Proinsulin
| insulin | |||||||
|---|---|---|---|---|---|---|---|
Chr. 11 p15.5 | |||||||
| |||||||
Proinsulin is the prohormone precursor to insulin made in the beta cells of the Pancreatic Islets, specialized regions of the pancreas. In humans, proinsulin is encoded by the INS gene.[1][2] The pancreatic islets only secrete between 1% and 3% of proinsulin intact.[3] However, because proinsulin has a longer half life than insulin, it can account for anywhere from 5–30% of the insulin-like structures circulating in the blood.[3] There are higher concentrations of proinsulin after meals and lower levels when a person is fasting.[3] Additionally, while proinsulin and insulin have structural differences, proinsulin does demonstrate some affinity for the insulin receptor. Due to the relative similarities in structure, proinsulin can produce between 5% and 10% of the metabolic activity similarly induced by insulin.[3]
Proinsulin is the final single chain protein structure secreted by cells before cleavage into mature insulin.[4] Proinsulin was discovered by Professor Donald F. Steiner of the University of Chicago in 1967.[5]
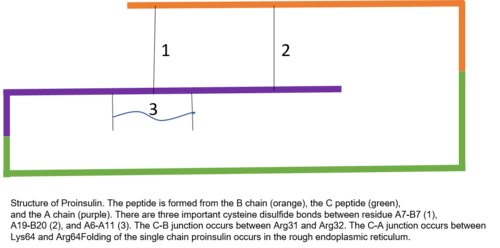
Structure
Proinsulin is made up of 86
The C peptide is between the A and B chains of proinsulin.[7] The connection between the A chain and C peptide is much more stable than the junction between the C peptide and B chain, with alpha helical features being exhibited near the C peptide-A chain connection.[10] The C peptide-A chain junction occurs between residues 64 and 65 of proinsulin. These are lysine and arginine molecules, respectively.[10] The C peptide-B chain connection is between two arginine residues at positions 31 and 32 of proinsulin.[10]
There is conservation of much of the structure of proinsulin among mammalian species, with much of the residue changes seen from one species to another present in the C peptide.[8][11] That said, the residues of the C peptide that are conserved across species interact with similarly conserved residues on the A and B chains.[8] Thus, it is hypothesized that these conserved residues are important for the functionality of mature insulin.[8]
-
3D Model of proinsulin - A chain is in blue, b chain in red, c peptide in orange. The dibasic cleavage for c peptide and a chain is in green KR (lysine and arginine), the one for c peptide and b chain is in cyan RR (arginine).
Synthesis and Post-translational Modification
Proinsulin is synthesized on membrane associated ribosomes found on the
The
Immunogenicity
When insulin was originally purified from
Medical Relevance
Historically, the focus of many insulin related metabolic diseases has focused on mature insulin. However, in recent years the importance of studying the structure and function of proinsulin or proinsulin:insulin ratio[18] in relation to these diseases has become increasingly clear.
Diabetes Mellitus
Increased levels of proinsulin in the circulatory system relative to mature insulin concentrations can indicate impending insulin resistance and the development of type 2 diabetes.[19] Additional problems with proinsulin that can lead to diabetes include mutations in the number of cysteines present, which could affect correct folding.[9] If the mutation causes only a mild change it could simply stress the endoplasmic reticulum’s ability to properly fold the protein.[9] This stress, after a while, would lead to a decrease in the number of β-cells producing mature insulin, and would then lead to diabetes mellitus.[9]
Neonatal Diabetes Mellitus
Postnatal proinsulin is crucial for metabolic regulation. However, proinsulin in
See also
- insulin
- preproinsulin
- signal peptide
- signal peptide peptidase
- proprotein convertase 1 (PC1)
- proprotein convertase 2 (PC2)
References
- ^ "Entrez Gene: INS insulin".
- S2CID 4363706.
- ^ a b c d "Interpretation for 80908 Proinsulin, Plasma". www.mayomedicallaboratories.com. Archived from the original on 2017-10-04. Retrieved 2017-03-09.
- PMID 40897.
- PMID 25561547.
- ^ Universal protein resource accession number P01308 for " INS_HUMAN" at UniProt.
- ^ PMID 4928892.
- ^ PMID 808541.
- ^ PMID 19395706.
- ^ PMID 20106974.
- S2CID 4363706.
- PMID 8119968.
- ^ PMID 7575420.
- PMID 3905477.
- ^ Tanyolac S, Goldfine ID, Kroon L. "Insulin Pharmacology, Type of Regimens and Adjustments". Endotext.com. Archived from the original on 2011-07-25. Retrieved 2011-03-18.
- S2CID 28616749.
- PMID 3924216.
- S2CID 52058695.
- S2CID 44874023.
- ^ PMID 16596360.