Porphyrin
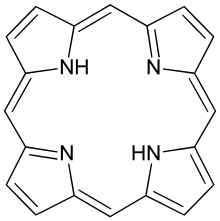
Porphyrins (
The parent of porphyrins is porphine, a rare chemical compound of exclusively theoretical interest. Substituted porphines are called porphyrins.[1] With a total of 26 π-electrons, of which 18 π-electrons form a planar, continuous cycle, the porphyrin ring structure is often described as aromatic.[2][3] One result of the large conjugated system is that porphyrins typically absorb strongly in the visible region of the electromagnetic spectrum, i.e. they are deeply colored. The name "porphyrin" derives from the Greek word πορφύρα (porphyra), meaning purple.[4]
Structure
Complexes of porphyrins
Concomitant with the displacement of two N-H protons, porphyrins bind metal ions in the N4 "pocket". The metal ion usually has a charge of 2+ or 3+. A schematic equation for these syntheses is shown:
- H2porphyrin + [MLn]2+ → M(porphyrinate)Ln−4 + 4 L + 2 H+, where M = metal ion and L = a ligand
- Representative porphyrins and derivatives
-
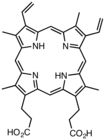
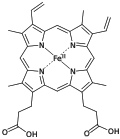
Derivatives of protoporphyrin IX are common in nature, the precursor to hemes.
-
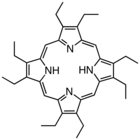
Octaethylporphyrin (H2OEP) is a synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, OEP2− is highly symmetrical.
-
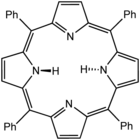
Tetraphenylporphyrin (H2TPP)is another synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, TPP2− is highly symmetrical. Another difference is that its methyne centers are occupied by phenyl groups.
-
Simplified view of heme, a complex of a protoporphyrin IX.
-
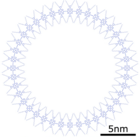
A macrocycle of 40 porphyrin molecules, model
-
A macrocycle of 40 porphyrin molecules, STM image
Ancient porphyrins
A geoporphyrin, also known as a petroporphyrin, is a porphyrin of geologic origin.
The field of
Biosynthesis
In non-photosynthetic
Two molecules of dALA are then combined by
The following scheme summarizes the biosynthesis of porphyrins, with references by EC number and the

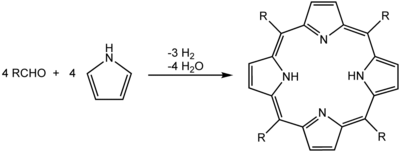
Laboratory synthesis
A common synthesis for porphyrins is the
Potential applications
Photodynamic therapy
Porphyrins have been evaluated in the context of photodynamic therapy (PDT) since they strongly absorb light, which is then converted to heat in the illuminated areas.[15] This technique has been applied in macular degeneration using verteporfin.[16]
PDT is considered a noninvasive cancer treatment, involving the interaction between light of a determined frequency, a photo-sensitizer, and oxygen. This interaction produces the formation of a highly reactive oxygen species (ROS), usually singlet oxygen, as well as superoxide anion, free hydroxyl radical, or hydrogen peroxide.[17] These high reactive oxygen species react with susceptible cellular organic biomolecules such as; lipids, aromatic amino acids, and nucleic acid heterocyclic bases, to produce oxidative radicals that damage the cell, possibly inducing apoptosis or even necrosis.[18]
Molecular electronics and sensors
Porphyrin-based compounds are of interest as possible components of
Biological applications
Porphyrins have been investigated as possible anti-inflammatory agents[22] and evaluated on their anti-cancer and anti-oxidant activity.[23] Several porphyrin-peptide conjugates were found to have antiviral activity against HIV in vitro.[24]
Toxicology
Heme biosynthesis is used as
Gallery
-
Lewis structure for meso-tetraphenylporphyrin
-
UV–vis readout for meso-tetraphenylporphyrin
-
Light-activated porphyrin. Monatomic oxygen. Cellular aging
Related species
In nature
Several heterocycles related to porphyrins are found in nature, almost always bound to metal ions. These include
| N4-macrocycle | Cofactor name | metal | comment |
|---|---|---|---|
| chlorin | chlorophyll | magnesium | several versions of chlorophyll exist (sidechain; exception being chlorophyll c) |
| bacteriochlorin | bacteriochlorophyll (in part) | magnesium | several versions of bacteriochlorophyll exist (sidechain; some use a usual chlorin ring) |
| sirohydrochlorin (an isobacteriochlorin) | siroheme | iron | Important cofactor in sulfur assimilation |
| biosynthetic intermediate en route to cofactor F430 and B12 | |||
| corrin | vitamin B12 | cobalt | several variants of B12 exist (sidechain) |
| corphin | Cofactor F430 | nickel | highly reduced macrocycle |
Synthetic
A benzoporphyrin is a porphyrin with a benzene ring fused to one of the pyrrole units. e.g. verteporfin is a benzoporphyrin derivative.[26]
Non-natural porphyrin isomers

The first synthetic porphyrin isomer was reported by Emanual Vogel and coworkers in 1986.[27] This isomer [18]porphyrin-(2.0.2.0) is named as porphycene, and the central N4 Cavity forms a rectangle shape as shown in figure.[28] Porphycenes showed interesting photophysical behavior and found versatile compound towards the photodynamic therapy.[29] This inspired Vogel and Sessler to took up the challenge of preparing [18]porphyrin-(2.1.0.1) and named it as corrphycene or porphycerin.[30] The third porphyrin that is [18]porphyrin-(2.1.1.0), was reported by Callot and Vogel-Sessler. Vogel and coworkers reported successful isolation of [18]porphyrin-(3.0.1.0) or isoporphycene.[31] The Japanese scientist Furuta[32] and Polish scientist Latos-Grażyński[33] almost simultaneously reported the N-confused porphyrins. The inversion of one of the pyrrolic subunits in the macrocyclic ring resulted in one of the nitrogen atoms facing outwards from the core of the macrocycle.

See also
- A porphyrin-related disease: porphyria
- Porphyrin coordinated to iron: heme
- A heme-containing group of enzymes: Cytochrome P450
- Porphyrin coordinated to magnesium: chlorophyll
- The one-carbon-shorter analogues: corroles, including vitamin B12, which is coordinated to a cobalt
- Corphins, the highly reduced porphyrin coordinated to nickel that binds the methyl coenzyme M reductase(MCR)
- Nitrogen-substituted porphyrins: phthalocyanine
References
- .
- PMID 25002069.
- .
- ^ Harper D, Buglione DC. "porphyria (n.)". The Online Etymology Dictionary. Retrieved 14 September 2014.
- PMID 26482230.
- ISBN 9781119951438.
- PMID 9533688.
- hdl:2262/73843.
- ^ ISBN 9780123932006.
- .
- .
- .
- .
- .
- ^ Giuntini F, Boyle R, Sibrian-Vazquez M, Vicente MG (2014). "Porphyrin conjugates for cancer therapy". In Kadish KM, Smith KM, Guilard R (eds.). Handbook of Porphyrin Science. Vol. 27. pp. 303–416.
- PMID 17636693.
- PMID 19659920.
- PMID 26317756.
- PMID 22649792.
- .
- S2CID 28058582.
- PMID 25863493.
- PMID 31384764.
- PMID 34033716.
- ISBN 978-1-4665-0260-4.
- S2CID 260491296.
- .
- S2CID 246165814.
- doi:10.1002/jpp.328.
- .
- PMID 10540393.
- .
- .