Ramberg–Bäcklund reaction
| Ramberg–Bäcklund reaction | |
|---|---|
| Named after | Birger Bäcklund
|
| Reaction type | Rearrangement reaction |
| Identifiers | |
| Organic Chemistry Portal | ramberg-baecklund-reaction |
| RSC ontology ID | RXNO:0000094 |
The Ramberg–Bäcklund reaction is an organic reaction converting an α-halo sulfone into an alkene in presence of a base with extrusion of sulfur dioxide.[1] The reaction is named after the two Swedish chemists Ludwig Ramberg and Birger Bäcklund. The carbanion formed by deprotonation gives an unstable episulfone that decomposes with elimination of sulfur dioxide. This elimination step is considered to be a concerted cheletropic extrusion.[citation needed]

The overall transformation is the conversion of the carbon–sulfur bonds to a carbon–carbon double bond. The original procedure involved halogenation of a
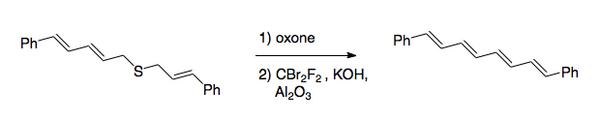
Applications
The Ramberg–Bäcklund reaction has several applications. Due to the nature of elimination, it can be applied to both small rings [3],
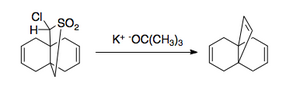
and large rings containing a double bond [4].
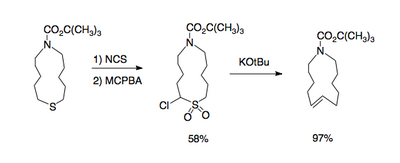
This reaction type gives access to 1,2-dimethylenecyclohexane[5]

and the epoxide variation [6] access to allyl alcohols.
Substrates
The necessary α-halo sulfones are accessible through oxidation of the corresponding α-halo sulfides with peracids such as
Mechanism
The
The Favorskii rearrangement and the Eschenmoser sulfide contraction are conceptually related reactions.
References
- ^ Ohme, R.; Preuschhof, H.; Heyne, H.-U. (1988). "Azoethane". Organic Syntheses; Collected Volumes, vol. 6, p. 78.
- ISSN 0365-3781.
- .
- .
- doi:10.1139/v00-103.
- .
- ISBN 9780471264187.
- ; Collected Volumes, vol. 8, p. 212.
- .
- .
