Prostacyclin
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Flolan, Veletri |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 42 seconds |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Prostacyclin (also called prostaglandin I2 or PGI2) is a prostaglandin member of the eicosanoid family of lipid molecules. It inhibits platelet activation and is also an effective vasodilator.
When used as a drug, it is also known as epoprostenol.[1] The terms are sometimes used interchangeably.[2]
Function
Prostacyclin chiefly prevents formation of the
Medical uses
It is used to treat
Degradation
Prostacyclin, which has a half-life of 42 seconds,[7] is broken down into 6-keto-PGF1, which is a much weaker vasodilator. A way to stabilize prostacyclin in its active form, especially during drug delivery, is to prepare prostacyclin in alkaline buffer. Even at physiological pH, prostacyclin can rapidly form the inactive hydration product 6-keto-prostaglandin F1α.[8]
Mechanism
Prostacyclin effect Mechanism Cellular response Classical
functionsVessel tone ↑cAMP, ↓ET-1
↓Ca2+, ↑K+↓SMC proliferation
↑VasodilationAntiproliferative ↑cAMP
↑PPARgamma↓Fibroblast growth
↑ApoptosisAntithrombotic ↓Thromboxane-A2
↓PDGF↓Platelet aggregation
↓Platelet adherence to vessel wallNovel
functionsAntiinflammatory ↓IL-1, IL-6
↑IL-10↓Proinflammatory cytokines
↑Antiinflammatory cytokinesAntimitogenic ↓VEGF
↓TGF-β↓Angiogenesis
↑ECM remodeling
As mentioned above, prostacyclin (PGI2) is released by healthy endothelial cells and performs its function through a
Members[9]
| PROSTACYCLINS | |||
|---|---|---|---|
| Flolan (epoprostenol sodium) for Injection |
Continuously infused | 2 ng/kg/min to start, increased by 2 ng/kg/min every 15 minutes or longer until suitable efficacy/tolerability balance is achieved | Class III Class IV |
| Veletri (epoprostenol) for Injection |
Continuously infused | 2 ng/kg/min to start, increased by 2 ng/kg/min every 15 minutes or longer until suitable efficacy/tolerability balance is achieved | Class III Class IV |
| Remodulin SC§ (treprostinil sodium) Injection |
Continuously infused | 1.25 ng/kg/min to start, increased by up to 1.25 ng/kg/min per week for 4 weeks, then up to 2.5 ng/kg/min per week until suitable efficacy/tolerability balance is achieved | Class II Class III Class IV |
| Ventavis (iloprost) Inhalation Solution |
Inhaled 6–9 times daily | 2.5 μg 6–9 times daily to start, increased to 5.0 μg 6–9 times daily if well tolerated | Class III Class IV |
Pharmacology

Synthetic prostacyclin analogues (iloprost, cisaprost) are used intravenously, subcutaneously or by inhalation:
- as a Raynaud's phenomenon or ischemiaof a limb;
- in pulmonary hypertension.
- in primary pulmonary hypertension (PPH)
The production of prostacyclin is inhibited by the action of
In patients with pulmonary hypertension, inhaled epoprostenol reduces pulmonary pressure, and improves right ventricular stroke volume in patients undergoing cardiac surgery. A dose of 60 μg is hemodynamically safe, and its effect is completely reversed after 25 minutes. No evidence of platelet dysfunction or an increase in surgical bleeding after administration of inhaled epoprostenol has been found.[10] The drug has been known to cause flushing, headaches and hypotension.[11]
Synthesis
Biosynthesis
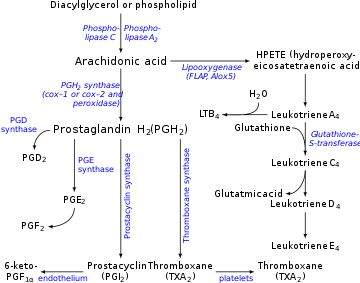
Prostacyclin is produced in endothelial cells, which line the walls of arteries and veins,[12] from prostaglandin H2 (PGH2) by the action of the enzyme prostacyclin synthase. Although prostacyclin is considered an independent mediator, it is called PGI2 (prostaglandin I2) in eicosanoid nomenclature, and is a member of the prostanoids (together with the prostaglandins and thromboxane). PGI2, derived primarily from COX-2 in humans, is the major arachidonate metabolite released from the vascular endothelium. This is a controversial point, some assign COX 1 as the major prostacyclin producing cyclooxygenase in the endothelial cells of the blood vessels.[13]
The series-3 prostaglandin PGH3 also follows the prostacyclin synthase pathway, yielding another prostacyclin, PGI3.[14] The unqualified term 'prostacyclin' usually refers to PGI2. PGI2 is derived from the ω-6 arachidonic acid. PGI3 is derived from the ω-3 EPA.
Artificial synthesis
Prostacyclin can be
Because prostacyclin is so chemically labile, quantitation of their inactive metabolites, rather than the active compounds, is used to assess their rate of synthesis.[17]
History
During the 1960s, a UK research team, headed by Professor John Vane, began to explore the role of prostaglandins in anaphylaxis and respiratory diseases. Working with a team from the Royal College of Surgeons, Vane discovered that aspirin and other oral anti-inflammatory drugs work by inhibiting the synthesis of prostaglandins. This critical finding opened the door to a broader understanding of the role of prostaglandins in the body.
A team at The Wellcome Foundation led by Salvador Moncada had identified a lipid mediator they called "PG-X," which inhibits platelet aggregation. PG-X, later known as prostacyclin, is 30 times more potent than any other then-known anti-aggregatory agent. They did this while searching for an enzyme that generates a fellow unstable prostanoid, Thromboxane A2[18]
In 1976, Vane and fellow researchers Salvador Moncada, Ryszard Gryglewski, and Stuart Bunting published the first paper on prostacyclin in Nature.[19] The collaboration produced a synthesized molecule, which was named epoprostenol. But, as with native prostacyclin, the epoprostenol molecule is unstable in solution and prone to rapid degradation.[citation needed] This presented a challenge for both in vitro experiments and clinical applications.
To overcome this challenge, the research team that discovered prostacyclin continued the research. The research team synthesized nearly 1,000 analogues.[citation needed]
References
- ^ "epoprostenol" at Dorland's Medical Dictionary
- PMID 1883670.
- ^ Pathologic Basis of Disease, Robbins and Cotran, 8th ed. Saunders Philadelphia 2010
- ^ "Epoprostenol Sodium Monograph for Professionals". Drugs.com. AHFS. 6 April 2020. Retrieved 22 October 2020.
- ^ "Flolan- epoprostenol sodium injection, powder, lyophilized, for solution Diluent- water solution". DailyMed. 15 November 2019. Retrieved 22 October 2020.
- ^ PMID 21687516.
- S2CID 25410558.
- PMID 6354353.
- ^ ^ REM_RefGuideWC_AUG07v.1
- PMID 12658208.
- ^ Nickson, C. (2015, October 28). Prostacyclin or Epoprostenol. Retrieved November 16, 2015, from http://lifeinthefastlane.com/ccc/prostacyclin-or-epoprostenol/
- ^ prostacyclin. (n.d.) Miller-Keane Encyclopedia and Dictionary of Medicine, Nursing, and Allied Health, Seventh Edition. (2003). Retrieved November 17, 2015 from http://medical-dictionary.thefreedictionary.com/prostacyclin
- PMID 23045674.
- PMID 2996649.
- .
- ^ Nickson C (15 October 2015). "Prostacyclin or Epoprostenol". Life in the Fast Lane. Archived from the original on 28 March 2015. Retrieved 16 November 2015.
- .
- PMID 1883670.
- S2CID 4279030.
External links
- "Epoprostenol". Drug Information Portal. U.S. National Library of Medicine.
- "Epoprostenol sodium". Drug Information Portal. U.S. National Library of Medicine.
