Progesterone (medication)
Antimineralocorticoid; Neurosteroid | |
| ATC code | |
|---|---|
| Legal status | |
| Legal status | |
IV: 3–90 minutes[17] | |
| Excretion | Bile and urine[18][19] |
| Identifiers | |
| |
dioxane, β-form) | |
| Melting point | 126 °C (259 °F) |
| |
| |
| (verify) | |
Progesterone (P4), sold under the brand name Prometrium among others, is a
Progesterone is
Progesterone was first isolated in pure form in 1934.
Medical uses
Hormone therapy
Menopause
Progesterone is used in combination with an
The REPLENISH trial was the first adequately
Oral progesterone has been found to significantly reduce
The combination of an estrogen and oral progesterone has been found to improve
In the French E3N-EPIC observational study, the risk of diabetes was significantly lower in women on menopausal hormone therapy, including with the combination of an oral or transdermal estrogen and oral progesterone or a progestin.[54]
Transgender women
Progesterone is used as a component of
Because some believe that progestogens are necessary for full breast development, progesterone is sometimes used in transgender women with the intention of enhancing breast development.[55][57][56] However, a 2014 review concluded the following on the topic of progesterone for enhancing breast development in transgender women:[57]
Our knowledge concerning the natural history and effects of different cross-sex hormone therapies on breast development in [transgender] women is extremely sparse and based on low quality of evidence. Current evidence does not provide evidence that progestogens enhance breast development in [transgender] women. Neither do they prove the absence of such an effect. This prevents us from drawing any firm conclusion at this moment and demonstrates the need for further research to clarify these important clinical questions.[57]
Data on menstruating women shows there is no correlation between water retention, and levels of progesterone or estrogen.
Pregnancy support
Vaginally dosed progesterone is being investigated as potentially beneficial in preventing preterm birth in women at risk for preterm birth. The initial study by Fonseca suggested that vaginal progesterone could prevent preterm birth in women with a history of preterm birth.[61] According to a recent study, women with a short cervix that received hormonal treatment with a progesterone gel had their risk of prematurely giving birth reduced. The hormone treatment was administered vaginally every day during the second half of a pregnancy.[62] A subsequent and larger study showed that vaginal progesterone was no better than placebo in preventing recurrent preterm birth in women with a history of a previous preterm birth,[63] but a planned secondary analysis of the data in this trial showed that women with a short cervix at baseline in the trial had benefit in two ways: a reduction in births less than 32 weeks and a reduction in both the frequency and the time their babies were in intensive care.[64]
In another trial, vaginal progesterone was shown to be better than placebo in reducing preterm birth prior to 34 weeks in women with an extremely short cervix at baseline.[65] An editorial by Roberto Romero discusses the role of sonographic cervical length in identifying patients who may benefit from progesterone treatment.[66] A meta-analysis published in 2011 found that vaginal progesterone cut the risk of premature births by 42 percent in women with short cervixes.[67][68] The meta-analysis, which pooled published results of five large clinical trials, also found that the treatment cut the rate of breathing problems and reduced the need for placing a baby on a ventilator.[69]
Fertility support
Progesterone is used for
Birth control
A progesterone vaginal ring is available for birth control when breastfeeding in a number of areas of the world.[26] An intrauterine device containing progesterone has also been marketed under the brand name Progestasert for birth control, including previously in the United States.[73]
Gynecological disorders
Progesterone is used to control persistent
Other uses
Progesterone is of unclear benefit for the reversal of mifepristone-induced abortion.[77] Evidence is insufficient to support use in traumatic brain injury.[78]
Progesterone has been used as a topical medication applied to the scalp to treat female and male pattern hair loss.[79][80][81][82][83] Variable effectiveness has been reported, but overall its effectiveness for this indication in both sexes has been poor.[80][81][84][83]
Breast pain
Progesterone is approved under the brand name Progestogel as a 1%
Premenstrual syndrome
Historically, progesterone has been widely used in the treatment of
Catamenial epilepsy
Progesterone can be used to treat catamenial epilepsy by supplementation during certain periods of the menstrual cycle.[92]
Available forms
Progesterone is available in a variety of different forms, including
In addition to approved pharmaceutical products, progesterone is available in unregulated custom
| Route | Form | Dose | Brand name | Availability[b] |
|---|---|---|---|---|
| Oral | Capsule | 100, 200, 300 mg | Prometrium[c] | Widespread |
| Tablet (SR) | 200, 300, 400 mg | Dubagest SR[c] | India | |
| Sublingual | Tablet | 10, 25, 50, 100 mg | Luteina[c] | Europe[d] |
Transdermal
|
Gel[e] | 1% (25 mg) | Progestogel | Europe |
Vaginal
|
Capsule | 100, 200 mg | Utrogestan | Widespread |
| Tablet | 100 mg | Endometrin[c] | Widespread | |
| Gel | 4, 8% (45, 90 mg) | Crinone[c] | Widespread | |
| Suppository | 200, 400 mg | Cyclogest | Europe | |
| Ring | 10 mg/day[f] | Fertiring[c] | South America[g] | |
| Rectal | Suppository | 200, 400 mg | Cyclogest | Europe |
Uterine
|
IUD | 38 mg | Progestasert | Discontinued |
Intramuscular
injection |
Oil solution | 2, 5, 10, 20, 25, 50, 100 mg/mL |
Proluton[c] | Widespread |
| Aq. susp. | 12.5, 30, 100 mg/mL | Agolutin[c] | Europe[h] | |
| Emulsion | 5, 10, 25 mg/mL | Di-Pro-Emulsion | Discontinued | |
| Microsph. | 20, 100 mg/mL | ProSphere[c] | Mexico | |
| Subcutaneous | Aq. soln. (inj.) | 25 mg/vial | Prolutex | Europe |
| Implant | 50, 100 mg | Proluton[c] | Discontinued | |
Intravenous
|
Aq. soln. (inj.) | 20 mg/mL | Primolut | Discontinued |
Sources and footnotes:
| ||||
Contraindications
Side effects
Progesterone is well tolerated, and many clinical studies have reported no side effects.
Vaginal progesterone may be associated with
Progesterone lacks undesirable
Central depression
Progesterone can produce
Sedation and cognitive and memory impairment with progesterone are attributable to its
Breast cancer
Breast cell proliferation has been found to be significantly increased by the combination of an
In large short-term
Most data on breast density changes and breast cancer risk are with oral progesterone.
| Therapy | <5 years | 5–14 years | 15+ years | |||
|---|---|---|---|---|---|---|
| Cases | RR (95% CI )
|
Cases | RR (95% CI )
|
Cases | RR (95% CI )
| |
| Estrogen alone | 1259 | 1.18 (1.10–1.26) | 4869 | 1.33 (1.28–1.37) | 2183 | 1.58 (1.51–1.67) |
| By estrogen | ||||||
| Conjugated estrogens | 481 | 1.22 (1.09–1.35) | 1910 | 1.32 (1.25–1.39) | 1179 | 1.68 (1.57–1.80) |
| Estradiol | 346 | 1.20 (1.05–1.36) | 1580 | 1.38 (1.30–1.46) | 435 | 1.78 (1.58–1.99) |
| Estropipate (estrone sulfate) | 9 | 1.45 (0.67–3.15) | 50 | 1.09 (0.79–1.51) | 28 | 1.53 (1.01–2.33) |
| Estriol | 15 | 1.21 (0.68–2.14) | 44 | 1.24 (0.89–1.73) | 9 | 1.41 (0.67–2.93) |
| Other estrogens | 15 | 0.98 (0.46–2.09) | 21 | 0.98 (0.58–1.66) | 5 | 0.77 (0.27–2.21) |
| By route | ||||||
| Oral estrogens | – | – | 3633 | 1.33 (1.27–1.38) | – | – |
| Transdermal estrogens
|
– | – | 919 | 1.35 (1.25–1.46) | – | – |
| Vaginal estrogens
|
– | – | 437 | 1.09 (0.97–1.23) | – | – |
| Estrogen and progestogen | 2419 | 1.58 (1.51–1.67) | 8319 | 2.08 (2.02–2.15) | 1424 | 2.51 (2.34–2.68) |
| By progestogen | ||||||
| (Levo)norgestrel | 343 | 1.70 (1.49–1.94) | 1735 | 2.12 (1.99–2.25) | 219 | 2.69 (2.27–3.18) |
| Norethisterone acetate | 650 | 1.61 (1.46–1.77) | 2642 | 2.20 (2.09–2.32) | 420 | 2.97 (2.60–3.39) |
| Medroxyprogesterone acetate | 714 | 1.64 (1.50–1.79) | 2012 | 2.07 (1.96–2.19) | 411 | 2.71 (2.39–3.07) |
| Dydrogesterone | 65 | 1.21 (0.90–1.61) | 162 | 1.41 (1.17–1.71) | 26 | 2.23 (1.32–3.76) |
| Progesterone | 11 | 0.91 (0.47–1.78) | 38 | 2.05 (1.38–3.06) | 1 | – |
| Promegestone | 12 | 1.68 (0.85–3.31) | 19 | 2.06 (1.19–3.56) | 0 | – |
| Nomegestrol acetate | 8 | 1.60 (0.70–3.64) | 14 | 1.38 (0.75–2.53) | 0 | – |
| Other progestogens | 12 | 1.70 (0.86–3.38) | 19 | 1.79 (1.05–3.05) | 0 | – |
| By progestogen frequency | ||||||
| Continuous
|
– | – | 3948 | 2.30 (2.21–2.40) | – | – |
| Intermittent
|
– | – | 3467 | 1.93 (1.84–2.01) | – | – |
| Progestogen alone | 98 | 1.37 (1.08–1.74) | 107 | 1.39 (1.11–1.75) | 30 | 2.10 (1.35–3.27) |
| By progestogen | ||||||
| Medroxyprogesterone acetate | 28 | 1.68 (1.06–2.66) | 18 | 1.16 (0.68–1.98) | 7 | 3.42 (1.26–9.30) |
| Norethisterone acetate | 13 | 1.58 (0.77–3.24) | 24 | 1.55 (0.88–2.74) | 6 | 3.33 (0.81–13.8) |
| Dydrogesterone | 3 | 2.30 (0.49–10.9) | 11 | 3.31 (1.39–7.84) | 0 | – |
| Other progestogens | 8 | 2.83 (1.04–7.68) | 5 | 1.47 (0.47–4.56) | 1 | – |
| Miscellaneous | ||||||
| Tibolone | – | – | 680 | 1.57 (1.43–1.72) | – | – |
| Notes: menopausal hormone therapy and breast cancer risk by the Collaborative Group on Hormonal Factors in Breast Cancer (CGHFBC). Fully adjusted relative risks for current versus never-users of menopausal hormone therapy. Source: See template.
| ||||||
| Study | Therapy | Hazard ratio (95% CI) |
|---|---|---|
| E3N-EPIC: Fournier et al. (2005) | Estrogen alone | 1.1 (0.8–1.6) |
| Estrogen plus progesterone Transdermal estrogen Oral estrogen |
0.9 (0.7–1.2) 0.9 (0.7–1.2) No events | |
| Estrogen plus progestin Transdermal estrogen Oral estrogen |
1.4 (1.2–1.7) 1.4 (1.2–1.7) 1.5 (1.1–1.9) | |
| E3N-EPIC: Fournier et al. (2008) | Oral estrogen alone | 1.32 (0.76–2.29) |
| Oral estrogen plus progestogen Progesterone Dydrogesterone Medrogestone Chlormadinone acetate Cyproterone acetate Promegestone Nomegestrol acetate Norethisterone acetate Medroxyprogesterone acetate |
Not analyzeda 0.77 (0.36–1.62) 2.74 (1.42–5.29) 2.02 (1.00–4.06) 2.57 (1.81–3.65) 1.62 (0.94–2.82) 1.10 (0.55–2.21) 2.11 (1.56–2.86) 1.48 (1.02–2.16) | |
| Transdermal estrogen alone | 1.28 (0.98–1.69) | |
| Transdermal estrogen plus progestogen Progesterone Dydrogesterone Medrogestone Chlormadinone acetate Cyproterone acetate Promegestone Nomegestrol acetate Norethisterone acetate Medroxyprogesterone acetate |
1.08 (0.89–1.31) 1.18 (0.95–1.48) 2.03 (1.39–2.97) 1.48 (1.05–2.09) Not analyzeda 1.52 (1.19–1.96) 1.60 (1.28–2.01) Not analyzeda Not analyzeda | |
| E3N-EPIC: Fournier et al. (2014) | Estrogen alone | 1.17 (0.99–1.38) |
| Estrogen plus progesterone or dydrogesterone | 1.22 (1.11–1.35) | |
| Estrogen plus progestin | 1.87 (1.71–2.04) | |
| CECILE: Cordina-Duverger et al. (2013) | Estrogen alone | 1.19 (0.69–2.04) |
| Estrogen plus progestogen Progesterone Progestins Progesterone derivatives Testosterone derivatives |
1.33 (0.92–1.92) 0.80 (0.44–1.43) 1.72 (1.11–2.65) 1.57 (0.99–2.49) 3.35 (1.07–10.4) | |
| Footnotes: a = Not analyzed, fewer than 5 cases. Sources: See template. | ||
| Study | Therapy | Hazard ratio (95% CI) |
|---|---|---|
| E3N-EPIC: Fournier et al. (2005)a | Transdermal estrogen plus progesterone <2 years 2–4 years ≥4 years |
0.9 (0.6–1.4) 0.7 (0.4–1.2) 1.2 (0.7–2.0) |
| Transdermal estrogen plus progestin <2 years 2–4 years ≥4 years |
1.6 (1.3–2.0) 1.4 (1.0–1.8) 1.2 (0.8–1.7) | |
| Oral estrogen plus progestin <2 years 2–4 years ≥4 years |
1.2 (0.9–1.8) 1.6 (1.1–2.3) 1.9 (1.2–3.2) | |
| E3N-EPIC: Fournier et al. (2008) | Estrogen plus progesterone <2 years 2–4 years 4–6 years ≥6 years |
0.71 (0.44–1.14) 0.95 (0.67–1.36) 1.26 (0.87–1.82) 1.22 (0.89–1.67) |
| Estrogen plus dydrogesterone <2 years 2–4 years 4–6 years ≥6 years |
0.84 (0.51–1.38) 1.16 (0.79–1.71) 1.28 (0.83–1.99) 1.32 (0.93–1.86) | |
| Estrogen plus other progestogens <2 years 2–4 years 4–6 years ≥6 years |
1.36 (1.07–1.72) 1.59 (1.30–1.94) 1.79 (1.44–2.23) 1.95 (1.62–2.35) | |
| E3N-EPIC: Fournier et al. (2014) | Estrogens plus progesterone or dydrogesterone <5 years ≥5 years |
1.13 (0.99–1.29) 1.31 (1.15–1.48) |
| Estrogen plus other progestogens <5 years ≥5 years |
1.70 (1.50–1.91) 2.02 (1.81–2.26) | |
| Footnotes: a = Oral estrogen plus progesterone was not analyzed because there was a low number of women who used this therapy. Sources: See template. | ||
Blood clots
Whereas the combination of estrogen and a progestin is associated with increased risk of
Overdose
Progesterone is likely to be relatively safe in
Interactions
There are several notable
Progesterone is a weak but significant agonist of the pregnane X receptor (PXR), and has been found to induce several hepatic cytochrome P450 enzymes, such as CYP3A4, especially when concentrations are high, such as with pregnancy range levels.[144][145][146][147] As such, progesterone may have the potential to accelerate the metabolism of various medications.[144][145][146][147]
Pharmacology
Pharmacodynamics
Progesterone is a progestogen, or an
The PRs are expressed widely throughout the body, including in the
The activities of progesterone besides those mediated by the PRs and mPRs are also of significance.
There are differences between progesterones and progestins, such as medroxyprogesterone acetate and norethisterone, with implications for pharmacodynamics and pharmacokinetics, as well as for efficacy, tolerability, and safety.[20]
Pharmacokinetics
The
The bioavailability of progesterone was commonly overestimated due to the immunoassay method of analysis failing to distinguish between progesterone itself and its metabolites.[160][129][130] Newer methods have adjusted the oral bioavailbility estimate from 6.2 to 8.6%[161] down to less than 2.4%.[5]
Chemistry
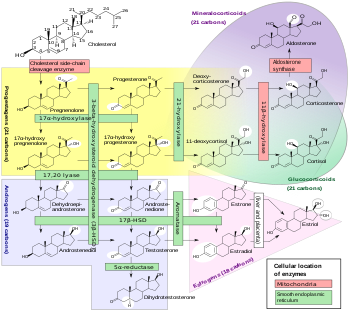
Progesterone is a
Derivatives
A large number of
A variety of synthetic
Synthesis
Chemical syntheses of progesterone have been published.[171]
History
Discovery and synthesis
The hormonal action of progesterone was discovered in 1929.[30][31][172] Pure crystalline progesterone was isolated in 1934 and its chemical structure was determined.[30][31] Later that year, chemical synthesis of progesterone was accomplished.[31][173] Shortly following its chemical synthesis, progesterone began being tested clinically in women.[31][102]
Injections and implants
In 1933 or 1934,
Subcutaneous pellet implants of progesterone were first studied in women in the late 1930s.[184][185][186][187][188] They were the first long-acting progestogen formulation.[189] Pellets were reported to be extruded out of the skin within a few weeks at high rates, even when implanted beneath the deep fascia, and also produced frequent inflammatory reactions at the site of implantation.[107][186][190] In addition, they were absorbed too slowly and achieved unsatisfactorily low progesterone levels.[107] Consequently, they were soon abandoned, in favor of other preparations such as aqueous suspensions.[107][190][191][189] However, subcutaneous pellet implants of progesterone were later studied as a form of birth control in women in the 1980s and early 1990s, though no preparations were ultimately marketed.[192][193][194][195]
In the 1950s, long-acting parenteral
Oral and sublingual
The first study of oral progesterone in humans was published in 1949.
Sublingual progesterone in women was first studied in 1944 by Robert Greenblatt.[241][242][190][225][243][229] Buccal progesterone tablets were marketed by Schering under the brand name Proluton Buccal Tablets by 1949.[244] Sublingual progesterone tablets were marketed under the brand names Progesterone Lingusorbs and Progesterone Membrettes by 1951.[245][246][247] A sublingual tablet formulation of progesterone has been approved under the brand name Luteina in Poland and Ukraine and remains marketed today.[94][95]
Progesterone was the first progestogen that was found to inhibit ovulation, both in animals and in women.
Unfortunately, the use of oral progesterone as a hormonal contraceptive was plagued by problems.
More modern clinical studies of oral progesterone demonstrating elevated levels of progesterone and end-organ responses in women, specifically progestational endometrial changes, were published between 1980 and 1983.[270][271][272][273] Up to this point, many clinicians and researchers apparently still thought that oral progesterone was inactive.[273][274][275] It was not until almost half a century after the introduction of progesterone in medicine that a reasonably effective oral formulation of progesterone was marketed.[103] Micronization of progesterone and suspension in oil-filled capsules, which allowed progesterone to be absorbed several-fold more efficiently by the oral route, was first studied in the late 1970s and described in the literature in 1982.[276][272][277] This formulation, known as oral micronized progesterone (OMP), was then introduced for medical use under the brand name Utrogestan in France in 1982.[272][34][33][22] Subsequently, oral micronized progesterone was introduced under the brand name Prometrium in the United States in 1998.[161][278] By 1999, oral micronized progesterone had been marketed in more than 35 countries.[161] In 2019, the first combination of oral estradiol and progesterone was introduced under the brand name Bijuva in the United States.[11][279]
A
Vaginal, rectal, and uterine
Vaginal progesterone suppositories were first studied in women by Robert Greenblatt in 1954.
Development of a progesterone-containing intrauterine device (IUD) for contraception began in the 1960s.[296] Incorporation of progesterone into IUDs was initially studied to help reduce the risk of IUD expulsion.[296] However, while addition of progesterone to IUDs showed no benefit on expulsion rates, it was unexpectedly found to induce endometrial atrophy.[296] This led in 1976 to the development and introduction of Progestasert, a progesterone-containing product and the first progestogen-containing IUD.[73][296][27] Unfortunately, the product had various problems that limited its use.[296][27][73] These included a short duration of efficacy of only one year, a high cost, a relatively high 2.9% failure rate, a lack of protection against ectopic pregnancy, and difficult and sometimes painful insertions that could necessitate use of a local anesthetic or analgesic.[296][27][73] As a result of these issues, Progestasert never became widely used, and was discontinued in 2001.[296][27][73] It was used mostly in the United States and France while it was marketed.[27]
Transdermal and topical
A topical gel formulation of progesterone, for direct application to the breasts as a local therapy for breast disorders such as
Society and culture
Generic names
Progesterone is the
Brand names
Progesterone is marketed under a large number of
Availability
Progesterone is widely available in countries throughout the world in a variety of formulations.[94][95] Progesterone in the form of oral capsules; vaginal capsules, tablets/inserts, and gels; and intramuscular oil have widespread availability.[94][95] The following formulations/routes of progesterone have selective or more limited availability:[94][95]
- A tablet of micronized progesterone which is marketed under the brand name Luteina is indicated for sublingual administration in addition to vaginal administration and is available in Poland and Ukraine.[94][95]
- A progesterone suppository which is marketed under the brand name Cyclogest is indicated for rectal administration in addition to vaginal administration and is available in Cyprus, Hong Kong, India, Malaysia, Malta, Oman, Singapore, South Africa, Thailand, Tunisia, Turkey, the United Kingdom, and Vietnam.[94][95]
- An
- A non-systemic topical gel formulation of progesterone for local application to the breasts to treat breast pain is marketed under the brand name Progestogel and is available in Belgium, Bulgaria, Colombia, Ecuador, France, Georgia, Germany, Hong Kong, Lebanon, Peru, Romania, Russia, Serbia, Switzerland, Tunisia, Venezuela, and Vietnam.[94][95] It was also formerly available in Italy, Portugal, and Spain, but was discontinued in these countries.[95]
- A progesterone intrauterine device was previously marketed under the brand name Progestasert and was available in Canada, France, the United States, and possibly other countries, but was discontinued.[95][303]
- Progesterone vaginal rings are marketed under the brand names Fertiring and Progering and are available in Chile, Ecuador, and Peru.[94][95]
- A sustained-release tablet formulation of oral micronized progesterone (also known as "oral natural micronized progesterone sustained release" or "oral NMP SR") is marketed in India under the brand names Lutefix Pro (CROSMAT Technology), Dubagest SR, Gestofit SR, and Susten SR, among many others.[280][304][305][306][307][308][309][281][94]
In addition to single-drug formulations, the following progesterone combination formulations are or have been marketed, albeit with limited availability:[94][95]
- A combination pack of progesterone capsules for oral use and estradiol gel for transdermal use is marketed under the brand name Estrogel Propak in Canada.[94][95]
- A combination pack of progesterone capsules and estradiol tablets for oral use is marketed in an under the brand name Duogestan in Belgium.[94][95]
- Progesterone and
- Progesterone and
- Progesterone and estradiol benzoate in an oil solution for use by intramuscular injection is marketed under the brand names Duogynon, Duoton Fort T P, Emmenovis, Gestrygen, Lutofolone, Menovis, Mestrolar, Metrigen Fuerte, Nomestrol, Phenokinon-F, Prodiol, Pro-Estramon-S, Proger F, Progestediol, and Vermagest and is available in Belize, Egypt, El Salvador, Ethiopia, Guatemala, Honduras, Italy, Lebanon, Malaysia, Mexico, Nicaragua, Taiwan, Thailand, and Turkey.[94][95]
- Progesterone and estradiol hemisuccinate in an oil solution for use by intramuscular injection is marketed under the brand name Hosterona in Argentina.[94][95]
- Progesterone and estrone for use by intramuscular injection is marketed under the brand name Synergon in Monaco.[94]
United States
As of November 2016[update], progesterone is available in the United States in the following formulations:[93]
- Oral: Capsules: Prometrium (100 mg, 200 mg, 300 mg)
- Vaginal: Tablets: Endometrin (100 mg); Gels: Crinone (4%, 8%)
- Intramuscular injection: Oil: Progesterone (50 mg/mL)
A 25 mg/mL concentration of progesterone oil for intramuscular injection and a 38 mg/device progesterone intrauterine device (Progestasert) have been discontinued.[93]
An oral combination formulation of micronized progesterone and estradiol in oil-filled capsules (brand name Bijuva) is marketed in the United States for the treatment of menopausal symptoms and endometrial hyperplasia.[311][11]
Progesterone is also available in unregulated custom preparations from
Research
Progesterone was studied as a
Progesterone has been assessed for the suppression of
An
References
- ISBN 978-1-4684-4834-4. Retrieved 4 July 2015.
- ^ "Regulatory Decision Summary for pms-Progesterone". Drug and Health Product Register. 23 October 2014.
- ^ "Reproductive health". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ "Health product highlights 2021: Annexes of products approved in 2021". Health Canada. 3 August 2022. Retrieved 25 March 2024.
- ^ PMID 10689005.
- PMID 30595525.
- S2CID 36586762.
- PMID 24606090.
- ISBN 978-1-4511-4847-3.
- ISBN 978-0-7234-3455-9.
- ^ PMID 25944519.
- ^ a b Khomyak NV, Mamchur VI, Khomyak EV (2014). "Клинико-фармакологические особенности современных лекарственных форм микронизированного прогестерона, применяющихся во время беременности" [Clinical and pharmacological features of modern dosage forms of micronized progesterone used during pregnancy.] (PDF). Доровье [Health]. 4: 90.
- ^ a b "Crinone® 4% and Crinone® 8% (progesterone gel)" (PDF). Watson Pharma, Inc. U.S. Food and Drug Administration. August 2013.
- S2CID 32772029.
- PMID 8513955.
- ^ S2CID 31974637.
The administration of progesterone in injectable or vaginal form is more efficient than by the oral route, since it avoids the metabolic losses of progesterone encountered with oral administration resulting from the hepatic first-pass effect (32). In addition, the injectable forms avoid the need for higher doses that cause a fairly large number of side-effects, such as somnolence, sedation, anxiety, irritability and depression (33).
- ^ PMID 945344.
- ^ "Prometrium (progesterone, USP) Capsules 100 mg" (PDF). Solvay Pharmaceuticals, Inc. U.S. Food and Drug Administration. 1998.
- ^ "Progesterone Injection USP in Sesame Oil for Intramuscular Use Only Rx Only" (PDF). U.S. Food and Drug Administration. January 2007.
- ^ S2CID 24616324.
- ^ PMID 28159148.
- ^ PMID 25113944.
- PMID 26443945.
- ^ S2CID 40753277.
- PMID 28989916.
- ^ ISBN 978-1-4614-6579-9.
- ^ ISBN 978-81-312-1150-2.
- ^ PMID 17924777.
- ^ PMID 27277331.
- ^ ISBN 978-1-4613-2157-6.
- ^ ISBN 978-0-19-513021-8.
- ^ ISBN 978-1-60980-062-8.
- ^ PMID 8616985.
- ^ PMID 6925387.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Progesterone - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ PMID 31282768.
- ^ S2CID 206990125.
- ^ PMID 29630427.
- PMID 31612748.
- PMID 30694918.
- ^ S2CID 4422872.
- S2CID 4419927.
- S2CID 17700754.
- ^ S2CID 20019151.
- ^ S2CID 10982395.
- ^ PMID 29962247.
- PMID 17431228.
- PMID 18374402.
- PMID 26944460.
- PMID 29962257.
- S2CID 20392538.
- S2CID 6077829.
- PMID 25835751.
- ^ a b c d e World Professional Association for Transgender Health (September 2011), Standards of Care for the Health of Transsexual, Transgender, and Gender Nonconforming People, Seventh Version (PDF), archived from the original (PDF) on 6 January 2016
- ^ ISBN 978-1-317-51460-2.
- ^ PMID 24618412.
- PMID 21845193.
- ISBN 978-0-323-29317-4.
Throughout the reproductive years, some women note swelling of the breast around the latter part of each menstrual cycle before the onset of menstruation. The water retention and subsequent swelling of breast tissue during this phase of the menstrual cycle are thought to be due to high levels of circulating progesterone stimulating the secretory cells of the breast.
- S2CID 22293838.
- S2CID 14904733.
- ^ Harris G (2 May 2011). "Hormone Is Said to Cut Risk of Premature Birth". The New York Times. Retrieved 5 May 2011.
- S2CID 31181784.
- S2CID 15577369.
- S2CID 14884358.
- S2CID 46366053.
- PMID 21472815.
- ^ Doheny K (14 December 2011). "Hormone Treatment May Drastically Reduce Preterm Births". WebMD.
- ^ "Progesterone helps cut risk of pre-term birth". Women's health. msnbc.com. 14 December 2011. Archived from the original on 15 December 2011. Retrieved 14 December 2011.
- PMID 25734349.
- PMID 26585269.
- S2CID 3610323.
- ^ ISBN 978-0-323-03309-1.
- PMID 22230306.
- PMID 22972055.
- PMID 7877394.
- PMID 26057457.
- PMID 28005271.
- ISBN 978-0-8247-9363-0.
- ^ PMID 10626111.
- ^ PMID 3063373.
- )
- ^ PMID 8435919.
- S2CID 5094700.
- ^ ISBN 978-94-011-6255-5.
- PMID 26327902.
- ^ ISBN 978-0-7020-4838-8.
- ^ PMID 15008609.
- ^ PMID 12725453.
- PMID 22419287.
- PMID 11588078.
- ISBN 978-1-934559-08-6.
- ^ a b c d "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 29 November 2016.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y "Progesterone".
- ^ a b c d e f g h i j k l m n o p q r s "Micromedex Products: Please Login".
- ^ PMID 26035149.
- ^ PMID 26418479.
- ^ S2CID 28399314.
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 26 July 2018.
- ISBN 978-3-13-179275-4.
- ISBN 978-0-7817-1750-2.
- ^ ISBN 978-93-5270-218-3.
- ^ ISBN 978-1-4471-2392-7.
- ISBN 978-1-139-49285-0.
- ^ "PROSPHERE CARNOT". Archived from the original on 30 December 2019.
- ISBN 978-3-7091-5694-0.
- ^ a b c d e f g Basic Sex Hormone Therapy. Schering A.G. 1962. p. 96.
- ^ a b Current Medicine and Drugs. 1962. p. 40.
Primolut Intravenous (Schering A.G. Berlin)
- ^ S2CID 74224514.
- ^ ISBN 978-0-7817-6587-9.
- ^ ISBN 978-1-111-80040-6.
- ^ ISBN 978-1-930513-83-9.
- ^ ISBN 978-3-211-27063-9.
- ^ PMID 29852783.
- ^ S2CID 34913189.
- ^ PMID 1576050.
- ^ PMID 12627037.
- S2CID 21796848.
- ^ ISBN 978-0-08-015745-0.
- ^ ISBN 978-1-4684-1784-5.
- ^ PMID 13211793.
- PMID 3189454.
- ^ S2CID 5867301.
- PMID 8612843.
- PMID 19709825.
- ^ PMID 29384406.
- ^ S2CID 205631264.
- ^ PMID 24485796.
- ^ S2CID 20808536.
- ^ S2CID 3850275.
- PMID 31474332.
- ^ PMID 27028912.
- ^ S2CID 4229701.
- PMID 23238854.
- ISBN 978-0-12-397769-4.
- S2CID 8221132.
- S2CID 5446017.
- S2CID 19338424.
- PMID 21350928.
- S2CID 195107326.
- PMID 24955220.
- S2CID 5830825.
- PMID 15181090.
- ^ PMID 22837389.
- ^ ISBN 978-3-642-55041-6.
- ^ ISBN 978-0-12-440906-4.
- ^ ISBN 978-1-60913-345-0.
- PMID 23756388.
- PMID 24530924.
- PMID 8282004.
- S2CID 41756726.
- S2CID 14952168.
- S2CID 221753076.
- ^ ISBN 978-0-521-22673-8.
- S2CID 35051390.
- S2CID 207407084.
- ^ ISBN 978-81-8061-427-9.]
It has been observed that micronized progesterone has no suppressive effects on high-density lipoprotein-cholesterol (HDL-C). Jensen et al have proved that oral micronized progesterone has no adverse effect on serum lipids. These preparations have the same antiestrogenic and antimineralocorticoid effect but no androgenic action. It does not affect aldosterone synthesis, blood pressure, carbohydrate metabolism or mood changes. No side effects have been reported as far as lipid profile, coagulation factors and blood pressure are concerned.
[permanent dead link - ISBN 978-0-87983-696-2.
- ^ Progesterone - Drugs.com, retrieved 23 August 2015
- ^ Kuhl H (2011). "Pharmacology of Progestogens" (PDF). Journal für Reproduktionsmedizin und Endokrinologie-Journal of Reproductive Medicine and Endocrinology. 8 (1): 157–177.
- ^ PMID 10090424.
- ^ ISBN 978-1-4757-2085-3.
- ^ ISBN 978-3-88763-075-1.
- PMID 13480263.
- ISSN 0378-5173.
- S2CID 19906601.
- ^ US 9802978, Guthrie DA, Lockwood MA, Natchus MG, Liotta DC, Stein DG, Sayeed I, "Progesterone phosphate analogs and uses related thereto", issued 31 October 2017, assigned to Emory University.
- S2CID 23608386.
- PMID 24900479.
- ^ "Progesterone conjugate - Levolta Pharmaceuticals". AdisInsight. Springer Nature Switzerland AG.
- ISBN 978-3-642-99941-3.
- ISBN 978-1-134-71411-7.
- ISBN 978-1-4684-5275-4.
- ^ ISBN 978-3-642-99941-3.
- ^ ISBN 978-1-4027-8264-0.
- ISBN 978-93-5270-218-3.
- ^ PMID 29648134.
- ^ ISSN 0035-9157.
- ^ ISSN 0025-7125.
- S2CID 25856898.
- PMID 20780598.
- PMID 21313067.
- ISBN 978-0-19-860695-6.
- ISSN 0002-9378.
- ISSN 1471-8391.
- ^ S2CID 71161306.
- PMID 18123090.
- PMID 14862159.
- ^ ISBN 978-3-662-23272-9.
- ^ PMID 13407238.
- S2CID 71319436.
- PMID 3320572.
- S2CID 25152238.
- PMID 7114130.
- PMID 6509984.
- PMID 12999984.
- OCLC 499306481.
- PMID 21025113.
- ^ ISBN 978-3-7091-5694-0.
- S2CID 78850713.
- PMID 14905411.
- PMID 13021207.
- ISBN 978-0-443-04706-0.
- ^ "Progesterone: Uses, Dosage & Side Effects".
- ^ "Agolutin Depot Label" (PDF). www.sukl.cz. Archived from the original (PDF) on 19 May 2019. Retrieved 15 January 2022.
- ^ "Clinomin Forte Label" (PDF). www.indufar.com.py. Archived from the original (PDF) on 18 August 2020. Retrieved 15 January 2022.
- PMID 18140399.
- PMID 14907837.
- ISSN 1935-4657.
- ISBN 978-3-642-94850-3.
- ^ Jores A, Nowakowski H (1960). Praktische Endokrinologie. G. Thieme. p. 295.
- PMID 14810456.
- S2CID 42166026.
- ^ ISBN 978-0-8247-8700-4.
- ^ PMID 8286694.
- ^ S2CID 39442952.
- ^ PMID 8013219.
- PMID 1893701.
- PMID 15432051.
- ^ Basic Sex Hormone Therapy. Schering A.G. 1962. p. 93,96.
Intravenous: The intravenous injection of sex hormones is restricted mainly to specific circumstances where a speedy elevation of hormone levels is required, for example, in treatment of threatened abortion. [...] Crystalline Suspension: With crystalline suspensions the crystalline size governs the rate of absorption and therefore the duration of action. The lack of standardisation of crystalline size in commercial products plus the limits imposed by needle bore, introduces marked variations in effect. The results from emulsified forms are even more unreliable. [...] Hormone Pellets for Implantation: The subcutaneous implantation of sterile tablets was the first means of achieving prolonged action. Such possible factors as encapsulation or extrusion and diminished absorption as the surface area of the pellet is reduced, may be a drawback. Implantation of testosterone (about eight 100 mg. pellets), repeated 6-monthly, is a satisfactory treatment for eunuchoidism and implantation of oestradiol (a 50 mg. pellet remains active for about a year or more) is sometimes a useful procedure. The implantation of progesterone is best discarded altogether; extrusion of pellets (even when placed beneath the deep fascia) and slowness of absorption, in relation to metabolic requirements, make it unsatisfactory and the new depot hormones should be given preference. [...] Sex Hormone Preparations of Schering A.G. Berlin [...] Trade Name: Primolut intravenous. Chemical Description: Progesterone in aqueous solution. Packing: Ampoules of 1 c. c. = 20 mg.
- ^ "Progesterone - IBSA". AdisInsight. Springer Nature Switzerland AG.
- ^ PMID 14164973.
- ^ ISSN 0065-2393.
- )
- ^ PMID 18133494.
- ^ PMID 15436649.
- PMID 14933526.
- PMID 12980155.
- ^ PMID 13084722.
- ^ PMID 13183192.
- ^ PMID 13405054.
- ^ PMID 13477811. Archived from the originalon 2 May 2019. Retrieved 2 May 2019.
- PMID 13504644.
- ISSN 0008-543X.
- ^ ISBN 978-1-5036-0441-4.
- PMID 13030701.
Cyclogesterin. A relatively new approach to progesterone therapy, Cyclogesterin establishes that this hormone can be effective by the oral route. Primarily indicated to induce menstruation in secondary amenorrhea by oral therapy, it contains 30 mg. of progesterone and 1 mg. of mixed natural estrogens per tablet. One tablet is given three times daily for five consecutive days and therapy is stopped. Menstruation follows in one to six days in the non-pregnant patient. The product is manufactured by the Upjohn Company.
- ^ Gutman J (1958). Modern Drug Encyclopedia and Therapeutic Index. Yorke Medical Group. p. 299.
- PMID 13901505.
- PMID 14216475.
- ^ United States. Patent Office (1955). Official Gazette of the United States Patent Office. U.S. Patent Office. pp. 2–.
- ISSN 0021-972X.
- ISSN 0021-972X.
- PMID 13073082.
- ISSN 0095-9561.
- ^ Remington JP, Cook EF, Martin EW (1951). Remington's Practice of Pharmacy: A Treatise on the Preparing, Standardizing, and Dispensing of Official and Extemporaneous Pharmaceutical Products, with Descriptions of Medicinal Substances, Their Properties, Uses and Doses. Also a Guide to Other Professional Services Rendered by the Pharmacist in Connection with Community Health. Intended for the Use of Pharmacists and Physicians and as a Textbook for Students. Mack Publishing Company. pp. 936–937.
- ^ Welsh AL (1951). Dermatological Formulary: A Guide for Medical Students and Resident Physicians in Dermatology. Educational Publishers. p. 155.
- ISBN 978-3-662-25655-8.
- ^ PMID 14232795.
The original observation of Makepeace et al. (1937) that progesterone inhibited ovulation in the rabbit was substantiated by Pincus and Chang (1953). In women, 300 mg of progesterone per day taken orally resulted in ovulation inhibition in 80% of cases (Pincus, 1956). The high dosage and frequent incidence of breakthrough bleeding limited the practical application of the method. Subsequently, the utilization of potent 19-norsteroids, which could be given orally, opened the field to practical oral contraception.
- ^ PMID 356615.
- ISSN 0002-9513.
- ISSN 0002-9513.
- PMID 13138262.
- PMID 13162007.
- ^ PMID 5848673.
At the Fifth International Conference on Planned Parenthood in Tokyo, Pincus (1955) reported an ovulation inhibition by progesterone or norethynodrel1 taken orally by women. This report indicated the beginning of a new era in the history of contraception. [...] That the cervical mucus might be one of the principal sites of action was suggested by the first studies of Pincus (1956, 1959) and of Ishikawa et al. (1957). These investigators found that no pregnancies occurred in women treated orally with large doses of progesterone, though ovulation was inhibited only in some 70% of the cases studied. [...] The mechanism of protection in this method—and probably in that of Pincus (1956) and of Ishikawa et al. (1957)—must involve an effect on the cervical mucus and/or endometrium and Fallopian tubes.
- ^ Pincus G (1955). "Some Effects of Progesterone and Related Compounds upon Reproduction and Early Development in Mammals". The Fifth International Conference on Planned Parenthood: Theme, Overpopulation and Family Planning: Report of the Proceedings, 24-29 October, 1955, Tokyo, Japan. International Planned Parenthood Federation. pp. 175–184.
- ^ Stone A, Kupperman HS (1955). "The Effects of Progesterone on Ovulation: A Preliminary Report". The Fifth International Conference on Planned Parenthood: Theme, Overpopulation and Family Planning: Report of the Proceedings, 24-29 October, 1955, Tokyo, Japan. International Planned Parenthood Federation. p. 185. Archived from the original on 2 May 2019. Retrieved 2 May 2019.
The results of testing the effects of progesterone on ovulation in 13 patients at the Margaret Sanger Research Bureau are presented. The patients had normal menstrual cycles and showed clear evidence of ovulation. Each patient was given 1000 [mg] of [oral] progesterone daily during the midperiod for 10 or 12 days during 16 cycles. Ovulation was inhibited in 6 cycles. No disturbance in menstrual rhythm was observed. 3 of 12 patients with longstanding infertility histories became pregnant within 2–4 months after the cessation of progesterone therapy.
- ^ Ishikawa M, Kyushiro F, Yoshio F, Takashi K, Masanao M, Michio M, et al. (1955). "Some Effects of Progesterone and Related Compounds upon Reproduction and Early Development in Mammals". The Fifth International Conference on Planned Parenthood: Theme, Overpopulation and Family Planning: Report of the Proceedings, 24-29 October, 1955, Tokyo, Japan. International Planned Parenthood Federation. pp. 186–187.
- S2CID 33729147.
- ^ Ishikawa M, Fujii K, Furusawa Y, Kobayashi T, Makino T, Matsumoto S, et al. "Unknown". J. Jap. Family Plann. Ass. 2: 51–56.
- ISSN 0083-6729.
Ishikawa et al. (1957) employing the same regime of progesterone administration also observed suppression of ovulation in a proportion of the cases taken to laparotomy. Although sexual intercourse was practised freely by the subjects of our experiments and those of Ishikawa el al., no pregnancies occurred. Since ovulation presumably took place in a proportion of cycles, the lack of any pregnancies may be due to chance, but Ishikawa et al. (1957) have presented data indicating that in women receiving oral progesterone the cervical mucus becomes impenetrable to sperm.
- S2CID 46312750.
- S2CID 22706524.
- ^ ISBN 978-1-4696-4001-3.
[...] Still, neither of the two researchers was completely satisfied with the results. Progesterone tended to cause "premature menses," or breakthrough bleeding, in approximately 20 percent of the cycles, an occurrence that disturbed the patients and worried Rock.17 in addition, Pincus was concerned about the failure to inhibit ovulation in all the cases. Only large doses of orally administered progesterone could insure the suppression of ovulation, and these doses were expensive. The mass use of this regimen as a birth control method was thus seriously imperiled.18 [...]
- ISBN 978-1-4214-0208-6.
43. The first study used progesterone continuously rather than cyclically. Women began by taking 5 mg of stilbestrol and 50 mg of progesterone, increasing the dose of stilbestrol by 5 mg and of progesterone by 50 mg every two weeks. By the end of twelve weeks, women were taking 30 mg stilbestrol and 300 mg of progesterone. If they had vaginal bleeding at any time, the doses were increased. "Pseudopregnancy," typescript, 15 July 1954, GP-LC. Rock also summarizes his early studies in John Rock, Celso-Ramon Garcia, and Gregory Pincus, "Synthetic Progestins in the Normal Human Menstrual Cycle," Recent Progress in Hormone Research, vol. 13 (New York: Academic Press, 1957), 323-24.
- ISBN 978-1-4214-0371-7.
In the early 1950s, independent of Pincus's work in Worcester, Rock successfully induced pregnancy in previously infertile women by treating them for several months with estrogen and progesterone. Although the steroids prevented pregnancy during the course of therapy, some of the women conceived when the treatment ended; this phenomenon became known as the "Rock rebound effect."58 When Pincus learned of Rock's work, he asked the physician to join forces in the hunt for an ovulation inhibitor, and Rock agreed. Pincus suggested two changes in the experimental regimen: use only progesterone (estrogen promoted cancer in laboratory animals) and administer the hormone for twenty days each month (to allow a period of menstruation). Rock achieved the same rate of success in curing infertility (about 15%), but a significant problem remained: tests indicated that about 15 percent of the women ovulated while taking the progesterone.59 Pincus and Rock needed to find an orally active compound that would completely inhibit ovulation. It was time to test the 19-nor steroids in humans. [...]
- S2CID 31577493.
- PMID 13380401.
- ISBN 978-0-300-16791-7.
- PMID 23384741.
- PMID 7370683.
- PMID 6784875.
- ^ PMID 7119381.
- ^ PMID 6315123.
- PMID 6991820.
It is generally accepted that orally administered progesterone has little biological effect.
- PMID 7193749.
- S2CID 44066213.
- PMID 2801843.
- ^ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations: 019781". Food and Drug Administration. 2 July 2010. Retrieved 7 July 2010.
- ^ "TherapeuticsMD Announces FDA Approval of TX-001HR: BIJUVA™ (Estradiol and Progesterone) Capsules for the Treatment of Moderate to Severe Vasomotor Symptoms Due to Menopause".
- ^ PMID 27042538.
- ^ a b c Kirk EP, Serat S, Burrows LJ, Mott LA, Yeo KJ, Fitzmaurice T, et al. (1997). "A pharmacokinetic study of micronized natural progesterone extended release tablets". Restore Health. Archived from the original on 4 March 2019.
- PMID 13211792.
- ^ PMID 6301793.
- ISSN 0095-9561.
- ISSN 0804-4643.
- ^ Unlisted Drugs. Unlisted Drugs. 1976. p. 360.
- ^ The Belfast Gazette. H.M. Stationery Office. January 1977. p. 158.
- ^ Negwer M (1978). Organic-chemical Drugs and Their Synonyms. Akademie-Verlag. p. 872.
- ISBN 978-1-4419-8456-2.
- ^ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations: 020701". Food and Drug Administration. 2 July 2010. Retrieved 7 July 2010.
- ^ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations: 022057". Food and Drug Administration. 2 July 2010. Retrieved 7 July 2010.
- ^ S2CID 72093345.
- PMID 26732558.
- ISSN 1810-2107.
- ISBN 978-1-4822-1772-8.
- ^ PMID 19445984.
- ^ Journal de Gynécologie, Obstétrique et Biologie de la Reproduction. Masson: 198, 214, 327. 1972 https://books.google.com/books?id=NjH9dmzFBfsC.
{{cite journal}}: Missing or empty|title=(help) - PMID 2642780.
- S2CID 23411589.
- .
- S2CID 24890723.
- ISBN 978-0-7514-0499-9.
- ^ Gelijns A (1991). Innovation in Clinical Practice: The Dynamics of Medical Technology Development. National Academies. pp. 195–. NAP:13513.
- ISSN 2319-5886.
- ^ Haleem S, Khan MI (March 2015). "Changing Indian Market Trends of NMP: A Review". International Journal of Pharma Research & Review. 4 (3): 28–30.
- ^ Nigam A (2018). "Luteal Phase Support: Why, When and How" (PDF). Pan Asian Journal of Obstetrics & Gynecology. 1 (2): 79–83. Archived from the original (PDF) on 19 August 2020.
- PMID 26894126.
- ISSN 2249-782X.
- ^ Singh N, Reddy A (April–June 2015). "Current Concepts in Management of Preterm Labour - A Review Article". Indian Obstetrics and Gynaecology. 5 (2).
- ^ "Estradiol/progesterone injection - Laboratorios Carnot". AdisInsight. Springer Nature Switzerland AG.
- ^ "Estradiol/progesterone - TherapeuticsMD". AdisInsight. Springer Nature Switzerland AG.
- ^ ISBN 978-0-12-137250-7.
- ^ S2CID 24859886.
- PMID 5558416.
- S2CID 32637425.
- PMID 14400846.
- ^ a b "Progesterone topical - Novavax". AdisInsight. Springer Nature Switzerland AG.
- ISBN 978-1-59392-004-3.
- ^ "Form 10-K Novavax, inc". ir.novavax.com. Archived from the original on 22 December 2019. Retrieved 15 January 2022.
Further reading
- Sitruk-Ware R, Bricaire C, De Lignieres B, Yaneva H, Mauvais-Jarvis P (October 1987). "Oral micronized progesterone. Bioavailability pharmacokinetics, pharmacological and therapeutic implications--a review". Contraception. 36 (4): 373–402. PMID 3327648.
- Ruan X, Mueck AO (November 2014). "Systemic progesterone therapy--oral, vaginal, injections and even transdermal?". Maturitas. 79 (3): 248–255. PMID 25113944.
