Prednisolone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Orapred, PediaPred, Millipred, others |
| Other names | 11,17-Dihydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-3-one |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a615042 |
| License data |
|
| Pregnancy category |
|
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 2–3.5 hours[2][3][4] |
| Excretion | urine |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Prednisolone is a
Side effects with short-term use include nausea, inability to concentrate, insomnia, or feeling tired.[5] More severe side effects include psychiatric problems, which may occur in about 5% of people.[9] Common side effects with long term use include bone loss, weakness, yeast infections, and easy bruising.[6] While short-term use in the later part of pregnancy is safe, long-term use or use in early pregnancy is occasionally associated with harm to the baby.[1] It is a glucocorticoid made from hydrocortisone (cortisol).[10]
Prednisolone was discovered and approved for medical use in 1955.[10] It is on the World Health Organization's List of Essential Medicines.[11] It is available as a generic drug.[6] In 2021, it was the 153rd most commonly prescribed medication in the United States, with more than 3 million prescriptions.[12][13]
Medical uses
Systemic use
Prednisolone is a
Prednisolone can also be used for allergic reactions ranging from seasonal allergies to drug allergic reactions.[22]
Prednisolone can also be used as an
Prednisolone in lower doses can be used in cases of primary adrenal insufficiency (Addison's disease).[24][25]
Topical use in ophthalmology

Topical prednisolone is a type of glucocorticoid, mainly used in the ophthalmic pathway as eye drops in numerous eye conditions,[26] including corneal injuries caused by chemicals, burns and alien objects, inflammation of the eyes, mild to moderate non-infectious allergies, disorders of the eyelid, conjunctiva or sclera, ocular inflammation caused by operation and optic neuritis.[27][28] Some side effects include glaucoma, blurring of vision, eye discomfort, impaired recovery of injured site, scarring of the optic nerve, clouding of lenses and urticaria.[28] However, their prevalence is not known.
Prednisolone eye drops are contraindicated in individuals who develop hypersensitivity reactions against prednisolone,[26] or individuals with the current conditions, such as tuberculosis of the eye, shingles affecting the eye, raised intraocular pressure, and eye infection caused by fungus.[29]
Prednisolone acetate ophthalmic suspension (eye drops) is an adrenocortical steroid product, prepared as a sterile ophthalmic suspension and used to reduce swelling, redness, itching, and allergic reactions affecting the eye.[30][8][31] It has been explored as a treatment option for bacterial keratitis.[32]
Prednisolone eye drops are used in
Ear drops
In addition, topical prednisolone can also be administered as ear drops.[36]

| Eye Drops | Ear Drops |
|---|---|
| Prednisolone Sodium Phosphate Ophthalmic Solution[26] | Prednisolone Sodium Phosphate 0.5% eye/ ear drops[36] |
| Pred Forte Ophthalmic Suspension (Prednisolone acetate ophthalmic suspension)[30] | |
| Suspension (Prednisolone Mild Ophthalmic)[26] | |
| Suspension (Prednisolone Acetate Ophthalmic)[26] |
Adverse effects
Adverse reactions from the use of prednisolone include:[37][7]
- Increased appetite, weight gain, nausea, and malaise
- Increased risk of infection
- Cardiovascular events
- Dermatological effects including reddening of face, bruising/skin discoloration, impaired wound healing, thinning of skin, skin rash, fluid build up and abnormal hair growth
- Hyperglycemia; patients with diabetes may need increased insulin or diabetic therapies
- Menstrual abnormalities
- Lower response to hormones, especially during stressful instances such as surgery or illness
- Change in electrolytes: rise in blood pressure, increased sodium and low potassium, leading to alkalosis
- GI system effects: swelling of stomach lining, reversible increase in liver enzymes, and risk of stomach ulcers
- Muscular and skeletal abnormalities, such as muscle weakness/muscle loss, osteoporosis (see steroid-induced osteoporosis), long bone fractures, tendon rupture, and back fractures
- Neurological effects, including involuntary movements (convulsions), headaches, and vertigo
- Psychosocial behavioral and emotional disturbances[38] with aggression being one of the most common cognitive symptoms or symptoms overall, especially with oral use.[39]
- Nasal septum perforation and bowel perforation (in some pathologic conditions).[40][41]
Withdrawal from prednisolone after long-term or high-dose use can lead to adrenal insufficiency.[38]
Pregnancy and breastfeeding
Although there are no major human studies of prednisolone use in pregnant women, studies in several animals show that it may cause birth defects including increased likelihood of
Prednisolone is found in breast milk of mothers taking prednisolone.[38]
Local adverse effects in the eye
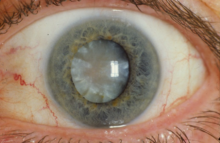
When used topically on the eye, the following are potential side-effects:
- Cataracts: Extended usage of corticosteroids may cause clouding at the back of the lens, also known as posterior subcapsular cataract. This type of cataract reduces the path of light from reaching the eye, which interferes with a person's reading vision. Consumption of prednisolone eye drops post surgery may also retard the healing process.[26]
- Corneal thinning: When corticosteroids are used in the long term, corneal and scleral thinning is also one of its consequences. When not ceased, thinning may ultimately lead to perforation of the cornea.[26]
- Glaucoma: Elongated use of corticosteroids has a chance of causing a raised intraocular pressure (IOP), injuring the optic nerve and weakening of visual awareness. Corticosteroids should be used cautiously in patients with concomitant conditions of glaucoma. Doctors track patients' IOP if they are using corticosteroid eye drops for more than 103 days.[26]
Pharmacology
Pharmacodynamics
As a
Pharmacokinetics
Prednisolone has a relatively short
Prednisolone is 70-90% plasma protein bound, it binds to proteins such as albumin.[45]
Both prednisolone phosphate and prednisolone acetate go through ester hydrolysis in the body to form prednisolone. It subsequently undergoes the usual metabolism of prednisolone. Concomitant use of prednisolone and strong CYP3A4 inhibitors such as ketoconazole is shown to cause a rise in plasma prednisolone concentrations by about 50% owing to a diminished clearance.[44]
Prednisolone predominantly undergoes kidney elimination and is excreted in the urine as sulphate and metabolites of glucuronide conjugate.[45]
Prednisone
Prednisone is a prodrug that is activated in the liver. When it enters the body, prednisone is triggered by the liver and body chemicals to turn into its active form, prednisolone.[47]
Chemistry
Prednisolone is a
Interactions
Drug interactions of prednisolone include other
Contraindications
This section is empty. You can help by adding to it. (October 2023) |
Minor contraindications and drug reactions
Co-administration of prednisolone eye drops with ophthalmic nonsteroidal anti-inflammatory agents (NSAIDs) may perhaps exacerbate its effects, causing unwanted side effects such as toxicity. The wound healing process may also be hindered.[51]
When used in low doses, corticosteroids serve as an anti-inflammatory agent. At higher doses, they are considered as immunosuppressives.[45] Corticosteroids inhibit the inflammatory response to a variety of inciting agents and, it is presumed, delay or slow healing.[52] They inhibit the edema, fibrin deposition, capillary dilation, leukocyte migration, capillary proliferation, fibroblast proliferation, deposition of collagen, and scar formation with inflammation.[53]
Special populations
Child use
Prolonged use of prednisolone eye drops in children may lead to a raised intraocular pressure. While this phenomenon is dose dependent, it is shown to have a greater effect especially in children under 6 years of age.[26]
Pregnancy and breastfeeding
Researches on animal reproduction have indicated that there is a trace of teratogenicity when doses are reduced by 10 times of the human recommended dose.[54] There is no sufficient information on human pregnancy at this moment. Use is only recommended when the potential benefits outweigh the potential risks of the pregnant mother and the fetus.[54]
Prednisolone when delivered systemically can be found in the mother's breast milk, however, there is no data provided for the extent of prednisolone found in the system after administering eye drops.[26][54] However, the presence of corticosteroids is recorded when they are administered systemically, and it could possibly affect the fetus' growth.[54] Therefore, use of prednisolone during breastfeeding is not advocated.[54]
Society and culture
Dosage forms
Prednisolone is supplied as oral liquid, oral suspension, oral syrup, oral tablet, and oral disintegrating tablet. It may be a generic medication or supplied as brands Flo-Pred (prednisolone acetate oral suspension),[37] Millipred (oral tablets),[22] Orapred (prednisolone sodium phosphate oral dissolving tablets),[7] Pediapred (prednisolone sodium phosphate oral solution),[38] Veripred 20, Prelone, Hydeltra-T.B.A., Hydeltrasol, Key-Pred, Cotolone, Predicort, Medicort, Predcor, Bubbli-Pred, Omnipred (prednisolone acetate ophthalmic suspension),[8] Pred Mild,[31] Pred Forte,[30] and others.[55]
Athletics
As a glucocorticosteroid, unauthorized or ad hoc use of prednisolone during competition via oral, intravenous, intramuscular or rectal routes is banned under World Anti-Doping Agency (WADA) anti-doping rules.[56]
Veterinary uses
Prednisolone is used in the treatment of inflammatory and allergic conditions in cats, dogs, horses, small mammals such as
It is used in a broad spectrum of diseases, for example, inflammation of scleral tissues, cornea, conjunctiva in dogs.[57] In horses, prednisolone acetate suspensions are priorly used to treat inflammation in the middle layer of the eye, also known as anterior uveitis and equine recurrent uveitis (ERU), which is the leading cause of visual impairment in horses.[58] Prednisolone acetate eye drops are not to be used in other animals such as birds.[57]
Prednisolone acetate eye drops are also prescribed to the dogs and cats to lessen swelling,
Cats with conjunctivitis usually are required to avoid using ophthalmic preparations of
References
- ^ a b "Prednisolone Use During Pregnancy". Drugs. 16 January 2000. Archived from the original on 21 December 2016. Retrieved 9 March 2020.
- S2CID 12218704.
- S2CID 33189235.
- PMID 30083083.
- ^ ISBN 978-924154765-9.
- ^ a b c d e "Prednisolone". The American Society of Health-System Pharmacists. Archived from the original on 23 December 2016. Retrieved 8 December 2016.
- ^ a b c d e "Orapred ODT- prednisolone sodium phosphate tablet, orally disintegrating". DailyMed. 11 September 2019. Archived from the original on 12 May 2021. Retrieved 9 March 2020.
- ^ a b c "Omnipred- prednisolone acetate suspension". DailyMed. 9 September 2019. Archived from the original on 13 May 2021. Retrieved 9 March 2020.
- ^ "Pevanti 10mg Tablets – Summary of Product Characteristics (SPC) – (eMC)". Medicines. UK. 1 December 2014. Archived from the original on 20 December 2016. Retrieved 13 December 2016.
- ^ ISBN 978-940240844-7. Archivedfrom the original on 10 September 2017.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Prednisolone - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- S2CID 24458998.
- S2CID 24656857.
- ^ PMID 24995449.
- S2CID 10687818.
- PMID 19450877.
- PMID 23024586.
- ^ "Prednisolone 5mg Tablets - Summary of Product Characteristics (SmPC) - (emc)". Medicines. UK. Archived from the original on 27 November 2022. Retrieved 27 November 2022.
- PMID 24789154.
- ^ a b "Millipred- prednisolone tablet". DailyMed. NIH. 8 April 2019. Archived from the original on 9 March 2020. Retrieved 9 March 2020.
- PMID 22016125.
- S2CID 206117017.
- ^ AMA Department of Drugs: AMA Drug Evaluations (6th ed.). Chicago, IL: American Medical Association. 1986.
- ^ a b c d e f g h i j "Prednisolone (ophthalmic): Drug information".
- ^ a b "Goodman & Gilman's: The Pharmacological Basis of Therapeutics, 13e > Ocular Pharmacy". AccessPharmacy. Archived from the original on 16 March 2022. Retrieved 16 March 2022.
- ^ a b "Prednisolone (Ophthalmic Route) Side Effects - Mayo Clinic". www.mayoclinic.org. Archived from the original on 25 February 2023. Retrieved 16 March 2022.
- ^ "Common and Rare Side Effects for prednisolone acetate ophthalmic (eye)". www.webmd.com. Archived from the original on 21 May 2022. Retrieved 16 March 2022.
- ^ a b c "Pred Forte- prednisolone acetate suspension/ drops". DailyMed. 19 July 2019. Archived from the original on 9 March 2020. Retrieved 9 March 2020.
- ^ a b "Pred Mild- prednisolone acetate suspension/ drops". DailyMed. 19 July 2019. Archived from the original on 9 March 2020. Retrieved 9 March 2020.
- PMID 25321340.
- S2CID 23472925.
- S2CID 214616647.
- S2CID 90234022.
- ^ a b "BNF: Prednisolone, Medicine Forms". Medicines Complete. Archived from the original on 16 March 2022. Retrieved 14 April 2022.
- ^ a b "Flo-Pred- prednisolone acetate suspension". DailyMed. 28 February 2019. Archived from the original on 9 May 2021. Retrieved 9 March 2020.
- ^ a b c d "Pediapred- prednisolone sodium phosphate solution". DailyMed. 29 January 2020. Archived from the original on 9 May 2021. Retrieved 9 March 2020.
- ^ "Prednisolone (Oral Route)". Mayo Clinic. Retrieved 1 August 2023.
- PMID 7425698.
- PMID 9830677.
- ^ Goldstein BG, Goldstein AO (13 September 2022). Dellavalle RP, Levy ML, Corona R (eds.). "Topical corticosteroids: Use and adverse effects".
- (PDF) from the original on 28 September 2020. Retrieved 27 June 2019.
- ^ PMID 29549463.
- ^ a b c d e "Prednisolone phosphate". go.drugbank.com. Archived from the original on 25 February 2023. Retrieved 16 March 2022.
- ^ "Prednisolone acetate". go.drugbank.com. Archived from the original on 25 February 2023. Retrieved 16 March 2022.
- ^ "What's the Difference Between Prednisolone and Prednisone?". Archived from the original on 10 October 2022. Retrieved 27 November 2022.
- ISBN 978-1-4757-2085-3. Archivedfrom the original on 10 September 2017.
- ISBN 978-3-88763-075-1. Archivedfrom the original on 10 September 2017.
- ^ "Prednisolone Oral: Uses, Side Effects, Interactions, Pictures, Warnings & Dosing - WebMD". www.webmd.com. Retrieved 29 February 2024.
- ^ "Prednisolone (Ophthalmic Route)". Mayo Foundation for Medical Education and Research.
- S2CID 27261595.
- PMID 26333699.
- ^ a b c d e "Prednisolone ophthalmic Use During Pregnancy". Drugs.com. Archived from the original on 25 February 2023. Retrieved 16 March 2022.
- ^ "Prednisolone: Uses, Dosage, Side Effects, Warnings". Drugs.com. 6 July 2022.
- ^ "Prohibited List" (PDF). World Anti-Doping Agency. January 2020. Archived (PDF) from the original on 9 March 2020.
- ^ a b c d "PrednisoLONE Acetate Ophthalmic Suspension". www.vetrxdirect.com. Archived from the original on 25 February 2023. Retrieved 16 March 2022.
- ^ a b c "Prednisolone Acetate Ophthalmic for Veterinary Use". www.wedgewoodpharmacy.com. Archived from the original on 4 July 2022. Retrieved 16 March 2022.
- ^ Gollakner R. "Prednisolone/Prednisone". VCA Hospitals. Archived from the original on 21 June 2021. Retrieved 3 July 2021.
- PMID 34746285.
External links
- "Prednisolone Ophthalmic". MedlinePlus.
- US patent 2837464, Arthur Nobile, "Process for production of dienes by corynebacteria", published 1958-06-03, issued 1958-06-03, assigned to Schering Corp
