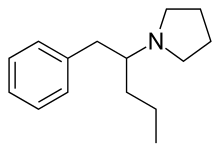
Prolintane
Source: Wikipedia, the free encyclopedia.
Stimulant and NDRI drug
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | oral, intranasal, rectal |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
JSmol) | |
| Melting point | 133 °C (271 °F) |
| Boiling point | 153 °C (307 °F) |
| |
| |
| | |
Prolintane (Catovit, Katovit, Promotil, Villescon) is a
MDPV, and propylhexedrine and it has a similar mechanism of action.[4] Many cases of prolintane abuse have been reported.[5]
Under the trade-name "Katovit", prolintane was commercialized by the Spanish pharmaceutical company, FHER. Katovit was sold until 2001, and was most often used by students and workers as a stimulant to provide energy, promote alertness and concentration.[medical citation needed]
See also
- α-PVP (β-ketone-prolintane, prolintanone)
- Methylenedioxypyrovalerone (MDPV)
- Pyrovalerone (Centroton, Thymergix)
- Phenylpropylaminopentane
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- PMID 4392006.
- ^ GB Patent 807835
- PMID 7437258.
- PMID 17725890.
