Product (chemistry)

Products are the species formed from
|
Spontaneous reaction
|
Catalysed reaction
|
Much of chemistry research is focused on the synthesis and characterization of beneficial products, as well as the detection and removal of undesirable products. Synthetic chemists can be subdivided into research chemists who design new chemicals and pioneer new methods for synthesizing chemicals, as well as process chemists who scale up chemical production and make it safer, more environmentally sustainable, and more efficient.[3] Other fields include natural product chemists who isolate products created by living organisms and then characterize and study these products.
Determination of reaction
The products of a chemical reaction influence several aspects of the reaction. If the products are lower in energy than the reactants, then the reaction will give off excess energy making it an
If the products are higher in chemical energy than the reactants then the reaction will require energy to be performed and is therefore an endergonic reaction. Additionally if the product is less stable than a reactant, then Leffler's assumption holds that the transition state will more closely resemble the product than the reactant.[6] Sometimes the product will differ significantly enough from the reactant that it is easily purified following the reaction such as when a product is insoluble and precipitates out of solution while the reactants remained dissolved.
History
Ever since the mid-nineteenth century, chemists have been increasingly preoccupied with synthesizing chemical products.[7] Disciplines focused on isolation and characterization of products, such as natural products chemists, remain important to the field, and the combination of their contributions alongside synthetic chemists has resulted in much of the framework through which chemistry is understood today.[7]
Much of
Biochemistry
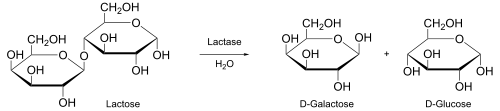
In
- Where S is substrate, P is product and E is enzyme.
Product promiscuity
Some enzymes display a form of promiscuity where they convert a single substrate into multiple different products. It occurs when the reaction occurs via a high energy transition state that can be resolved into a variety of different chemical products.[9]
Product inhibition
Some enzymes are inhibited by the product of their reaction binds to the enzyme and reduces its activity.[10] This can be important in the regulation of metabolism as a form of negative feedback controlling metabolic pathways.[11] Product inhibition is also an important topic in biotechnology, as overcoming this effect can increase the yield of a product.[12]
See also
- Chemical reaction
- Chemical equilibrium
- Second law of thermodynamics
References
- ISBN 978-0-9678550-9-7.
- ISBN 978-0-9678550-9-7.
- ^ a b Henry, Celia M. "DRUG DEVELOPMENT". Chemical and Engineering News. Retrieved 13 September 2014.
- ISBN 978-0-9678550-9-7.
- ISBN 978-0-9678550-9-7.
- ISBN 978-0-9678550-9-7.
- ^ S2CID 17719341.
- S2CID 12573784.
- S2CID 4394693.
- PMID 14149677.
- PMID 231784.
- PMID 15939352.



