Pyrovalerone
Source: Wikipedia, the free encyclopedia.
Chemical compound
 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
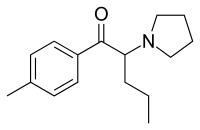
Pyrovalerone (Centroton, 4-Methyl-β-keto-prolintane, Thymergix, O-2371)MDPV and prolintane (Promotil, Katovit).
muscle tremors. Withdrawal following abuse upon discontinuation often results in depression
.
The R-enantiomer of pyrovalerone is devoid of pharmacologic activity.[5]
See also
Look up pyrovalerone in Wiktionary, the free dictionary.
- 4-Et-PVP
- α-Pyrrolidinohexiophenone (α-PHP)
- α-Pyrrolidinopentiothiophenone (α-PVT)
- Methylenedioxypyrovalerone (MDPV)
- Naphyrone (O-2482)
- Prolintane (Promotil, Katovit)
- 4'-Methyl-α-pyrrolidinohexiophenone (MPHP, 4-MPHP)
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ US Patent 3314970
- PMID 4402508.
- PMID 9895.
- PMID 16480278.
antagonists
receptor agonists
- Beloranib§
- Bimagrumab§
- Desiccated thyroid‡
- Metformin
- Metreleptin
- Naltrexone
- Setmelanotide
- Topiramate
- ZGN-1061
- Zonisamide
- Water
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
gastrointestinal system is a stub. You can help Wikipedia by expanding it. |
