Quinone
The quinones are a class of
The archetypical member of the class isThe name is derived from that of
Properties
Quinones are oxidized derivatives of aromatic compounds and are often readily made from reactive aromatic compounds with
The term quinone is also used more generally for a large class of compounds formally derived from aromatic quinones through replacement of some hydrogen atoms by other atoms or radicals.
-
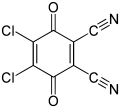
Chloranil, a reagent in organic chemistry
-
Lawsone, a dye present in the leaves of the henna plant
-
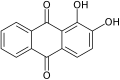
Alizarin, a common red dye
-
DDQ, a reagent in organic chemistry
-
Daunorubicin, an anticancer drug
Reactions
Quinones form polymers by formation of hydrogen bonds with ρ-hydroquinone.[5]
Reduction
Quinones are oxidizing agents, sometimes reversibly so. Relative to benzoquinone, more strongly oxidizing quinones include chloranil and 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (also known as DDQ).[6]
The oxidizing power of quinones is enhanced by the presence of acids.[7] In acidic conditions, quinone undergoes two-electron and two-proton reduction to hydroquinone.

In alkaline conditions, quinones undergo a reversible single-step, two-electron reduction. In neutral conditions, quinones may undergo either a one-proton, two-electron reduction or a two-electron reduction. In
9,10-Anthraquinone-2,7-disulphonic acid (AQDS) a quinone similar to one found naturally in rhubarb has been used as a charge carrier in metal-free flow batteries.[9]
Addition
Quinones undergo addition reaction to form 1,4-addition products.[10] An example of 1,4-addition reaction is the addition of hydrogen chloride to form chlorohydroquinone:

Quinones can undergo Diels–Alder reactions.[10] The quinone acts as the dienophile and reacts with a diene at a carbon-carbon double bond.
In
Occurrence and uses
Production of hydrogen peroxide
A large scale industrial application of quinones is for the production of hydrogen peroxide. 2-Alkylanthraquinones are hydrogenated to the corresponding hydroquinones (quinizarins), which then transfer H
2 to oxygen:
- dihydroanthraquinone + O
2 → anthraquinone + H
2O
2
in this way, several million metric tons of H
2O
2 are produced annually.[12]
1,4-Naphthoquinone, derived by oxidation of naphthalene with chromium trioxide.[13] It is the precursor to anthraquinone.
Biochemistry
Numerous quinones are significant roles in biology. Vitamin K, which involved in coagulation of blood, is a quinone.

Quinones are conjectured to occur in all respiring organisms.
The auto-oxidation of the neurotransmitter dopamine and its precursor L-Dopa generates the comparatively stable dopamine quinone which inhibits the functioning of dopamine transporter (DAT) and the TH enzyme and leads to low mitochondrial ATP production.[15]
The benzoquinone blattellaquinone is a sex pheromone in cockroaches. In the spray of bombardier beetles, hydroquinone reacts with hydrogen peroxide to produce a fiery blast of steam, a deterrent in the animal world.
Medical
Several quinones are of pharmacological interest. They form a major class of anticancer cytotoxins. One example is
Another quinone-containing drug is Mecarbinate (dimecarbine), made by the reaction of ethyl N-methyl-β-aminocrotonate with para-benzoquinone. Others include Amendol, Oxyphemedol, Phemedol all in FR5142 (M) ― 1967-06-05.[clarification needed] Note: These are all indoles made via the Nenitzescu indole synthesis. The antineoplastic Apaziquone.
Benzoquinone compounds are a metabolite of paracetamol.[17]
Dyes
Many natural and artificial coloring substances (
Photography
A commercial application of quinones is in black-and-white photography. Black-and-white film is covered with an emulsion containing silver bromide or silver iodide crystals, which exposure to light activates. Hydroquinone is used to reduce the activated silver ions to metallic silver. During this process, hydroquinone is oxidized to quinone. All silver halide not activated by light or reduced by hydroquinone is removed, leaving a negative by deposited silver where the film had been struck by light.[18]
Nomenclature
Quinones are commonly named with a prefix that indicates the parent aromatic hydrocarbon ("benzo-" for benzene, "naphtho-" for naphthalene, "anthra-" for anthracene, etc.) and the "-quinone" suffix. Infix multipliers "-di-", "-tri-", "-tetra-" (etc.) are used when there are 4, 6, 8 (etc.) carbonyls. The position of the carbonyl groups can be indicated before the prefix (as in "1,4,5,8-naphthodiquinone") or after it ("anthra-1,4-quinone").
Structural analogues of quinones
- Quinone methide – where one O is replaced by C
- Xylylene – where both O's are replaced by C's
- Quinone imine – where one O is replaced by N, illustrated by NAPQI
- Quinone diimine – where both O's are replaced by N's, illustrated by the antiseptic ambazone
- Azaxylylene – where both O's are replaced by one N and one C, illustrated by various fuchsine dyes like pararosaniline
References
- ISBN 9780470772119.
- ISBN 9780470772126.
- ^ The Chemical News and Journal of Physical Science. Griffin, Bohn and Company. 1773.
- .
- OCLC 642506595
- ISSN 2090-3529.
- ISSN 1932-7447.
- S2CID 4459692.
- ^ ISBN 978-0-08-012948-8, retrieved 2022-11-17
- S2CID 1362687.
- .
- ; Collected Volumes, vol. 4, p. 698.
- ^ PMID 1913977.
- PMID 24653659.
- ^ Liu H., "Extraction and Isolation of Compounds from Herbal Medicines" in 'Traditional Herbal Medicine Research Methods', ed by Willow JH Liu 2011 John Wiley and Sons, Inc.
- PMID 6424115.
- OCLC 974377227.
External links
- Quinones at the U.S. National Library of Medicine Medical Subject Headings (MeSH)