B cell
| B lymphocyte cell | |
|---|---|
 Transmission electron micrograph of a human B cell | |
| Details | |
| System | Immune system |
| Identifiers | |
| Latin | lymphocytus B |
| MeSH | D001402 |
| FMA | 62869 |
| Anatomical terms of microanatomy | |
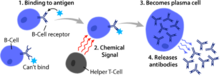

B cells, also known as B lymphocytes, are a type of white blood cell of the lymphocyte subtype.[1] They function in the humoral immunity component of the adaptive immune system.[1] B cells produce antibody molecules which may be either secreted or inserted into the plasma membrane where they serve as a part of B-cell receptors.[2] When a naïve or memory B cell is activated by an antigen, it proliferates and differentiates into an antibody-secreting effector cell, known as a plasmablast or plasma cell.[2] In addition, B cells present antigens (they are also classified as professional antigen-presenting cells, APCs) and secrete cytokines.[1] In mammals, including marsupials [3] B cells mature in the bone marrow, which is at the core of most bones.[4] In birds, B cells mature in the bursa of Fabricius, a lymphoid organ where they were first discovered by Chang and Glick,[5] which is why the B stands for bursa and not bone marrow, as commonly believed.
B cells, unlike the other two classes of lymphocytes, T cells and natural killer cells, express B cell receptors (BCRs) on their cell membrane.[1] BCRs allow the B cell to bind to a foreign antigen, against which it will initiate an antibody response.[1] B cell receptors are extremely specific, with all BCRs on a B cell recognizing the same epitope.[6]
Development
B cells develop from

B cells undergo two types of selection while developing in the bone marrow to ensure proper development, both involving B cell receptors (BCR) on the surface of the cell. Positive selection occurs through antigen-independent signalling involving both the pre-BCR and the BCR.[10][11] If these receptors do not bind to their ligand, B cells do not receive the proper signals and cease to develop.[10][11] Negative selection occurs through the binding of self-antigen with the BCR; if the BCR can bind strongly to self-antigen, then the B cell undergoes one of four fates: clonal deletion, receptor editing, anergy, or ignorance (B cell ignores signal and continues development).[11] This negative selection process leads to a state of central tolerance, in which the mature B cells do not bind self antigens present in the bone marrow.[9]
To complete development, immature B cells migrate from the bone marrow into the spleen as

Activation

B cell activation occurs in the
B cell activation is enhanced through the activity of CD21, a surface receptor in complex with surface proteins CD19 and CD81 (all three are collectively known as the B cell coreceptor complex).[18] When a BCR binds an antigen tagged with a fragment of the C3 complement protein, CD21 binds the C3 fragment, co-ligates with the bound BCR, and signals are transduced through CD19 and CD81 to lower the activation threshold of the cell.[19]
T cell-dependent activation
Antigens that activate B cells with the help of T-cell are known as T cell-dependent (TD) antigens and include foreign proteins.[1] They are named as such because they are unable to induce a humoral response in organisms that lack T cells.[1] B cell responses to these antigens takes multiple days, though antibodies generated have a higher affinity and are more functionally versatile than those generated from T cell-independent activation.[1]
Once a BCR binds a TD antigen, the antigen is taken up into the B cell through

Once activated, B cells participate in a two-step differentiation process that yields both short-lived plasmablasts for immediate protection and long-lived plasma cells and memory B cells for persistent protection.[17] The first step, known as the extrafollicular response, occurs outside lymphoid follicles but still in the SLO.[17] During this step activated B cells proliferate, may undergo immunoglobulin class switching, and differentiate into plasmablasts that produce early, weak antibodies mostly of class IgM.[22]
The second step consists of activated B cells entering a lymphoid follicle and forming a germinal center (GC), which is a specialized microenvironment where B cells undergo extensive proliferation, immunoglobulin class switching, and affinity maturation directed by somatic hypermutation.[23] These processes are facilitated by TFH cells within the GC and generate both high-affinity memory B cells and long-lived plasma cells.[17] Resultant plasma cells secrete large amounts of antibody and either stay within the SLO or, more preferentially, migrate to bone marrow.[23]
T cell-independent activation
Antigens that activate B cells without T cell help are known as T cell-independent (TI) antigens[1] and include foreign polysaccharides and unmethylated CpG DNA.[17] They are named as such because they are able to induce a humoral response in organisms that lack T cells.[1] B cell response to these antigens is rapid, though antibodies generated tend to have lower affinity and are less functionally versatile than those generated from T cell-dependent activation.[1]
As with TD antigens, B cells activated by TI antigens need additional signals to complete activation, but instead of receiving them from T cells, they are provided either by recognition and binding of a common microbial constituent to toll-like receptors (TLRs) or by extensive crosslinking of BCRs to repeated epitopes on a bacterial cell.[1] B cells activated by TI antigens go on to proliferate outside lymphoid follicles but still in SLOs (GCs do not form), possibly undergo immunoglobulin class switching, and differentiate into short-lived plasmablasts that produce early, weak antibodies mostly of class IgM, but also some populations of long-lived plasma cells.[24]
Memory B cell activation
Memory B cell activation begins with the detection and binding of their target antigen, which is shared by their parent B cell.[25] Some memory B cells can be activated without T cell help, such as certain virus-specific memory B cells, but others need T cell help.[26] Upon antigen binding, the memory B cell takes up the antigen through receptor-mediated endocytosis, degrades it, and presents it to T cells as peptide pieces in complex with MHC-II molecules on the cell membrane.[25] Memory T helper (TH) cells, typically memory follicular T helper (TFH) cells, that were derived from T cells activated with the same antigen recognize and bind these MHC-II-peptide complexes through their TCR.[25] Following TCR-MHC-II-peptide binding and the relay of other signals from the memory TFH cell, the memory B cell is activated and differentiates either into plasmablasts and plasma cells via an extrafollicular response or enter a germinal center reaction where they generate plasma cells and more memory B cells.[25][26] It is unclear whether the memory B cells undergo further affinity maturation within these secondary GCs.[25] In vitro activation of memory B cells can be achieved through stimulation with various activators, such as pokeweed mitogen or anti-CD40 monoclonal antibodies, however, a study found a combination of R-848 and recombinant human IL-2 to be the most efficient activator.[27]
B cell types
- Plasmablast
- A short-lived, proliferating antibody-secreting cell arising from B cell differentiation.[1] Plasmablasts are generated early in an infection and their antibodies tend to have a weaker affinity towards their target antigen compared to plasma cell.[17] Plasmablasts can result from T cell-independent activation of B cells or the extrafollicular response from T cell-dependent activation of B cells.[1]
- Plasma cell
- A long-lived, non-proliferating antibody-secreting cell arising from B cell differentiation.[1] There is evidence that B cells first differentiate into a plasmablast-like cell, then differentiate into a plasma cell.[17] Plasma cells are generated later in an infection and, compared to plasmablasts, have antibodies with a higher affinity towards their target antigen due to affinity maturation in the germinal center (GC) and produce more antibodies.[17] Plasma cells typically result from the germinal center reaction from T cell-dependent activation of B cells, though they can also result from T cell-independent activation of B cells.[24]
- Lymphoplasmacytoid cell
- A cell with a mixture of B lymphocyte and plasma cell morphological features that is thought to be closely related to or a subtype of plasma cells. This cell type is found in pre-malignant and malignant Waldenström's macroglobulinemia.[28]
- Memory B cell
- Dormant B cell arising from B cell differentiation.[1] Their function is to circulate through the body and initiate a stronger, more rapid antibody response (known as the anamnestic secondary antibody response) if they detect the antigen that had activated their parent B cell (memory B cells and their parent B cells share the same BCR, thus they detect the same antigen).[26] Memory B cells can be generated from T cell-dependent activation through both the extrafollicular response and the germinal center reaction as well as from T cell-independent activation of B1 cells.[26]
- B-2 cell
- FO B cells and MZ B cells.[29]
- Follicular (FO) B cell(also known as a B-2 cell)
- Most common type of B cell and, when not circulating through the blood, is found mainly in the lymphoid follicles of secondary lymphoid organs (SLOs).[17] They are responsible for generating the majority of high-affinity antibodies during an infection.[1]
- Marginal-zone (MZ) B cell
- Found mainly in the marginal zone of the spleen and serves as a first line of defense against blood-borne pathogens, as the marginal zone receives large amounts of blood from the general circulation.[30] They can undergo both T cell-independent and T cell-dependent activation, but preferentially undergo T cell-independent activation.[17]
- B-1 cell
- Arises from a developmental pathway different from FO B cells and MZ B cells.[29] In mice, they predominantly populate the peritoneal cavity and pleural cavity, generate natural antibodies (antibodies produced without infection), defend against mucosal pathogens, and primarily exhibit T cell-independent activation.[29] A true homologue of mouse B-1 cells has not been discovered in humans, though various cell populations similar to B-1 cells have been described.[29]
- Regulatory B (Breg) cell
- An immunosuppressive B cell type that stops the expansion of pathogenic, pro-inflammatory lymphocytes through the secretion of IL-10, IL-35, and TGF-β.[31] Also, it promotes the generation of regulatory T (Treg) cells by directly interacting with T cells to skew their differentiation towards Tregs.[31] No common Breg cell identity has been described and many Breg cell subsets sharing regulatory functions have been found in both mice and humans.[31] It is currently unknown if Breg cell subsets are developmentally linked and how exactly differentiation into a Breg cell occurs.[31] There is evidence showing that nearly all B cell types can differentiate into a Breg cell through mechanisms involving inflammatory signals and BCR recognition.[31]
Autoimmune disease can result from abnormal B cell recognition of self-antigens followed by the production of autoantibodies.
Abnormal B cells may be relatively large and some diseases include this in their names, such as
Patients with B cell alymphocytosis are predisposed to infections.[35]
Epigenetics
A study that investigated the methylome of B cells along their differentiation cycle, using whole-genome bisulfite sequencing (WGBS), showed that there is a hypomethylation from the earliest stages to the most differentiated stages. The largest methylation difference is between the stages of germinal center B cells and memory B cells. Furthermore, this study showed that there is a similarity between B cell tumors and long-lived B cells in their DNA methylation signatures.[36]
See also
References
- ^ ISBN 9780815342434.
- ^ a b Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2002). "B Cells and Antibodies". Molecular Biology of the Cell (4th ed.). Garland Science.
- .
- PMID 25656707.
- ISSN 0032-5791.
- PMID 30863406.
- PMID 33392513.
- ^ PMID 20969583.
- ^ PMID 22378602.
- ^ S2CID 43158480.
- ^ PMID 18725575.
- PMID 10429672.
- ^ PMID 12810111.
- PMID 23348416.
- PMID 20192804.
- S2CID 24791216.
- ^ S2CID 9769697.
- PMID 22885687.
- PMID 11367532.
- PMID 23298205.
- ^ PMID 25677493.
- S2CID 2455541.
- ^ S2CID 5362003.
- ^ PMID 22529295.
- ^ PMID 22158414.
- ^ S2CID 20825732.
- PMID 23454005.
- S2CID 1508656.
- ^ S2CID 23355423.
- PMID 15771569.
- ^ PMID 25902480.
- ^ S2CID 11593298.
- PMID 22224767.
- PMID 27866585.
- ^ Grammatikos Alexandros, Donati Matthew, Johnston Sarah L., Gompels Mark M. Peripheral B Cell Deficiency and Predisposition to Viral Infections: The Paradigm of Immune Deficiencies. Frontiers in Immunology (12)2021 https://www.frontiersin.org/articles/10.3389/fimmu.2021.731643 DOI=10.3389/fimmu.2021.731643
- PMID 26053498.
