Colorectal cancer
| Colorectal cancer | |
|---|---|
| Other names | Colon cancer, rectal cancer, bowel cancer |
| Prevention | Screening from age of 45 to 75 |
| Treatment | Surgery, radiation therapy, chemotherapy, targeted therapy[5] |
| Prognosis | Five-year survival rate 65% (US)[6] |
| Frequency | 9.4 million (2015)[7] |
| Deaths | 551,000 (2018)[8] |
Colorectal cancer (CRC), also known as bowel cancer, colon cancer, or rectal cancer, is the development of
Colorectal cancer may be diagnosed by
Treatments used for colorectal cancer may include some combination of surgery, radiation therapy, chemotherapy, and targeted therapy.[5] Cancers that are confined within the wall of the colon may be curable with surgery, while cancer that has spread widely is usually not curable, with management being directed towards improving quality of life and symptoms.[5] The five-year survival rate in the United States was around 65% in 2014.[6] The individual likelihood of survival depends on how advanced the cancer is, whether or not all the cancer can be removed with surgery, and the person's overall health.[1] Globally, colorectal cancer is the third most common type of cancer, making up about 10% of all cases.[14] In 2018, there were 1.09 million new cases and 551,000 deaths from the disease.[8] It is more common in developed countries, where more than 65% of cases are found.[2] It is less common in women than men.[2]
Signs and symptoms
The signs and symptoms of colorectal cancer depend on the location of the
Rectal bleeding or anemia are high-risk symptoms in people over the age of 50.[17] Weight loss and changes in a person's bowel habit are typically only concerning if they are associated with rectal bleeding.[17][18]
Cause
75–95% of colorectal cancer cases occur in people with little or no genetic risk.
Pathogenic Escherichia coli may increase the risk of colorectal cancer by producing the genotoxic metabolite, colibactin.[30]
Inflammatory bowel disease
People with
Genetics
Those with a family history in two or more
Mutations in the pair of genes (POLE and POLD1) have been associated with familial colon cancer.[40]
Most deaths due to colon cancer are associated with metastatic disease. A gene that appears to contribute to the potential for metastatic disease, metastasis associated in colon cancer 1 (MACC1), has been isolated.
Pathogenesis
Colorectal cancer is a disease originating from the
Beyond the defects in the
Other proteins responsible for programmed cell death that are commonly deactivated in colorectal cancers are
Approximately 70% of all
Comprehensive,
Mismatch repair (MMR) deficient tumours are characterized by a relatively high amount of poly-nucleotide tandem repeats.[54] This is caused by a deficiency in MMR proteins – which are typically caused by epigenetic silencing and or inherited mutations (e.g., Lynch syndrome).[55] 15 to 18 percent of colorectal cancer tumours have MMR deficiencies, with 3 percent developing due to Lynch syndrome.[56] The role of the mismatch repair system is to protect the integrity of the genetic material within cells (i.e., error detecting and correcting).[55] Consequently, a deficiency in MMR proteins may lead to an inability to detect and repair genetic damage, allowing for further cancer-causing mutations to occur and colorectal cancer to progress.[55]
The polyp to cancer progression sequence is the classical model of colorectal cancer
Field defects
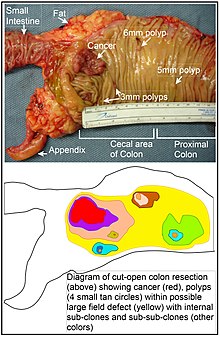
The term "field cancerization" was first used in 1953 to describe an area or "field" of epithelium that has been preconditioned (by what were largely unknown processes at the time) to predispose it towards development of cancer.
Field defects are important in progression to colon cancer.[62][63]
However, as pointed out by Rubin, "The vast majority of studies in cancer research has been done on well-defined tumors in vivo, or on discrete neoplastic foci in vitro. Yet there is evidence that more than 80% of the somatic mutations found in mutator phenotype human colorectal tumors occur before the onset of terminal clonal expansion."[64][65] Similarly, Vogelstein et al.[66] pointed out that more than half of somatic mutations identified in tumors occurred in a pre-neoplastic phase (in a field defect), during growth of apparently normal cells. Likewise, epigenetic alterations present in tumors may have occurred in pre-neoplastic field defects.[67]
An expanded view of field effect has been termed "etiologic field effect", which encompasses not only molecular and pathologic changes in pre-neoplastic cells but also influences of exogenous environmental factors and molecular changes in the local microenvironment on neoplastic evolution from tumor initiation to death.[68]
Epigenetics
Epigenetic alterations are much more frequent in colon cancer than genetic (mutational) alterations. As described by Vogelstein et al.,[66] an average cancer of the colon has only 1 or 2 oncogene mutations and 1 to 5 tumor suppressor mutations (together designated "driver mutations"), with about 60 further "passenger" mutations. The oncogenes and tumor suppressor genes are well studied and are described above under Pathogenesis.[69][70]
In addition to epigenetic alteration of expression of miRNAs, other common types of epigenetic alterations in cancers that change gene expression levels include direct hypermethylation or hypomethylation of CpG islands of protein-encoding genes and alterations in histones and chromosomal architecture that influence gene expression.[71] As an example, 147 hypermethylations and 27 hypomethylations of protein coding genes were frequently associated with colorectal cancers. Of the hypermethylated genes, 10 were hypermethylated in 100% of colon cancers, and many others were hypermethylated in more than 50% of colon cancers.[72] In addition, 11 hypermethylations and 96 hypomethylations of miRNAs were also associated with colorectal cancers.[72] Abnormal (aberrant) methylation occurs as a normal consequence of normal aging and the risk of colorectal cancer increases as a person gets older.[73] The source and trigger of this age-related methylation is unknown.[73][74] Approximately half of the genes that show age-related methylation changes are the same genes that have been identified to be involved in the development of colorectal cancer.[73] These findings may suggest a reason for age being associated with the increased risk of developing colorectal cancer.[73]
Epigenetic reductions of DNA repair enzyme expression may likely lead to the genomic and epigenomic instability characteristic of cancer.[75][76][67] As summarized in the articles Carcinogenesis and Neoplasm, for sporadic cancers in general, a deficiency in DNA repair is occasionally due to a mutation in a DNA repair gene, but is much more frequently due to epigenetic alterations that reduce or silence expression of DNA repair genes.[77]
Epigenetic alterations involved in the development of colorectal cancer may affect a person's response to chemotherapy.[78]
Genomics and epigenomics
Consensus molecular subtypes (CMS) classification of colorectal cancer was first introduced in 2015. CMS classification so far has been considered the most robust classification system available for CRC that has a clear biological interpretability and the basis for future clinical stratification and subtype-based targeted interventions.[79]
A novel Epigenome-based Classification (EpiC) of colorectal cancer was proposed in 2021 introducing 4 enhancer subtypes in people with CRC. Chromatin states using 6 histone marks are characterized to identify EpiC subtypes. A combinatorial therapeutic approach based on the previously introduced consensus molecular subtypes (CMSs) and EpiCs could significantly enhance current treatment strategies.[80]
Diagnosis
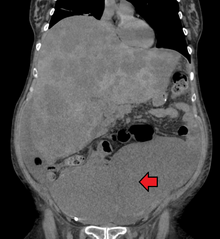
Colorectal cancer diagnosis is performed by sampling of areas of the colon suspicious for possible tumor development, typically during colonoscopy or sigmoidoscopy, depending on the location of the lesion.[20] It is confirmed by microscopical examination of a tissue sample.[citation needed]
Medical imaging
A colorectal cancer is sometimes initially discovered on CT scan.[81]
Presence of
MRI is also performed after completion of neoadjuvant chemoradiotherapy to identify patients who achieve complete response. Patients with complete response on both MRI and endoscopy may not require surgical resection and can avoid unnecessary surgical morbidity and complications.[82] Patients selected for non-surgical treatment of rectal cancer should have periodic MRI scans, receive physical examinations, and undergo endoscopy procedures to detect any tumor re-growth which can occur in a minority of these patients. When local recurrence occurs, periodic follow up can detect it when it is still small and curable with salvage surgery. In addition, MRI tumor regression grades can be assigned after chemoradiotherapy which correlate with patients' long-term survival outcomes.[83]
Histopathology


The
Staging
Staging of the cancer is based on both radiological and pathological findings. As with most other forms of cancer, tumor staging is based on the TNM system which considers how much the initial tumor has spread and the presence of metastases in lymph nodes and more distant organs.[20] The AJCC 8th edition was published in 2018.[89]
Prevention
It has been estimated that about half of colorectal cancer cases are due to lifestyle factors, and about a quarter of all cases are preventable.[90] Increasing surveillance, engaging in physical activity, consuming a diet high in fiber, and reducing smoking and alcohol consumption decrease the risk.[91][92]
Lifestyle
Lifestyle risk factors with strong evidence include lack of exercise, cigarette smoking, alcohol, and obesity.[93][94][95] The risk of colon cancer can be reduced by maintaining a normal body weight through a combination of sufficient exercise and eating a healthy diet.[96]
Current research consistently links eating more red meat and processed meat to a higher risk of the disease.[97] Starting in the 1970s, dietary recommendations to prevent colorectal cancer often included increasing the consumption of whole grains, fruits and vegetables, and reducing the intake of red meat and processed meats. This was based on animal studies and retrospective observational studies. However, large scale prospective studies have failed to demonstrate a significant protective effect, and due to the multiple causes of cancer and the complexity of studying correlations between diet and health, it is uncertain whether any specific dietary interventions will have significant protective effects.[98]: 432–433 [99]: 125–126 In 2018 the National Cancer Institute stated that "There is no reliable evidence that a diet started in adulthood that is low in fat and meat and high in fiber, fruits, and vegetables reduces the risk of CRC by a clinically important degree."[93][100]
According to the
The 2014 World Health Organization cancer report noted that it has been hypothesized that dietary fiber might help prevent colorectal cancer, but that most studies at the time had not yet studied the correlation.
Higher physical activity is recommended.
Medication and supplements
Aspirin and celecoxib appear to decrease the risk of colorectal cancer in those at high risk.[110][111] Aspirin is recommended in those who are 50 to 60 years old, do not have an increased risk of bleeding, and are at risk for cardiovascular disease to prevent colorectal cancer.[112] It is not recommended in those at average risk.[113]
There is tentative evidence for calcium supplementation, but it is not sufficient to make a recommendation.[114] Vitamin D intake and blood levels are associated with a lower risk of colon cancer.[115][116]
Screening
As more than 80% of colorectal cancers arise from
The three main screening tests are colonoscopy,
Fecal occult blood testing (FOBT) of the stool is typically recommended every two years and can be either guaiac-based or immunochemical.[20] If abnormal FOBT results are found, participants are typically referred for a follow-up colonoscopy examination. When done once every 1–2 years, FOBT screening reduces colorectal cancer deaths by 16% and among those participating in screening, colorectal cancer deaths can be reduced up to 23%, although it has not been proven to reduce all-cause mortality.[121] Immunochemical tests are accurate and do not require dietary or medication changes before testing.[122] However, research in the UK has found that for these immunochemical tests, the threshold for further investigation is set at a point that may miss more than half of bowel cancer cases. The research suggests that the NHS England's Bowel Cancer Screening Programme could make better use of the test's ability to provide the exact concentration of blood in faeces (rather than only whether it is above or below a cutoff level).[123][124]
Other options include virtual colonoscopy and stool DNA screening testing (FIT-DNA). Virtual colonoscopy via a CT scan appears as good as standard colonoscopy for detecting cancers and large adenomas but is expensive, associated with radiation exposure, and cannot remove any detected abnormal growths as standard colonoscopy can.[20] Stool DNA screening test looks for biomarkers associated with colorectal cancer and precancerous lesions, including altered DNA and blood hemoglobin. A positive result should be followed by colonoscopy. FIT-DNA has more false positives than FIT and thus results in more adverse effects.[10] Further study is required as of 2016 to determine whether a three-year screening interval is correct.[10]
Recommendations
In the United States, screening is typically recommended between ages 50 and 75 years.[10][125] The American Cancer Society recommends starting at the age of 45.[126] For those between 76 and 85 years old, the decision to screen should be individualized.[10] For those at high risk, screenings usually begin at around 40.[20][127]
Several screening methods are recommended including stool-based tests every 2 years,
In Canada, among those 50 to 75 years old at normal risk, fecal immunochemical testing or FOBT is recommended every two years or sigmoidoscopy every 10 years.[133] Colonoscopy is less preferred.[133]
Some countries have national colorectal screening programs which offer FOBT screening for all adults within a certain age group, typically starting between ages 50 and 60. Examples of countries with organised screening include the United Kingdom,[134] Australia,[135] the Netherlands,[136] Hong Kong, and Taiwan.[137]
The UK Bowel Cancer Screening Programme aims to find warning signs in people aged 60 to 74, by recommending a faecal immunochemical test (FIT) every two years. FIT measures blood in faeces, and people with levels above a certain threshold may have bowel tissue examined for signs of cancer. Growths having cancerous potential are removed.[138][124]
Treatment
The treatment of colorectal cancer can be aimed at cure or palliation. The decision on which aim to adopt depends on various factors, including the person's health and preferences, as well as the stage of the tumor.[139] Assessment in multidisciplinary teams is a critical part of determining whether the patient is suitable for surgery or not.[140] When colorectal cancer is caught early, surgery can be curative. However, when it is detected at later stages (for which metastases are present), this is less likely and treatment is often directed at palliation, to relieve symptoms caused by the tumour and keep the person as comfortable as possible.[20]
Surgery


At an early stage, colorectal cancer may be removed during a colonoscopy using one of several techniques, including
If there are only a few metastases in the liver or lungs, these may also be removed.
Chemotherapy
In both cancer of the
In Stage I colon cancer, no chemotherapy is offered, and surgery is the definitive treatment. The role of chemotherapy in Stage II colon cancer is debatable, and is usually not offered unless risk factors such as T4 tumor, undifferentiated tumor, vascular and perineural invasion or inadequate lymph node sampling is identified.[144] It is also known that the people who carry abnormalities of the mismatch repair genes do not benefit from chemotherapy. For Stage III and Stage IV colon cancer, chemotherapy is an integral part of treatment.[20]
If cancer has spread to the
The primary difference in the approach to low stage rectal cancer is the incorporation of radiation therapy. Often, it is used in conjunction with chemotherapy in a
Stage IV colorectal cancer due to peritoneal carcinomatosis can be treated using HIPEC combined with cytoreductive surgery, in some people.[149][150][151] Also, T4 colorectal cancer can be treated with HIPEC to avoid future relapses.[152]
Radiation therapy
While a combination of
Immunotherapy
On the other hand, in a prospective phase 2 study published in June 2022 in The New England Journal of Medicine, 12 patients with Deficient Mismatch Repair (dMMR) stage II or III rectal adenocarcinoma were administered single-agent dostarlimab, an anti–PD-1 monoclonal antibody, every three weeks for six months. After a median follow-up of 12 months (range, 6 to 25 months), all 12 patients had a complete clinical response with no evidence of tumor on MRI, 18F-fluorodeoxyglucose–positron-emission tomography, endoscopic evaluation, digital rectal examination, or biopsy. Moreover, no patient in the trial needed chemoradiotherapy or surgery, and no patient reported adverse events of grade 3 or higher. However, although the results of this study are promising, the study is small and has uncertainties about long-term outcomes.[161]
Palliative care
Palliative care is recommended for any person who has advanced colon cancer or who has significant symptoms.[162][163]
Involvement of palliative care may be beneficial to improve the quality of life for both the person and his or her family, by improving symptoms, anxiety and preventing admissions to the hospital.[164]
In people with incurable colorectal cancer, palliative care can consist of procedures that relieve symptoms or complications from the cancer but do not attempt to cure the underlying cancer, thereby improving quality of life. Surgical options may include non-curative surgical removal of some of the cancer tissue, bypassing part of the intestines, or stent placement. These procedures can be considered to improve symptoms and reduce complications such as bleeding from the tumor, abdominal pain and intestinal obstruction.[165] Non-operative methods of symptomatic treatment include radiation therapy to decrease tumor size as well as pain medications.[166]
Psychosocial Intervention
In addition to medical intervention, a variety of psychosocial interventions have been implemented in the treatment of colorectal cancer.[167] Depression and anxiety are highly prevalent in patients diagnosed with CRC, therefore psychosocial interventions are necessary.[168][169] Many patients continue to experience symptoms of anxiety and depression following treatment, regardless of treatment outcome.[168][170] Societal stigmas associated with colorectal cancer present further psychosocial challenges for CRC patients and their families.[171][172]
Depression and Anxiety
Colorectal cancer patients have a 51% higher risk of experiencing depression than individuals without the disease.[173] Additionally, CRC patients are at high risk of experiencing severe anxiety, low self-esteem, poor self-concept, and social anxiety.[174][175]
Post-Treatment Distress
Regardless of treatment outcome, many CRC patients experience ongoing symptoms of anxiety, depression, and distress.[176]
Survivorship of CRC can involve significant lifestyle adjustments.[177] Postoperative afflictions may include stomas, bowel issues, incontinence, odor, and changes to sexual functioning.[177][178] These changes can result in distorted body image, social anxiety, depression, and distress—all of which contribute to a poorer quality of life.[177][179]
Colorectal cancer is the second leading cause of cancer-related death worldwide.[180] Transitioning into palliative care and contending with mortality can be a deeply distressing experience for a CRC patient and their loved ones.
Stigma
Colorectal cancer is highly stigmatized and can elicit feelings of disgust from patients, healthcare professionals, family, intimate partners, and the general public.[181] Patients with stomas are especially vulnerable to stigmatization due to unavoidable odors, gas, and unpleasant noises from stoma bags.[181] Additionally, associated CRC risk factors like poor diet, alcohol consumption, and lack of physical activity prompt negative assumptions of blame and personal responsibility onto CRC patients.[182] Judgement from others along with internalized self-blame and embarrassment can negatively affect self-esteem, sociability, and quality of life.[182]
Methods of Intervention
Face-to-face interventions such as clinician-patient talk therapy, body-mind-spirit practices, and support group sessions have been identified as most effective in reducing anxiety and depression in CRC patients.[183] Additionally, journaling exercises and over-the-phone talk therapy sessions have been implemented.[183] Though deemed less effective, these non-face-to-face interventions are economically inclusive and have been found to reduce both depression and anxiety in CRC patients.[183]
Follow-up
The U.S.
Routine
For people who have undergone curative surgery or adjuvant therapy (or both) to treat non-metastatic colorectal cancer, intense surveillance and close follow-up have not been shown to provide additional survival benefits.[186]
Exercise
Exercise may be recommended in the future as secondary therapy to cancer survivors. In epidemiological studies, exercise may decrease colorectal cancer-specific mortality and all-cause mortality. Results for the specific amounts of exercise needed to observe a benefit were conflicting. These differences may reflect differences in tumour biology and the expression of biomarkers. People with tumors that lacked
Another potential biomarker may be
Physical activity provides benefits to people with non-advanced colorectal cancer. Improvements in aerobic fitness, cancer-related fatigue and health-related quality of life have been reported in the short term.[189] However, these improvements were not observed at the level of disease-related mental health, such as anxiety and depression.[189]
Prognosis
Fewer than 600 genes are linked to outcomes in colorectal cancer.[51] These include both unfavorable genes, where high expression is related to poor outcome, for example the heat shock 70 kDa protein 1 (HSPA1A), and favorable genes where high expression is associated with better survival, for example the putative RNA-binding protein 3 (RBM3).[51] The prognosis is also correlated with a poor fidelity of the pre-mRNA splicing apparatus, and thus a high number of deviating alternative splicing.[190]
Recurrence rates
The average five-year recurrence rate in people where surgery is successful is 5% for stage I cancers, 12% in stage II and 33% in stage III. However, depending on the number of risk factors it ranges from 9–22% in stage II and 17–44% in stage III.[191]
Survival rates
In Europe the
Survival is directly related to detection and the type of cancer involved, but overall is poor for symptomatic cancers, as they are typically quite advanced. Survival rates for early stage detection are about five times that of late stage cancers. People with a tumor that has not breached the
Whilst the impact of colorectal cancer on those who survive varies greatly there will often be a need to adapt to both physical and psychological outcomes of the illness and its treatment.[197] For example, it is common for people to experience incontinence,[198] sexual dysfunction,[199] problems with stoma care[200] and fear of cancer recurrence[201] after primary treatment has concluded.
A qualitative systematic review published in 2021 highlighted that there are three main factors influencing adaptation to living with and beyond colorectal cancer: support mechanisms, severity of late effects of treatment and psychosocial adjustment. Therefore, it is essential that people are offered appropriate support to help them better adapt to life following treatment.[202]
Epidemiology
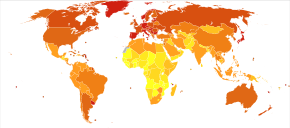
Globally more than 1 million people get colorectal cancer every year[20] resulting in about 715,000 deaths as of 2010 up from 490,000 in 1990.[203]
As of 2012[update], it is the second most common cause of cancer in women (9.2% of diagnoses) and the third most common in men (10.0%)[14]: 16 with it being the fourth most common cause of cancer death after lung, stomach, and liver cancer.[204] It is more common in developed than developing countries.[205] Global incidence varies 10-fold, with highest rates in Australia, New Zealand, Europe and the US and lowest rates in Africa and South-Central Asia.[206]
United States
In 2022, the incidence of colorectal cancer in the United States was anticipated to be about 151,000 adults, including over 106,000 new cases of colon cancer (some 54,000 men and 52,000 women) and about 45,000 new cases of rectal cancer.[207] Since the 1980s, the incidence of colorectal cancer decreased, dropping by about 2% annually from 2014 to 2018 in adults aged 50 and older, due mainly to improved screening.[207] However, incidence of colorectal cancer has increased in individuals aged 25 to 50. In early 2023, the American Cancer Society (ACS) reported that 20% of diagnoses (of colon cancer) in 2019 were in patients under age 55, which is about double the rate in 1995, and rates of advanced disease increased by about 3% annually in people younger than 50. It predicted that, in 2023, an estimated 19,550 diagnoses and 3,750 deaths would be in people younger than 50.[208] Colorectal cancer also disproportionately affects the Black community, where the rates are the highest of any racial/ethnic group in the US. African Americans are about 20% more likely to get colorectal cancer and about 40% more likely to die from it than most other groups. Black Americans often experience greater obstacles to cancer prevention, detection, treatment, and survival, including systemic racial disparities that are complex and go beyond the obvious connection to cancer.
United Kingdom
In the UK about 41,000 people a year get colon cancer making it the fourth most common type.[209]
Australia
One in 19 men and one in 28 women in Australia will develop colorectal cancer before the age of 75; one in 10 men and one in 15 women will develop it by 85 years of age.[210]
Papua New Guinea
In the developing countries like Papua New Guinea and other Pacific Island States including the Solomon Islands, colorectal cancer is a very rare cancer amongst the people, which is least common compared to lung, stomach, liver or breast cancer. It is estimated that at least 8 in 100,000 of the people are most likely to developed colorectal cancer every year, which is unlike lung or breast cancer, where for the latter alone is 24 in 100,000 women.[211]
Early Onset Colorectal Cancer (EOCC)
A diagnosis of colorectal cancer in patients under 50 years of age is referred to as Early Onset Colorectal Cancer (EOCC).[212][213]Instances of EOCC have increased over the last decade, specifically in patient populations aged 20-40 years old throughout North America, Europe, Australia, and China.[213][214]
Incidence by Age
Incidences of colorectal cancer in younger populations have increased over the last decade.[215][216][217]While advancements in diagnostic procedure may have some impact, reduced likelihood of screening among these populations suggests detection bias is not a major contributor to this trend. More likely, cohort-effects are at play.[217]
The population experiencing the greatest rise in EOCC cases are men and women aged 20-29 years old, with incidences increasing by 7.9% a year between 2004 and 2016.[217] Similarly, though less severe, men and women aged 30-39 experienced an increase in cases at a rate of 3.4% a year during that same time period. Despite these increases, the mortality rate for colorectal cancer has remained the same.[217]
Risk Factors
Risk factors associated with EOCC are akin to those of all colorectal cancer cases.[218] Observed cohort-effects are likely the product of generational shifts in lifestyle and environmental factors.[219][218]
Preventative Screening
In 2018, the American Cancer Society modified their previous screening guideline for colorectal cancer from age 50 down to age 45 following the recognition of increasing cases of EOCC.[220] Individuals under the age of 60 have been identified as most susceptible to non-participation in colorectal cancer screening.[221]
History
This section needs expansion. You can help by adding to it. (January 2024) |
Rectal cancer has been diagnosed in an Ancient Egyptian
Society and culture
In the United States, March is colorectal cancer awareness month.[118]
Research
This section needs to be updated. The reason given is: all sources > 5 years. (January 2024) |
Preliminary
- The Cancer Genome Atlas[53]
- The Colorectal Cancer Atlas integrating genomic and proteomic data pertaining to colorectal cancer tissues and cell lines have been developed.[227]
See also
- Fecal occult blood
- Adenoma-carcinoma sequence
References
- ^ a b c d "General Information About Colon Cancer". NCI. May 12, 2014. Archived from the original on July 4, 2014. Retrieved June 29, 2014.
- ^ ISBN 978-92-832-0443-5.
- ^ a b c "Colorectal Cancer Prevention (PDQ®)". National Cancer Institute. February 27, 2014. Archived from the original on July 5, 2014. Retrieved June 29, 2014.
- ^ PMID 28826375.
- ^ a b c d e f g "Colon Cancer Treatment (PDQ®)". NCI. May 12, 2014. Archived from the original on July 5, 2014. Retrieved June 29, 2014.
- ^ a b "SEER Stat Fact Sheets: Colon and Rectum Cancer". NCI. Archived from the original on June 24, 2014. Retrieved June 18, 2014.
- PMID 27733282.
- ^ S2CID 52188256.
- ^ "Colorectal Cancer Signs and Symptoms | Signs of Colorectal Cancer". www.cancer.org. Retrieved February 8, 2023.
- ^ PMID 27304597.
- ^ "First Colonoscopies Now Recommended at Age 45". ThedaCare. Retrieved December 30, 2022.
- S2CID 40187047.
- from the original on July 14, 2014.
- ^ ISBN 978-92-832-0443-5.
- ISBN 978-1-4051-6910-3. Archivedfrom the original on September 28, 2015.
- PMID 30065255.
- ^ PMID 21619747.
- PMID 21624112.
- ^ S2CID 7640363.
- ^ S2CID 25299272.
- ^ a b "Colorectal Cancer 2011 Report: Food, Nutrition, Physical Activity, and the Prevention of Colorectal Cancer" (PDF). World Cancer Research Fund & American Institute for Cancer Research. 2011. Archived (PDF) from the original on September 9, 2016.
- S2CID 221359557.
- PMID 23575393.
- PMID 22818936.
- PMID 21307158.
- S2CID 2256436.
- PMID 21960713.
- PMID 25233845.
- ^ license.
- S2CID 216033220.
- PMID 21822822.
- PMID 26004415.
- ^ PMID 19596953.
- ^ PMID 28922695.
- S2CID 36836169.
- PMID 19822006.
- S2CID 21385340.
- PMID 31520728.
- PMID 29445907.
- PMID 28427513.
- S2CID 8854895.
- ^ Stein U (2013) MACC1 – a novel target for solid cancers. Expert Opin Ther Targets
- PMID 17892325.
- ^ "What is the relationship between Ashkenazi Jews and colorectal cancer?". WebMD. Retrieved October 17, 2019.
- ^ PMID 33127962.
- S2CID 4254940.
- ^ Chakravarthi S, Krishnan B, Madhavan M (1999). "Apoptosis and expression of p53 in colorectal neoplasms". Indian J. Med. Res. 86 (7): 95–102.
- PMID 21472043.
- ^ PMID 20018966.
- PMID 15310786.
- ^ PMID 28818916.
- S2CID 205383514.
- ^ PMID 22810696.
- PMID 26875156.
- ^ PMID 28693799.
- PMID 29955148.
- ^ PMID 25492499.
- S2CID 36456541.
- PMID 31622622.
- S2CID 6736946.
- PMID 16174847.
- PMID 18164807.
- PMID 20689513. 28 minute video
- S2CID 44981539.
- PMID 10655514.
- ^ PMID 23539594.
- ^ PMID 23671730.
- PMID 24925058.
- ^ Wilbur B, ed. (2009). The World of the Cell (7th ed.). San Francisco, C.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ Kimball's Biology Pages. Archived December 31, 2017, at the Wayback Machine "Oncogenes" Free full text
- PMID 22082348.
- ^ PMID 22389639.
- ^ PMID 22009203.
- PMID 27256564.
- PMID 17412712.
- PMID 21278452.
- ^ "Colorectal Cancer". The Lecturio Medical Concept Library. Retrieved July 22, 2021.
- PMID 24574767.
- PMID 26457759.
- S2CID 235269540.
- ^ a b "Colorectal Cancer". The Lecturio Medical Concept Library. Retrieved July 10, 2021.
- S2CID 257583845.
- S2CID 257583845.
- S2CID 34693873.
- ^ "Colon, Rectosigmoid, and Rectum Equivalent Terms and Definitions C180-C189, C199, C209, (Excludes lymphoma and leukemia M9590 – M9992 and Kaposi sarcoma M9140) – Colon Solid Tumor Rules 2018. July 2019 Update" (PDF). National Cancer Institute. Archived (PDF) from the original on January 16, 2020.
- ^ "Colorectal cancer types". Cancer Treatment Centers of America. October 4, 2018. Retrieved January 16, 2020.
- .
- PMID 9767106.
- ^ "TNM staging of colorectal carcinoma (AJCC 8th edition)". www.pathologyoutlines.com. Retrieved February 24, 2019.
- PMID 22158327.
- ISBN 978-0-19-974797-9. Archivedfrom the original on September 28, 2015.
- ISBN 978-3-540-37696-5. Archivedfrom the original on October 3, 2015.
- ^ a b "Colorectal Cancer Prevention Overview". National Cancer Institute. March 1, 2018. Retrieved October 26, 2018.
- ^ "Cancer prevention". World Health Organization. Retrieved October 27, 2018.
- PMID 36361914.
- PMID 27557308.
- ^ "Colorectal Cancer – Risk Factors and Prevention". June 25, 2012.
- ISBN 978-92-832-0443-5.
- ^ ISBN 978-92-832-0443-5.
Several large prospective cohort studies of dietary fibre and colon cancer risk have not supported an association, although an inverse relation was seen in the large European Prospective Investigation into Cancer and Nutrition (EPIC) study and a recent meta-analysis. The variation in findings from prospective studies needs to be better understood; dietary fibre is complex and heterogeneous, and the relation with colorectal cancer could differ by dietary source. (p. 127)
- ^ "Colorectal Cancer Prevention Description of Evidence". National Cancer Institute. March 1, 2018. Retrieved October 26, 2018.
- ^ "Colorectal cancer". World Cancer Research Fund International. Archived from the original on July 15, 2023. Retrieved December 20, 2023.
- S2CID 58632705.
- PMID 30031175.
Despite the longstanding hypothesis that a high-fiber diet may protect against colorectal cancer... epidemiologic studies associating dietary fiber intake with subsequent risk of colorectal cancer have yielded inconsistent results... Nonetheless, based on existing evidence, the most recent expert report from the World Cancer Research Fund and American Institute for Cancer Research in 2017 concludes that there is probable evidence
- S2CID 251280745.
- PMID 22196090.
- S2CID 8026021.
- S2CID 24764995.
- PMID 27510511.
- S2CID 7256176.
- PMID 20594533.
- PMID 27859394.
- PMID 27064677.
- ^ Agency for Healthcare Research and Quality. "Aspirin or Nonsteroidal Anti-inflammatory Drugs for the Primary Prevention of Colorectal Cancer". United States Department of Health & Human Services. Archived from the original on January 5, 2016.
2010/2011
- PMID 18254022.
- PMID 21876081.
- PMID 21672549.
- ^ "What Can I Do to Reduce My Risk of Colorectal Cancer?". Centers for Disease Control and Prevention. April 2, 2014. Archived from the original on February 26, 2015. Retrieved March 5, 2015.
- ^ PMID 21954677.
- PMID 22219318.
- PMID 28828493.
- S2CID 26338156.
- PMID 24658694.
- S2CID 239113610. Retrieved August 5, 2022.
- ^ PMID 33342370.
- ^ PMID 31683290.
- PMID 29846947.
- U.S. Preventive Services Task Force. 2008. Archived from the originalon February 7, 2015. Retrieved December 19, 2011.
- ^ PMID 24922745.
- ^ "Five Things Physicians and Patients Should Question" (PDF). Choosing Wisely: An Initiative of the ABIM Foundation. American Gastroenterological Association. Archived from the original (PDF) on August 9, 2012. Retrieved August 17, 2012.
- S2CID 29354772.
- PMID 22393133.
- PMID 25881903.
- ^ PMID 26903355.
- ^ "NHS Bowel Cancer Screening Programme". cancerscreening.nhs.uk. Archived from the original on November 29, 2014.
- ^ "Home – Bowel Cancer Australia". bowelcanceraustralia.org. Archived from the original on December 24, 2014.
- ^ "Bevolkingsonderzoek darmkanker". rivm.nl. Archived from the original on December 17, 2014.
- PMID 32903904.
- S2CID 239113610.
- PMID 21943995.
- PMID 32150483.
- S2CID 57427824.
- ^ PMID 23235660.
- ^ a b "Colorectal (Colon) Cancer". Cleveland Clinic. Retrieved July 9, 2021.
- PMID 25430983.
- PMID 21180382.
- PMID 25918280.
- PMID 23997942.
- PMID 31633039.
- PMID 26941982.
- PMID 32686435.
- S2CID 21282326.
- PMID 37099280.
- ISBN 978-0-7817-7207-5.
- ^ PMID 31543678.
- PMID 27489505.
- PMID 32030236.
- PMID 28492495.
- ^ PMID 29208439.
- PMID 37968259.
- ^ "FDA grants accelerated approval to pembrolizumab for first tissue/site agnostic indication". U.S. Food and Drug Administration. February 9, 2019.
- S2CID 249395846.
- ^ "Palliative or Supportive Care". American Cancer Society. Archived from the original on August 21, 2014. Retrieved August 20, 2014.
- ^ "ASCO Provisional Clinical Opinion: The Integration of Palliative Care into Standard Oncology Care". ASCO. Archived from the original on August 21, 2014. Retrieved 20 August 2014.
- S2CID 39881122.
- PMID 17913495.
- PMID 15236729.
- PMID 29884182.
- ^ PMID 30709020.
- PMID 36421342.
- PMID 26315692.
- PMID 23047793.
- PMID 21954081.
- PMID 36421342.
- PMID 30709020.
- PMID 23759360.
- PMID 30709020.
- ^ PMID 21954081.
- PMID 23759360.
- PMID 33786669.
- PMID 37213277.
- ^ PMID 23047793.
- ^ PMID 21954081.
- ^ PMID 29884182.
- ^ a b "National Comprehensive Cancer Network" (PDF). nccn.org. Archived (PDF) from the original on March 25, 2009.
- ^ PMID 16260687.
- PMID 31483854.
- PMID 22610066.
- ^ PMID 22570317.
- ^ PMID 32361988.
- PMID 36821799.
- S2CID 51934598.
- PMID 20332485.
- ISBN 978-0-7817-7153-5.
- PMID 36945303.
- ^ "Five-Year Survival Rates". National Cancer Institute. Retrieved May 9, 2021.
- S2CID 149443256.
- PMID 26521054.
- S2CID 29087556.
- S2CID 42069306.
- PMID 20183104.
- PMID 17823231.
- S2CID 231771176.
- S2CID 1541253.
- ^ WHO (February 2010). "Cancer". World Health Organization. Archived from the original on December 29, 2010. Retrieved January 5, 2011.
- PMID 20952724.
- ^ Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010). "Colorectal Cancer Incidence, Mortality and Prevalence Worldwide in 2008 – Summary". Archived from the original on October 17, 2012.; "GLOBOCAN 2008 v2.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 10". Lyon, France: International Agency for Research on Cancer. Archived from the original on May 8, 2011.
- ^ a b "Colorectal cancer: Statistics". Cancer.net, American Society of Clinical Oncology. February 2022. Retrieved May 13, 2022.
- ^ Katella K. "Colorectal Cancer: What Millennials and Gen Zers Need to Know". YaleMedicine.
- ^ "Bowel cancer | About bowel cancer | Cancer Research UK". www.cancerresearchuk.org. Archived from the original on March 9, 2017. Retrieved May 12, 2017.
- ISBN 978-1-74249-677-1.
- ^ Cancer in Papua New Guinea: an Overview, 2016. Cancer series No. 176. Cat. No. CAN 88. Papua New Guinea Department of Health. 2016.
- PMID 37213277.
- ^ PMID 34885046.
- PMID 31097539.
- PMID 37213277.
- PMID 34885046.
- ^ PMID 31097539.
- ^ PMID 34885046.
- PMID 37213277.
- PMID 31097539.
- PMID 33313657.
- PMID 21170260.
- PMID 24976724.
- PMID 26295972.
- PMID 26850374.
- PMID 26558688.
- ^ "Colorectal Cancer Atlas". Archived from the original on January 13, 2016.
