Reduction of nitro compounds
The reduction of nitro compounds are
Aromatic nitro compounds
Reduction to anilines
The reduction of nitroaromatics is conducted on an industrial scale.[1] Many methods exist, such as:
- Catalytic hydrogenation using: platinum(IV) oxide, or Urushibara nickel.[6]
- Iron in acidic media.[7][8][9]
- Sodium hydrosulfite[10]
- Sodium sulfide (or hydrogen sulfide and base). Illustrated by the selective reduction of dinitrophenol to the nitroaminophenol.[11]
- Tin(II) chloride[12]
- Titanium(III) chloride
- Samarium[13]
- Hydroiodic acid[14]
Reduction to hydroxylamines
Several methods have been described for the production of aryl hydroxylamines from aryl nitro compounds:
- Raney nickel and hydrazine at 0-10 °C[15]
- Electrolytic reduction[16]
- Zinc metal in aqueous ammonium chloride[17]
- Catalytic Rhodium on carbon with excess hydrazine monohydrate at room temperature [18]
Reduction to hydrazine compounds
Treatment of nitroarenes with excess zinc metal results in the formation of N,N'-diarylhydrazine.[19]
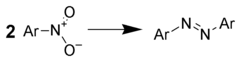
Reduction to azo compounds
Treatment of aromatic nitro compounds with metal hydrides gives good yields of azo compounds. For example, one could use:
- Lithium aluminium hydride[20]
- Zinc metal with sodium hydroxide.[19] (Excess zinc will reduce the azo group to a hydrazino compound.)
Aliphatic nitro compounds
Reduction to hydrocarbons
Hydrodenitration (replacement of a nitro group with hydrogen) is difficult to achieve but can be effected by catalytic hydrogenation over platinum on silica gel at high temperatures.[21] The reaction can also be effected through radical reaction with tributyltin hydride and a radical initiator, AIBN as an example.[22]
Reduction to amines
Aliphatic nitro compounds can be reduced to aliphatic amines by several reagents:
- Catalytic hydrogenation using
- Iron metal in refluxing acetic acid[25]
- Samarium diiodide[26]
- Raney nickel, platinum on carbon, or zinc dust and formic acid or ammonium formate[6]
α,β-Unsaturated nitro compounds can be reduced to saturated amines by:
- Catalytic hydrogenation over palladium-on-carbon
- Iron metal
- Lithium aluminium hydride[27] (Note: Hydroxylamines and oximes are typical impurities.)
- Lithium borohydride or sodium borohydride and trimethylsilyl chloride[28]
- Red-Al[29]
Reduction to hydroxylamines
Aliphatic nitro compounds can be reduced to aliphatic hydroxylamines using diborane.[30]
The reaction can also be carried out with zinc dust and ammonium chloride:[31][32][33]
- R-NO2 + 4 NH4Cl + 2 Zn → R-NH-OH + 2 ZnCl2 + 4 NH3 + H2O
Reduction to oximes
Nitro compounds are typically reduced to oximes using metal salts, such as tin(II) chloride[34] or chromium(II) chloride.[35] Additionally, catalytic hydrogenation using a controlled amount of hydrogen can generate oximes.[36]
References
- ISBN 978-3527306732.
- ^ Allen, C. F. H.; VanAllan, J. (1955). "2-Amino-p-cymene". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 63. - ^ Bavin, P. M. G. (1973). "2-Aminofluorene". Organic Syntheses; Collected Volumes, vol. 5, p. 30.
- ISBN 978-0-471-72091-1.
- hdl:2027.42/25034.
- ^ doi:10.1039/b009711j.
- doi:10.15227/orgsyn.044.0034).
{{cite journal}}: CS1 maint: multiple names: authors list (link - doi:10.15227/orgsyn.011.0032.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - .
- ^ Redemann, C. T.; Redemann, C. E. (1955). "5-Amino-2,3-dihydro-1,4-phthalazinedione". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 69. - .
- ISBN 9780470842898.
- .
- .
- .
- ^ Harman, R. E. (1963). "Chloro-p-benzoquinone". Organic Syntheses; Collected Volumes, vol. 4, p. 148.
- ^ Kamm, O. (1941). "β-Phenylhydroxylamine". Organic Syntheses; Collected Volumes, vol. 1, p. 445.
- PMID 29847002.
- ^ a b Bigelow, H. E.; Robinson, D. B. (1955). "Azobenzene". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 103. - PMID 18102934.
- .
- ^ T. V. (Babu) RajanBabu, Philip C. Bulman Page, Benjamin R. Buckley, "Tri-n-butylstannane" Encyclopedia of Reagents for Organic Synthesis 2004, John Wiley & Sons. doi:10.1002/047084289X.rt181.pub2
- .
- ^ Dauben, Jr., H. J.; Ringold, H. J.; Wade, R. H.; Pearson, D. L.; Anderson, Jr., A. G. (1963). "Cycloheptanone". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 4, p. 221. - .
- .
- .
- .
- .
- doi:10.1021/jo01020a002.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ISBN 978-0-08-010746-2, retrieved 2021-01-27
- PMID 24341483.
- .
- .
- .
- .