
- ^ PMID 22903767.
- ^ PMID 21310617.
- ^ PMID 24232370.
- ^ PMID 23124938.
- PMID 21251008.
- PMID 24991424.
- PMID 19366365.
- S2CID 205784538.
- PMID 34656181.
- PMID 23520729.
- ^ PMID 23760373.
- ^ PMID 19197342.
- ^ (PDF) from the original on 4 November 2018. Retrieved 4 November 2018.
- S2CID 23471069.
- ^ ISBN 978-3319133591. Archivedfrom the original on 1 May 2021. Retrieved 30 March 2015.
- ^ ISBN 978-1464803109.
- ^ PMID 22926181.
- ^ PMID 23584253.
- ^ PMID 23807359.
- ^ PMID 23744504.
- PMID 23733039.
- PMID 23790997.
- ^ ISBN 978-1449645175. Archivedfrom the original on 12 April 2021. Retrieved 29 March 2015.
- ^ ISBN 978-3642391491. Archivedfrom the original on 1 May 2021. Retrieved 29 March 2015.
- ISBN 978-0-323-95389-4
- PMID 28757186.
- PMID 30560777.
- ^ Ireland's Health Services. Health Care Worker Information (PDF). Retrieved 27 March 2020.
- ^ Australian government Department of Health Mumps Laboratory Case Definition (LCD)
- PMID 35262737.
- ^ Centers for Disease Control and Prevention; World Health Organization (2001). "History and epidemiology of global smallpox eradication". Smallpox: disease, prevention, and intervention (training course) (Presentation). Atlanta: Centers for Disease Control and Prevention (published 25 August 2014). cdc:27929. Archived (PDF) from the original on 17 March 2017. Retrieved 17 June 2021.
- PMID 8174658.
- PMID 12174806.
- PMID 11016876.
- PMID 20585374.
- S2CID 52799168. Retrieved 18 March 2020.
- ^ "Playing the Numbers Game: R0". National Emerging Special Pathogen Training and Education Center. 30 January 2020. Archived from the original on 12 May 2020. Retrieved 27 December 2020.
[...] while infections that require sexual contact like HIV have a lower R0 (2-5).
- hdl:10665/70863. WHO/CDS/CSR/GAR/2003.11.
A number of researchers have estimated the basic reproduction number by fitting models to the initial growth of epidemics in a number of countries. Their observations indicate that the SARS-CoV is less transmissible than initially thought with estimates of Ro in the range of 2-4.
- PMID 31425581.
- ^ Freeman C (6 November 2014). "Magic formula that will determine whether Ebola is beaten". The Telegraph. Telegraph.Co.Uk. Archived from the original on 12 January 2022. Retrieved 30 March 2020.
- PMID 33012864. Archived from the original(PDF) on 11 December 2020.
- PMID 36845803.
- PMID 28166851.
The median of the R0 mean estimate for the ongoing epidemic (overall) is 1.78 (interquartile range: 1.44, 1.80)
- PMID 17634159.
The reproduction number across influenza seasons and countries lied in the range 0.9–2.0 with an overall mean of 1.3, and 95% confidence interval (CI) 1.2–1.4.
- S2CID 227259435.
- PMID 23911335.
- PMID 26132768.
- PMID 17251095.
In general, the number of infecteds grows until the number of susceptibles has fallen to Sth. At this point, the average number of secondary infections created by an infected person drops below 1 and therefore the number of infecteds starts to decrease. However, right at this inflection point, the maximum number of infecteds is present. These infecteds will create on average less than 1, but still more than zero further infections, leading to additional depletion of susceptibles and therefore causing an overshoot.
- PMID 22701558.
- ^ Bergstrom CT, Dean N (1 May 2020). "Opinion: What the Proponents of 'Natural' Herd Immunity Don't Say". The New York Times. Archived from the original on 3 June 2020. Retrieved 30 May 2020.
- ^ S2CID 19197608.
- from the original on 30 January 2020. Retrieved 5 July 2019.
- PMID 34806766.
- ^ ISBN 978-1284057102. Archivedfrom the original on 1 May 2021. Retrieved 30 March 2015.
- PMID 24757524.
- PMID 23375475.
- PMID 22939022.
- PMID 22841972.
- PMID 22189699.
- PMID 20956021.
- PMID 22235228.
- ^ ISBN 978-8131236246. Archivedfrom the original on 1 May 2021. Retrieved 30 March 2015.
- ISBN 978-0199661756. Archivedfrom the original on 1 May 2021. Retrieved 30 March 2015.
- PMID 26144877.
- S2CID 206342684.
- PMID 25136129.
- ^ PMID 23072714.
- ^ PMID 23357859.
- ^ PMID 4960501.
- PMID 20392713.
- S2CID 235404539.
- .
- PMID 7044193.
External links
- PMID 20474777.
- A visual simulation of herd immunity written by Shane Killian and modified by Robert Webb
- Herd immunity simulation






