Carbon-14
 | |
| General | |
|---|---|
| Symbol | 14C |
| Names | carbon-14, 14C, C-14, radiocarbon |
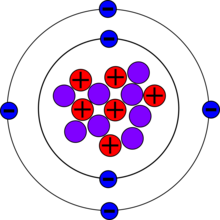
| Protons (Z) | 6 |
| Neutrons (N) | 8 |
| Nuclide data | |
| Natural abundance | 1 part per trillion = |
Decay mode | Decay energy (MeV) |
| Beta | 0.156476[2] |
| Isotopes of carbon Complete table of nuclides | |
Carbon-14, C-14, 14
C or radiocarbon, is a
There are three naturally occurring
The different isotopes of carbon do not differ appreciably in their chemical properties. This resemblance is used in chemical and biological research, in a technique called carbon labeling: carbon-14 atoms can be used to replace nonradioactive carbon, in order to trace chemical and biochemical reactions involving carbon atoms from any given organic compound.
Radioactive decay and detection
Carbon-14 goes through radioactive beta decay:
- 14
6C → 14
7N + e− +
ν
e + 156.5 keV
By emitting an
As usual with beta decay, almost all the decay energy is carried away by the beta particle and the neutrino. The emitted beta particles have a maximum energy of about 156 keV, while their weighted mean energy is 49 keV.[8] These are relatively low energies; the maximum distance traveled is estimated to be 22 cm in air and 0.27 mm in body tissue. The fraction of the radiation transmitted through the dead skin layer is estimated to be 0.11. Small amounts of carbon-14 are not easily detected by typical Geiger–Müller (G-M) detectors; it is estimated that G-M detectors will not normally detect contamination of less than about 100,000 disintegrations per minute (0.05 µCi). Liquid scintillation counting is the preferred method[9] although more recently, accelerator mass spectrometry has become the method of choice; it counts all the carbon-14 atoms in the sample and not just the few that happen to decay during the measurements; it can therefore be used with much smaller samples (as small as individual plant seeds), and gives results much more quickly. The G-M counting efficiency is estimated to be 3%. The half-distance layer in water is 0.05 mm.[10]
Radiocarbon dating
Radiocarbon dating is a
One of the frequent uses of the technique is to date organic remains from archaeological sites. Plants
C in plants and animals when they die approximately equals the level of 14
C in the atmosphere at that time. However, it decreases thereafter from radioactive decay, allowing the date of death or fixation to be estimated. The initial 14
C level for the calculation can either be estimated, or else directly compared with known year-by-year data from tree-ring data (dendrochronology) up to 10,000 years ago (using overlapping data from live and dead trees in a given area), or else from cave deposits (speleothems), back to about 45,000 years before the present. A calculation or (more accurately) a direct comparison of carbon-14 levels in a sample, with tree ring or cave-deposit carbon-14 levels of a known age, then gives the wood or animal sample age-since-formation. Radiocarbon is also used to detect disturbance in natural ecosystems; for example, in peatland landscapes, radiocarbon can indicate that carbon which was previously stored in organic soils is being released due to land clearance or climate change.[14][15]
Cosmogenic nuclides are also used as proxy data to characterize cosmic particle and solar activity of the distant past.[16][17]
Origin
Natural production in the atmosphere

2: Decay of carbon-14
3: The "equal" equation is for living organisms, and the unequal one is for dead organisms, in which the C-14 then decays (See 2).
Carbon-14 is produced in the upper
- 14
7N + n → 14
6C + p
The highest rate of carbon-14 production takes place at altitudes of 9 to 15 kilometres (30,000 to 49,000 ft) and at high geomagnetic latitudes.
The rate of 14
C production can be modelled, yielding values of 16,400
C increase (2%) has been associated with a 5480 BC event, which is unlikely to be a solar energetic particle event.[25]
Carbon-14 may also be produced by lightning[26][27] but in amounts negligible, globally, compared to cosmic ray production. Local effects of cloud-ground discharge through sample residues are unclear, but possibly significant.
Other carbon-14 sources
Carbon-14 can also be produced by other neutron reactions, including in particular
C production by thermal neutron irradiation of targets (e.g., in a nuclear reactor) are summarized in the table.
Carbon-14 may also be radiogenic (cluster decay of 223
Ra, 224
Ra, 226
Ra). However, this origin is extremely rare.
| Parent isotope | Natural abundance, % | Cross section for thermal neutron capture, b | Reaction |
|---|---|---|---|
| 14 N |
99.634 | 1.81 | 14 N(n,p)14 C |
| 13 C |
1.103 | 0.0009 | 13 C(n,γ)14 C |
| 17 O |
0.0383 | 0.235 | 17 O(n,α)14 C |
Formation during nuclear tests

C, New Zealand[30] and Austria.[31] The New Zealand curve is representative for the Southern Hemisphere, the Austrian curve is representative for the Northern Hemisphere. Atmospheric nuclear weapon tests almost doubled the concentration of 14
C in the Northern Hemisphere.[32] The annotated PTBT label is representative of the Partial Nuclear Test Ban Treaty.
The above-ground
One side-effect of the change in atmospheric carbon-14 is that this has enabled some options (e.g., bomb-pulse dating[33]) for determining the birth year of an individual, in particular, the amount of carbon-14 in tooth enamel,[34][35] or the carbon-14 concentration in the lens of the eye.[36]
In 2019, Scientific American reported that carbon-14 from nuclear bomb testing has been found in the bodies of aquatic animals found in one of the most inaccessible regions of the earth, the Mariana Trench in the Pacific Ocean.[37]
The concentration of carbon-14 in atmospheric CO2, reported as the ratio of carbon-14 to carbon-12 with respect to a standard, has now (approximately since the year 2022) declined to levels similar to those prior to the above-ground nuclear tests of the 1950s and 1960s.[38][39] Although the extra carbon-14 atoms generated during those nuclear tests have not disappeared from the atmosphere, oceans and biosphere,[40] they are diluted because of the Suess effect.
Emissions from nuclear power plants
Carbon-14 is produced in coolant at boiling water reactors (BWRs) and pressurized water reactors (PWRs). It is typically released to the atmosphere in the form of carbon dioxide at BWRs, and methane at PWRs.[41] Best practice for nuclear power plant operator management of carbon-14 includes releasing it at night, when plants are not photosynthesizing.[42] Carbon-14 is also generated inside nuclear fuels (some due to transmutation of oxygen in the uranium oxide, but most significantly from transmutation of nitrogen-14 impurities), and if the spent fuel is sent to nuclear reprocessing then the carbon-14 is released, for example as CO2 during PUREX.[43][44]
Occurrence
Dispersion in the environment
After production in the upper atmosphere, the carbon-14 atoms react rapidly to form mostly (about 93%) 14
CO (carbon monoxide), which subsequently oxidizes at a slower rate to form 14
CO
2, radioactive carbon dioxide. The gas mixes rapidly and becomes evenly distributed throughout the atmosphere (the mixing timescale in the order of weeks). Carbon dioxide also dissolves in water and thus permeates the oceans, but at a slower rate.[21] The atmospheric half-life for removal of 14
CO
2 has been estimated to be roughly 12 to 16 years in the northern hemisphere. The transfer between the ocean shallow layer and the large reservoir of bicarbonates in the ocean depths occurs at a limited rate.[29]
In 2009 the activity of 14
C was 238 Bq per kg carbon of fresh terrestrial biomatter, close to the values before atmospheric nuclear testing (226 Bq/kg C; 1950).[45]
Total inventory
The inventory of carbon-14 in Earth's biosphere is about 300
The following inventory of carbon-14 has been given:[47]- Global inventory: ~8500 PBq (about 50 t)
- Atmosphere: 140 PBq (840 kg)
- Terrestrial materials: the balance
- From nuclear testing (until 1990): 220 PBq (1.3 t)
In fossil fuels
Many human-made chemicals are derived from fossil fuels (such as petroleum or coal) in which 14
C is greatly depleted because the age of fossils far exceeds the half-life of 14
C. The relative absence of 14
CO
2 is therefore used to determine the relative contribution (or mixing ratio) of fossil fuel oxidation to the total carbon dioxide in a given region of the Earth's atmosphere.[48]
Dating a specific sample of fossilized carbonaceous material is more complicated. Such deposits often contain trace amounts of carbon-14. These amounts can vary significantly between samples, ranging up to 1% of the ratio found in living organisms, a concentration comparable to an apparent age of 40,000 years.[49] This may indicate possible contamination by small amounts of bacteria, underground sources of radiation causing the 14
N(n,p)14
C reaction, direct uranium decay (although reported measured ratios of 14
C/U in uranium-bearing ores[50] would imply roughly 1 uranium atom for every two carbon atoms in order to cause the 14
C/12
C ratio, measured to be on the order of 10−15), or other unknown secondary sources of carbon-14 production. The presence of carbon-14 in the isotopic signature of a sample of carbonaceous material possibly indicates its contamination by biogenic sources or the decay of radioactive material in surrounding geologic strata. In connection with building the Borexino solar neutrino observatory, petroleum feedstock (for synthesizing the primary scintillant) was obtained with low 14
C content. In the Borexino Counting Test Facility, a 14
C/12
C ratio of 1.94×10−18 was determined;[51] probable reactions responsible for varied levels of 14
C in different petroleum reservoirs, and the lower 14
C levels in methane, have been discussed by Bonvicini et al.[52]
In the human body
Since many sources of human food are ultimately derived from terrestrial plants, the relative concentration of carbon-14 in human bodies is nearly identical to the relative concentration in the atmosphere. The rates of disintegration of
Carbon-14 can be used as a radioactive tracer in medicine. In the initial variant of the urea breath test, a diagnostic test for Helicobacter pylori, urea labeled with approximately 37 kBq (1.0 μCi) carbon-14 is fed to a patient (i.e., 37,000 decays per second). In the event of a H. pylori infection, the bacterial urease enzyme breaks down the urea into ammonia and radioactively-labeled carbon dioxide, which can be detected by low-level counting of the patient's breath.[55]
See also
References
- ^ .
- ^ a b Waptstra AH, Audi G, Thibault C. "AME atomic mass evaluation 2003". IAEA.org. Archived from the original on 5 May 2023.
- PMID 17737092.
- S2CID 27534222.
- PMID 35783167.
- ^ "What is carbon dating?". National Ocean Sciences Accelerator Mass Spectrometry Facility. Archived from the original on July 5, 2007. Retrieved 2007-06-11.
- ln(2))
- ^ Nicols AL. "14C Comments on evaluation of decay data" (PDF). www.nucleide.org. LNHB. Archived (PDF) from the original on 2011-08-15. Retrieved 30 October 2021.
- ^ "Appendix B: The Characteristics of Common Radioisotopes". Radiation Safety Manual for Laboratory Users. Princeton University. Archived from the original on 2013-10-02.
- ^ "Carbon-14". Material Safety Data Sheet. University of Michigan. Archived from the original on 2013-03-12.
- PMID 15407879.
- ^ "Carbon 14:age calculation". C14dating.com. Archived from the original on 2007-06-10. Retrieved 2007-06-11.
- ^ "Class notes for Isotope Hydrology EESC W 4886: Radiocarbon 14C". Martin Stute's homepage at Columbia. Archived from the original on 2006-09-24. Retrieved 2007-06-11.
- S2CID 205232299.
- ISSN 2169-8953.
- hdl:11585/770531.
- S2CID 230508539.
- S2CID 118602346.
- S2CID 119301845.
- ^ (PDF) from the original on 2015-12-22.
- ^ .
- S2CID 4368820. Archived from the original(PDF) on 2015-07-06.
- S2CID 55137950.
- PMID 26497389.
- PMID 28100493.
- .
- S2CID 4388159.
- doi:10.2172/7114972.
- ^ .
- ^ Manning MR, Melhuish WH (1994). "Atmospheric δ14C record from Wellington". Trends: A Compendium of Data on Global Change. Carbon Dioxide Information Analysis Center. Archived from the original on 2014-02-01. Retrieved 2007-06-11.
- ^ Levin I, Kromer B, Schoch-Fischer H, Bruns M, Münnich M, Berdau D, Vogel JW, Münnich KO (1994). "δ14C record from Vermunt". Trends: A Compendium of Data on Global Change. Carbon Dioxide Information Analysis Center. Archived from the original on 2008-09-23. Retrieved 2009-03-25.
- ^ "Radiocarbon dating". University of Utrecht. Archived from the original on 2007-12-09. Retrieved 2008-02-19.
- from the original on 2014-10-20.
- ^ "Radiation in Teeth Can Help Date, ID Bodies, Experts Say". National Geographic News. 2005-09-22. Archived from the original on 2007-04-25.
- S2CID 4407447.
- PMID 18231610.
- ^ Levy A (15 May 2019). "'Bomb Carbon' Has Been Found in Deep-Ocean Creatures". Scientific American.
- ^ Jones, Nicola (27 July 2022). "Carbon dating hampered by rising fossil-fuel emissions". Nature News. Retrieved 5 November 2023.
- PMID 35854150.
- .
- ^ "EPRI | Product Abstract | Impact of Nuclear Power Plant Operations on Carbon-14 Generation, Chemical Forms, and Release". www.epri.com. Archived from the original on 2016-08-18. Retrieved 2016-07-07.
- ^ "EPRI | Product Abstract | Carbon-14 Dose Calculation Methods at Nuclear Power Plants". www.epri.com. Archived from the original on 2016-08-18. Retrieved 2016-07-07.
- ^ Otlet RL, Fulker MJ, Walker AJ (1992). "Environmental Impact of Atmospheric Carbon-14 Emissions Resulting from the Nuclear Energy Cycle.". In Taylor RE, Long A, Kra RS (eds.). Radiocarbon After Four Decades. New York, NY: Springer.
- ^ "Carbon-14 and the environment". Institute for Radiological Protection and Nuclear Safety.
- ^ "Carbon-14 and the environment". Institute for Radiological Protection and Nuclear Safety. Archived from the original on 2015-04-18.
- ^ "Human Health Fact Sheet – Carbon 14" (PDF). Argonne National Laboratory, EVS. August 2005. Archived from the original (PDF) on 2011-07-16.
- ISBN 978-0-7506-7463-8.
- ^ "The Basics: 14C and Fossil Fuels". NOAA ESRL GMD Education and Outreach. Archived from the original on 25 September 2015. Retrieved 9 Dec 2015.
All other atmospheric carbon dioxide comes from young sources–namely land-use changes (for example, cutting down a forest in order to create a farm) and exchange with the ocean and terrestrial biosphere. This makes 14C an ideal tracer of carbon dioxide coming from the combustion of fossil fuels. Scientists can use 14C measurements to determine the age of carbon dioxide collected in air samples, and from this can calculate what proportion of the carbon dioxide in the sample comes from fossil fuels.
- from the original on 2013-07-24.
- Bibcode:1985Metic..20..676J.
- .
- arXiv:hep-ex/0308025.
- ^ Rowland RE. "The Radioactivity of the Normal Adult Body". rerowland.com. Archived from the original on 2011-02-05.
- ^ Ionizing Radiation Exposure of the Population of the United States | NCRP Report No. 93. National Council on Radiation Protection and Measurements. 1987. Archived from the original on 2007-07-11.)
- ^ "Society of Nuclear Medicine Procedure Guideline for C-14 Urea Breath Test" (PDF). snm.org. 2001-06-23. Archived from the original (PDF) on 2007-09-26. Retrieved 2007-07-04.
Further reading
- Kamen MD (1985). Radiant Science, Dark Politics: A Memoir of the Nuclear Age. Berkeley: University of California Press. ISBN 978-0-520-04929-1.
External links
- What is Carbon Dating?, Woods Hole Oceanographic Institute

