Reductive amination
| Reductive amination | |
|---|---|
| Reaction type | Coupling reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000335 |
Reductive amination (also known as reductive alkylation) is a form of

Reaction process
Reductive amination occurs between a carbonyl such as an aldehyde or ketone and an amine in the presence of a reducing agent.[2] The reaction conditions are neutral or weakly acidic.[2]

The amine first reacts with the carbonyl group to form a
There are two ways to conduct a reductive amination reaction: direct and indirect.[2]
Direct Reductive Amination
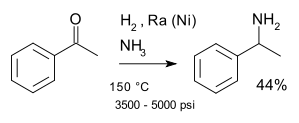
In a direct reaction, the carbonyl and amine starting materials and the reducing agent are combined and the reductions are done sequentially.[2] These are often one pot reactions since the imine intermediate is not isolated before the final reduction to the product.[2] Instead, as the reaction proceeds, the imine becomes favoured for reduction over the carbonyl starting material.[2] The two most common methods for direct reductive amination are hydrogenation with catalytic platinum, palladium, or nickel catalysts and the use of hydride reducing agents like cyanoborohydride (NaBH3CN).[2]
Indirect Reductive Amination
Indirect reductive amination, also called a stepwise reduction, isolates the imine intermediate.[2] In a separate step, the isolated imine intermediate is reduced to form the amine product.[2]
Designing a reductive amination reaction
There are many considerations to be made when designing a reductive amination reaction.[5]
- Chemoselectivity issues may arise since the carbonyl group is also reducible.
- The reaction between the carbonyl and amine are in equilibrium, with favouring for the carbonyl side unless water is removed from the system.
- Reducible intermediates may appear in the reaction which can affect chemoselectivity.
- The amine substrate, imine intermediate or amine product might deactivate the catalyst.
- Acyclic imines have E/Z isomers. This makes it difficult to create enantiopure chiral compounds through stereoselective reductions.
To solve the last issue, asymmetric reductive amination reactions can be used to synthesize an enantiopure product of chiral amines.[5] In asymmetric reductive amination, a carbonyl that can be converted from achiral to chiral is used.[6] The carbonyl undergoes condensation with an amine in the presence of H2 and a chiral catalyst to form the imine intermediate, which is then reduced to form the amine.[6] However, this method is still limiting to synthesize primary amines which are non-selective and prone to overalkylation.[6]
Common reducing agents
Sodium Borohydride
NaBH4 reduces both imines and carbonyl groups.[3] However, it is not very selective and can reduce other reducible functional groups present in the reaction.[3] To ensure that this does not occur, reagents with weak electrophilic carbonyl groups, poor nucleophilic amines and sterically hindered reactive centres should not be used, as these properties do not favour the reduction of the carbonyl to form an imine and increases the chance that other functional groups will be reduced instead.[3]
Sodium Cyanoborohydride
Sodium cyanoborohydride is soluble in hydroxylic solvents, stable in acidic solutions, and has different selectivities depending on the pH.[2] At low pH values, it efficiently reduces aldehydes and ketones.[7] As the pH increases, the reduction rate slows and instead, the imine intermediate becomes preferential for reduction.[7] For this reason, NaBH3CN is an ideal reducing agent for one-pot direct reductive amination reactions that don't isolate the intermediate imine.[2]
When used as a reducing agent, NaBH3CN can release toxic by-products like HCN and NaCN during work up.[2]
This reaction is related to the
A classic
Additionally, there exist many systems which catalyze reductive amination with a hydrogenation catalyst.[11] Generally, catalysis is preferred to stoichiometric reactions to enable the reaction to be more efficient, more atom economic, and to produce less waste.[12] This can be either a homogeneous catalytic system or heterogeneous system.[11] These systems provide an alternative method which is efficient, requires fewer volatile reagents and is redox economic.[11][13] As well, this method can be used in the reduction of alcohols, along with aldehydes and ketones to form the amine product.[11] One example of a heterogeneous catalytic system is the reductive amination of alcohols using the Ni-catalyzed system.[11][14]

Nickel is commonly used as a catalyst for reductive amination because of its abundance and relatively good catalytic activity.[11][15] An example of a homogeneous catalytic system is the reductive amination of ketones done with an iridium catalyst.[16] Additionally, it has been shown to be effective to use a homogeneous Iridium (III) catalyst system to reductively aminate carboxylic acids, which in the past has been more difficult than aldehydes and ketones.[12] Homogeneous catalysts are often favored because they are more environmentally and economically friendly compared to most heterogeneous systems. [11]
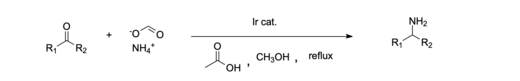
In industry, tertiary amines such as
In green chemistry
Reductive amination is commonly used over other methods for introducing amines to alkyl substrates, such as SN2-type reactions with halides, since it can be done in mild conditions and has high selectivity for nitrogen-containing compounds.[17][18] Reductive amination can occur sequentially in one-pot reactions, which eliminates the need for intermediate purifications and reduces waste.[17] Some multistep synthetic pathways have been reduced to one step through one-pot reductive amination.[17] This makes it a highly appealing method to produce amines in green chemistry.
Biochemistry
In biochemistry,
In popular culture
In the critically acclaimed drama
See also
References
- ^ S2CID 248001189.
- ^ PMID 11667239.
- ^ .
- ISSN 0040-4020.
- ^ PMID 24158548, retrieved 6 November 2023
- ^ S2CID 240250685.
- ^ ISSN 0002-7863.
- ISBN 9780582444072.
- Comptes rendus(in French). 172: 223.
- ; Collected Volumes, vol. 3, p. 717.
- ^ ISSN 2073-4344.
- ^ ISSN 0021-9517.
- PMID 19294720.
- .
- S2CID 245795966.
- S2CID 199000460.
- ^ S2CID 248767494.
- S2CID 225001665.
- ^ Metzler, D. E. "Biochemistry—The Chemical Reactions of Living Cells, Vol. 2" 2nd Ed. Academic Press: San Diego, 2003.
- ^ ISSN 2296-4185.
- PMID 21291399.
- ^ S2CID 252271457.
External links
- Current methods for reductive amination
- Industrial reductive amination at BASF