Renal cell carcinoma
This article needs more primary sources. (April 2021) |  |
| Renal cell carcinoma | |
|---|---|
 | |
| Micrograph of the most common type of renal cell carcinoma (clear cell)—on right of the image; non-tumour kidney is on the left of the image. Nephrectomy specimen. H&E stain | |
| Specialty | Oncology |
Renal cell carcinoma (RCC) is a kidney cancer that originates in the lining of the proximal convoluted tubule, a part of the very small tubes in the kidney that transport primary urine. RCC is the most common type of kidney cancer in adults, responsible for approximately 90–95% of cases.[1] It is more common in men (with a male-to-female ratio of up to 2:1).[2] It is most commonly diagnosed in the elderly (especially in people over 75 years of age).[3]
Initial treatment is most commonly either partial or complete removal of the affected kidney(s).
The body is remarkably good at hiding the symptoms and as a result people with RCC often have advanced disease by the time it is discovered.
RCC is also associated with a number of
Signs and symptoms
Historically, medical practitioners expected a person to present with three findings. This classic triad
Other signs and symptom may include
Risk factors
Lifestyle
The greatest risk factors for RCC are lifestyle-related; smoking, obesity and hypertension (high blood pressure) have been estimated to account for up to 50% of cases.[16] Occupational exposure to some chemicals such as asbestos, cadmium, lead, chlorinated solvents, petrochemicals and PAH (polycyclic aromatic hydrocarbon) has been examined by multiple studies with inconclusive results.[17][18][19] Another suspected risk factor is the long term use of
Finally, studies have found that women who have had a hysterectomy are at more than double the risk of developing RCC than those who have not.[21] Moderate alcohol consumption, on the other hand, has been shown to have a protective effect.[22]
Genetics
Hereditary factors have a minor impact on individual susceptibility with immediate relatives of people with RCC having a two to fourfold increased risk of developing the condition.
The most significant disease affecting risk however is not genetically linked – patients with acquired cystic disease of the kidney requiring dialysis are 30 times more likely than the general population to develop RCC.[26]
Pathophysiology
The tumour arises from the cells of the
Diagnosis
The first steps taken to diagnose this condition are consideration of the signs and symptoms, and a
Although this disease lacks characterization in the early stages of tumor development, considerations based on diverse clinical manifestations, as well as resistance to
Classification
Renal cell carcinoma (RCC) is not a single entity, but rather a collection of different types of
| Classification of the Common Histological Subtypes of Renal Cell Carcinoma[25] | |||
|---|---|---|---|
| Renal Cell Carcinoma Subtype | Frequency | Genetic Abnormalities | Characteristics |
|
Clear Cell Renal Cell Carcinoma (CCRCC)  |
60–70% |
|
|
| Papillary Renal Cell Carcinoma (PRCC)
cuboid cells with scanty cytoplasm and Type 2 PRCC consist of papillae covered by large eosinophilic cells arranged in an irregular or pseudostratified manner. |
10–15% |
| |
Chromophobe Renal Cell Carcinoma (ChRCC)
 |
3–5% |
|
|
| Clinical, Pathological and Genetic Features of Uncommon RCC Subtypes Included in the 2004 WHO Classification of RCC Pathology[30][33] | ||||
|---|---|---|---|---|
| RCC subtype | Clinical features | Cell/Tissue Characteristics | Genetics | Prognosis |
| Multilocular Cystic RCC |
|
Clear cytoplasm, small dark nuclei | 3p deletion as observed in CCRCC |
|
| Carcinoma of the Collecting Ducts of Bellini |
|
High-grade tumor cells with eosinophilic cytoplasm | Variable results: LOH on chromosomes 1q, 6p, 8p,9p, 13q, 19q32 and 21q; c-erB2 amplification associated with unfavorable outcome |
|
| Medullary Carcinoma |
|
Haemorrhage and necrosis , high-grade tumour cells with eosinophilic cytoplasm
|
Not well defined |
|
| Xp11.2 Translocation Carcinoma |
|
|
overexpression of the TFE3 protein
|
|
| Mucinous Tubular Spindle Cell Carcinoma |
|
Tubules, extracellular spindle cells
|
Not well defined; Losses involving chromosomes 1, 4, 6, 8, 9, 11, 13, 14, 15, 18, 22 reported; 3p alterations and gain of chromosome 7, and 17 not present |
|
| Post-Neuroblastoma Renal Cell Carcinoma |
|
Eosinophilic cells with oncocytoid features (same as CCRCC) | Not well defined; Loss of multiple chromosomal loci observed | Similar to other common RCC subtypes |
Array-based karyotyping can be used to identify characteristic chromosomal aberrations in renal tumors with challenging morphology.
The 2004 World Health Organization (WHO) classification of genitourinary tumours recognizes over 40 subtypes of renal neoplasms. Since the publication of the latest iteration of the WHO classification in 2004, several novel renal tumour subtypes have been described:[37]
- clear cell renal cell carcinoma with smooth muscle stroma[38]
- Mucinous tubular and spindle cell carcinoma (MTSCC) [37]
- Multilocular cystic clear cell renal cell carcinoma[37]
- Tubulocystic renal cell carcinoma
- Thyroid-like follicular renal cell carcinoma
- Acquired cystic kidney disease-associated renal cell carcinoma
- Renal cell carcinoma with t(6;11) translocation (TFEB)
- Hybrid oncocytoma/chromophobe renal cell carcinoma
- Hereditary leiomyomatosis and renal cell carcinoma(HLRCC)
Laboratory tests
Laboratory tests are generally conducted when the patient presents with signs and symptoms that may be characteristic of kidney impairment. They are not primarily used to diagnose kidney cancer, due to its asymptomatic nature and are generally found incidentally during tests for other illnesses such as gallbladder disease.[39] In other words, these cancers are not detected usually because they do not cause pain or discomfort when they are discovered. Laboratory analysis can provide an assessment on the overall health of the patient and can provide information in determining the staging and degree of metastasis to other parts of the body (if a renal lesion has been identified) before treatment is given.[citation needed]
Urine analysis
The presence of blood in urine is a common presumptive sign of renal cell carcinoma. The
Complete blood cell count
The CBC provides a quantified measure of the different cells in the whole blood sample from the patient. Such cells examined for in this test include red blood cells (
Blood chemistry
Blood chemistry tests are conducted if renal cell carcinoma is suspected as
Radiology
The characteristic appearance of renal cell carcinoma (RCC) is a solid renal lesion which disturbs the renal contour. It will frequently have an irregular or lobulated margin and may be seen as a lump on the lower pelvic or abdomen region. Traditionally, 85 to 90% of solid renal masses will turn out to be RCC but cystic renal masses may also be due to RCC.
Deciding on the
The main imaging tests performed in order to identify renal cell carcinoma are pelvic and abdominal CT scans, ultrasound tests of the kidneys (ultrasonography), MRI scans, intravenous pyelogram (IVP) or renal angiography.
Computed tomography
Contrast-enhanced
Ultrasound
Ultrasonographic examination can be useful in evaluating questionable
A
Magnetic resonance imaging
Intravenous pyelogram
Renal angiography
Renal
Staging
The staging of renal cell carcinoma is the most important factor in predicting its prognosis.[57] Staging can follow the TNM staging system, where the size and extent of the tumour (T), involvement of lymph nodes (N) and metastases (M) are classified separately. Also, it can use overall stage grouping into stage I–IV, with the 1997 revision of AJCC described below:[57]
| Stage I | Tumour of a diameter of 7 cm (approx. 2 3⁄4 inches) or smaller, and limited to the kidney. No lymph node involvement or metastases to distant organs. |
| Stage II | Tumour larger than 7.0 cm but still limited to the kidney. No lymph node involvement or metastases to distant organs. |
| Stage III any of the following |
Tumor of any size with involvement of a nearby lymph node but no metastases to distant organs. Tumour of this stage may be with or without spread to fatty tissue around the kidney, with or without spread into the large veins leading from the kidney to the heart. |
| Tumour with spread to fatty tissue around the kidney and/or spread into the large veins leading from the kidney to the heart, but without spread to any lymph nodes or other organs. | |
| Stage IV any of the following |
Tumour that has spread directly through the fatty tissue and the fascia ligament-like tissue that surrounds the kidney. |
| Involvement of more than one lymph node near the kidney | |
| Involvement of any lymph node not near the kidney | |
| Distant metastases, such as in the lungs, bone, or brain. |
At diagnosis, 30% of renal cell carcinomas have spread to the ipsilateral renal vein, and 5–10% have continued into the inferior vena cava.[58]
Histopathology
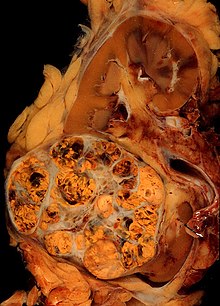


The gross and microscopic appearance of renal cell carcinomas is highly variable. The renal cell carcinoma may present reddened areas where
Recent studies have brought attention to the close association of the type of cancerous cells to the aggressiveness of the condition. Some studies suggest that these cancerous cells accumulate glycogen and lipids, their cytoplasm appear "clear", the nuclei remain in the middle of the cells, and the cellular membrane is evident.
The most common cell type exhibited by renal cell carcinoma is the
The recommended histologic grading schema for RCC is the Fuhrman system (1982), which is an assessment based on the microscopic morphology of a neoplasm with haematoxylin and eosin (H&E staining). This system categorises renal cell carcinoma with grades 1, 2, 3, 4 based on nuclear characteristics. The details of the Fuhrman grading system for RCC are shown below:[63]
| Grade Level | Nuclear Characteristics |
|---|---|
| Grade I | Nuclei appear round and uniform, 10 μm; nucleoli are inconspicuous or absent. |
| Grade II | Nuclei have an irregular appearance with signs of lobe formation, 15 μm; nucleoli are evident. |
| Grade III | Nuclei appear very irregular, 20 μm; nucleoli are large and prominent. |
| Grade IV | Nuclei appear bizarre and multilobated, 20 μm or more; nucleoli are prominent. |
Nuclear grade is believed to be one of the most imperative prognostic factors in patients with renal cell carcinoma.[25] However, a study by Delahunt et al. (2007) has shown that the Fuhrman grading is ideal for clear cell carcinoma but may not be appropriate for chromophobe renal cell carcinomas and that the staging of cancer (accomplished by CT scan) is a more favourable predictor of the prognosis of this disease.[64] In relation to renal cancer staging, the Heidelberg classification system of renal tumours was introduced in 1976 as a means of more completely correlating the histopathological features with the identified genetic defects.[65]
Prevention
The risk of renal cell carcinoma can be reduced by maintaining a normal body weight.[66]
Management

The type of treatment depends on multiple factors and the individual, some of which include the stage of renal cell carcinoma (organs and parts of the body affected/unaffected), type of renal cell carcinoma, pre-existing or comorbid conditions and overall health and age of the person.[10][67] Every form of treatment has both risks and benefits; a health care professional will provide the best options that suit the individual circumstances.
If it has spread outside of the kidneys, often into the
Active surveillance
Active surveillance or "watchful waiting" is becoming more common as small renal masses or tumours are being detected and also within the older generation when surgery is not always suitable.[68] Active surveillance involves completing various diagnostic procedures, tests and imaging to monitor the progression of the RCC before embarking on a more high risk treatment option like surgery.[68] In the elderly, patients with co-morbidities, and in poor surgical candidates, this is especially useful.
Surgery
Different procedures may be most appropriate, depending on circumstances.
The recommended treatment for renal cell cancer may be nephrectomy or partial nephrectomy, surgical removal of all or part of the kidney.[4] This may include some of the surrounding organs or tissues or lymph nodes. If cancer is only in the kidneys, which is about 60% of cases, it can be cured roughly 90% of the time with surgery.
Small renal tumors (< 4 cm) are treated increasingly by partial nephrectomy when possible.[69][70][71] Most of these small renal masses manifest indolent biological behavior with excellent prognosis.[72] Nephron-sparing partial nephrectomy is used when the tumor is small (less than 4 cm in diameter) or when the patient has other medical concerns such as diabetes or hypertension.[10] The partial nephrectomy involves the removal of the affected tissue only, sparing the rest of the kidney, Gerota's fascia and the regional lymph nodes. This allows for more renal preservation as compared to the radical nephrectomy, and this can have positive long-term health benefits.[73] Larger and more complex tumors can also be treated with partial nephrectomy by surgeons with a lot of kidney surgery experience.[74]
Surgical nephrectomy may be "radical" if the procedure removes the entire affected kidney including Gerota's fascia, the adrenal gland which is on the same side as the affected kidney, and the regional retroperitoneal lymph nodes, all at the same time.[10] This method, although severe, is effective. But it is not always appropriate, as it is a major surgery that contains the risk of complication both during and after the surgery and can have a longer recovery time.[75] It is important to note that the other kidney must be fully functional, and this technique is most often used when there is a large tumour present in only one kidney.

In cases where the tumor has spread into the renal vein, inferior vena cava, and possibly the right atrium, this portion of the tumor can be surgically removed, as well. When the tumor involved the inferior vena cava, it is important to classify which parts of the vena cava are involved and to plan accordingly, as sometimes complete resection will involve an incision into the chest with increased morbidity. For this reason, Dr.
Laparoscopic cryotherapy can also be done on smaller lesions. Typically a biopsy is taken at the time of treatment. Intraoperative ultrasound may be used to help guide placement of the freezing probes. Two freeze/thaw cycles are then performed to kill the tumor cells. As the tumor is not removed followup is more complicated (see below) and overall disease-free rates are not as good as those obtained with surgical removal.
Surgery for metastatic disease: If metastatic disease is present surgical treatment may still a viable option. Radical and partial nephrectomy can still occur, and in some cases, if the metastasis is small this can also be surgically removed.[10] This depends on what stage of growth and how far the disease has spread.
Percutaneous ablative therapies
A probe is placed through the skin and into the tumor using real-time imaging of both the probe tip and the tumor by
The two main types of ablation techniques that are used for renal cell carcinoma are
Radio frequency ablation uses an electrode probe which is inserted into the affected tissue, to send radio frequencies to the tissue to generate heat through the friction of water molecules. The heat destroys the tumor tissue.[10] Cell death will generally occur within minutes of being exposed to temperatures above 50 °C.
Cryoablation also involves the insertion of a probe into the affected area,[10] however, cold is used to kill the tumor instead of heat. The probe is cooled with chemical fluids which are very cold. The freezing temperatures cause the tumor cells to die by causing osmotic dehydration, which pulls the water out of the cell destroying the enzyme, organelles, cell membrane and freezing the cytoplasm.[81]
Targeted drugs
Cancers often grow in an unbridled fashion because they are able to evade the immune system.
Other targeted therapy medications inhibit growth factors that have been shown to promote the growth and spread of tumours.[85][86] Most of these medications were approved within the past ten years.[87] These treatments are:[88]
- Nivolumab[89]
- Axitinib[90]
- Sunitinib[91]
- Cabozantinib[89]
- Everolimus
- Lenvatinib
- Pazopanib
- Bevacizumab
- Sorafenib
- Tivozanib
- Temsirolimus[92]
- Interleukin-2 (IL-2) has produced "durable remissions" in a small number of patients, but with substantial toxicity.[93]
- Interferon-α
For patients with metastatic cancer, sunitinib probably results in more progression of the cancer than pembrolizumab, axitinib and avelumab.[94] In comparison to pembrolizumab and axitinib, it probably results in more death, but it may slightly reduce serious unwanted effects.[94] When compared with combinations of immunotherapy (nivolumab and ipilimumab), sunitinib may lead to more progression and serious effects.[94] There may be little to no difference in progression, survival and serious effects between pazopanib and sunitib.[94]
Activity has also been reported for ipilimumab[95] but it is not an approved medication for renal cancer.[96]
More medications are expected to become available in the near future as several clinical trials are currently being conducted for new targeted treatments,
Chemotherapy
Adjuvant and neoadjuvant therapy
Adjuvant therapy, which refers to therapy given after a primary surgery, had for a long time not been found to be beneficial in renal cell cancer.[98] However in 2021 Pembrolizumab was approved for adjuvant treatment after showing promising disease-free survival improvements.[99]
Conversely, neoadjuvant therapy is administered before the intended primary or main treatment. In some cases neoadjuvant therapy has been shown to decrease the size and stage of the RCC to then allow it to be surgically removed.[86] This is a new form of treatment and the effectiveness of this approach is still being assessed in clinical trials.
Metastasis
Metastatic renal cell carcinoma (mRCC) is the spread of the primary renal cell carcinoma from the kidney to other organs. Approximately 25–30% of people have this metastatic spread by the time they are diagnosed with renal cell carcinoma.[100] This high proportion is explained by the fact that clinical signs are generally mild until the disease progresses to a more severe state.[101] The most common sites for metastasis are the lymph nodes, lung, bones, liver and brain.[11] How this spread affects the staging of the disease and hence prognosis is discussed in the "Diagnosis" and "Prognosis" sections.
MRCC has a poor prognosis compared to other cancers, although average survival times have increased in the last few years due to treatment advances. Average survival time in 2008 for the metastatic form of the disease was under a year,[102] and by 2013 this improved to an average of 22 months.[103] Despite this improvement the five-year survival rate for mRCC remains under 10%[104] and 20–25% of patients remain unresponsive to all treatments and in these cases, the disease has a rapid progression.[103]
The available treatments for RCC discussed in the "Treatment" section are also relevant for the metastatic form of the disease. Options include interleukin-2, which is a standard therapy for advanced renal cell carcinoma.[98] From 2007 to 2013, seven new treatments have been approved specifically for mRCC (sunitinib, temsirolimus, bevacizumab, sorafenib, everolimus, pazopanib and axitinib).[8] These new treatments are based on the fact that renal cell carcinomas are very vascular tumours – they contain a large number of blood vessels. The drugs aim to inhibit the growth of new blood vessels in the tumours, hence slowing growth and in some cases, reducing the size of the tumours.[105] Side effects unfortunately are quite common with these treatments and include:[106]
- Gastrointestinal effects – nausea, vomiting, diarrhea, anorexia
- Respiratory effects – coughing, dyspnea (difficulty breathing)
- Cardiovascular effects – hypertension (high blood pressure)
- Neurological effects – intracranial hemorrhage (bleeding into the brain), thrombosis (blood clots) in the brain
- Effects on the skin and mucous membranes – rashes, hand-foot syndrome, stomatitis
- Bone marrow suppression – resulting in reduced white blood cells, increasing the risk of infections plus anemia and reduced platelets
- Renal effects – impaired kidney function
- Fatigue
Radiotherapy and chemotherapy are more commonly used in the metastatic form of RCC to target the secondary tumours in the bones, liver, brain and other organs. While not curative, these treatments do provide relief for symptoms associated with the spread of tumours.[103]
Prognosis
The prognosis is influenced by several factors, including tumour size, degree of invasion and metastasis, histologic type, and nuclear grade.[25] Staging is the most important factor in the outcome of renal cell cancer. The following numbers are based on patients first diagnosed in 2001 and 2002 by the National Cancer Data Base:[107]
| Stage | Description | 5 Year Survival Rate |
|---|---|---|
| I | Confined to the kidney | 81% |
| II | Extend through the renal capsule, confined to Gerota's Fascia | 74% |
| III | Include the renal vein, or the hilar lymph nodes | 53% |
| IV | Includes tumors that are invasive to adjacent organs (except the adrenal glands), or distant metastases | 8% |
A Korean study estimated a
Histological grade is related to the aggressiveness of the cancer, and it is classified in 4 grades, with 1 having the best prognosis (five-year survival over 89%), and 4 with the worst prognosis (46% of five-year survival).
Some people have the renal cell cancer detected before they have symptoms (incidentally) because of the CT scan (Computed Tomography Imaging) or ultrasound. Incidentally diagnosed renal cell cancer (no symptoms) differs in outlook from those diagnosed after presenting symptoms of renal cell carcinoma or metastasis. The five-year survival rate was higher for incidental than for symptomatic tumours: 85.3% versus 62.5%. Incidental lesions were significantly lower stage than those that cause symptoms, since 62.1% patients with incidental renal cell carcinoma were observed with Stage I lesions, against 23% were found with symptomatic renal cell carcinoma.[110]
If it has metastasized to the lymph nodes, the five-year survival is around 5% to 15%. For metastatic renal cell carcinoma, factors which may present a poor prognosis include a low
Renal cell carcinoma is one of the cancers most strongly associated with paraneoplastic syndromes, most often due to ectopic hormone production by the tumour. The treatment for these complications of RCC is generally limited beyond treating the underlying cancer.
Epidemiology
The incidence of the disease varies according to geographic, demographic and, to a lesser extent, hereditary factors. There are some known risk factors, however the significance of other potential risk factors remains more controversial. The incidence of the cancer has been increasing in frequency worldwide at a rate of approximately 2–3% per decade[102] until the last few years where the number of new cases has stabilised.[17]
The incidence of RCC varies between sexes, ages, races and geographic location around the world. Men have a higher incidence than women (approximately 1.6:1)[98] and the vast majority are diagnosed after 65 years of age.[98] Asians reportedly have a significantly lower incidence of RCC than whites and while African countries have the lowest reported incidences, African Americans have the highest incidence of the population in the United States.[17] Developed countries have a higher incidence than developing countries, with the highest rates found in North America, Europe and Australia / New Zealand.[114]
History
Miril published the earliest unequivocal case of renal carcinoma in 1810.[116] He described the case of Françoise Levelly, a 35-year-old woman, who presented to Brest Civic Hospital on April 6, 1809, supposedly in the late stages of pregnancy.[115]
Koenig published the first classification of renal tumours based on macroscopic morphology in 1826. Koenig divided the tumors into scirrhous, steatomatous, fungoid and medullary forms.[117]
Hypernephroma controversy
Following the classification of the tumour, researchers attempted to identify the tissue of origin for renal carcinoma.
The
In 1893, Paul Sudeck challenged the theory postulated by Grawitz by publishing descriptions of renal tumours in which he identified atypical features within
Vigorous criticism of Grawitz was provided by Oskar Stoerk in 1908, who considered the
Foot and Humphreys, and Foote et al. introduced the term Renal Celled Carcinoma to emphasize a
Convincing evidence to settle the debate was offered by Oberling et al. in 1959 who studied the
See also
- Dysuria
- Interferon
- Interleukin-2
- Kidney cancer
- Knudson hypothesis[121]
- Rapamycin
- Stauffer syndrome
- Vinblastine
- Vasculogenic Mimicry
References
- ^ a b c d e f g Curti, B; Jana, BRP; Javeed, M; Makhoul, I; Sachdeva, K; Hu, W; Perry, M; Talavera, F (26 February 2014). Harris, JE (ed.). "Renal Cell Carcinoma". Medscape Reference. WebMD. Archived from the original on 7 March 2014. Retrieved 7 March 2014.
- ISBN 978-94-92671-19-6.
- PMID 25449206.
- ^ PMID 18391630.
- ^ "Kidney cancer". nhs.uk. 2017-10-23. Archived from the original on 3 October 2017. Retrieved 19 March 2018.
- ISBN 978-0721601878.
- ^ a b c Master, VA (November 2013). "Renal Cell Carcinoma". Merck Manual Professional. Merck Sharp & Dohme Corp. Archived from the original on 7 March 2014. Retrieved 7 March 2014.
- ^ S2CID 11526724.
- ^ PMID 29208439.
- ^ PMID 16339096.
- ^ PMID 8778606.
- ^ PMID 14532767.
- ISBN 978-1-62703-061-8.
- ISBN 978-1-62703-061-8.
- ISBN 978-1-62703-061-8.
- PMID 23468995.
- ^ PMID 21741761.
- S2CID 21271953.
- S2CID 29328889.
- PMID 21911634.
- PMID 18711701.
- PMID 22398178.
- PMID 20865085.
- S2CID 17905393.
- ^ S2CID 24535138.
- PMID 18187049.
- PMID 21515214.
- ISBN 978-0-7817-7153-5.
- PMID 23026395.
- ^ S2CID 2101851.
- ISBN 9780808924500.
- ISBN 9780808924500.
- PMID 16442207.
- PMID 18990225.
- PMID 18246049.
- S2CID 24420204.
- ^ PMID 24364021.
- PMID 21602815.
- PMID 19948453.
- PMID 14974475.
- PMID 20121140.
- PMID 22762401.
- PMID 12663704.
- PMID 19602467.
- PMID 6675440.
- S2CID 260321430.
- S2CID 1916092.
- ^ PMID 8421912.
- ^ PMID 916043.
- PMID 22025734.
- ^ PMID 17997455.
- PMID 3969475.
- PMID 1863349.
- PMID 3195400.
- PMID 18215972.
- PMID 11517066.
- ^ a b Kidney Cancer / General Information Archived 2011-11-01 at the Wayback Machine at Weill Cornell Medical College, James Buchanan Brady Foundation, Department of Urology
- PMID 9843299.
- ^ "Clear-cell Carcinoma, Hypernephroid Tumour, or Hypernephroma". Archived from the original on 2010-05-07. Retrieved 2010-03-31.
- PMID 1016084.
- ^ "Renal clear cell carcinoma (Grawitz tumor)". Atlas of Pathology. January 30, 2009. Archived from the original on March 10, 2009.
- PMID 23433880.
- .
- S2CID 27154220.
- S2CID 34796951.
- PMID 27557308.
- ISBN 978-1-62703-061-8.
- ^ ISBN 978-1-62703-061-8.
- PMID 9772865.
- PMID 10037361.
- PMID 9720519.
- S2CID 26763264.
- PMID 20171688.
- S2CID 829345.
- PMID 23510704.
- PMID 17175095.
- PMID 16098354.
- PMID 14767273.
- PMID 18380384.
- ISBN 978-1-62703-061-8.
- ^ ISBN 978-1-62703-061-8.
- S2CID 5743758.
- S2CID 12651841.
- ^ ISBN 978-1-62703-061-8.
- S2CID 36871651.
- ^ ISBN 978-1-62703-061-8.
- S2CID 15100003.
- PMID 22638109.
- ^ PMID 26406149.
- S2CID 27524034.
- PMID 17215529.
- ^ "TORISEL® (temsirolimus) -Safety Info- Renal Cell Carcinoma Treatment". www.torisel.com. Archived from the original on 13 April 2018. Retrieved 19 March 2018.
- ^ Renal Cell Carcinoma Archived 2016-07-05 at the Wayback Machine MedlinePlus Medical Encyclopedia. Retrieved on 2010-09-10
- ^ PMID 33058158.
- PMID 18049334.
- ^ "Archived copy" (PDF). Archived (PDF) from the original on 2015-02-06. Retrieved 2014-10-29.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ "Kidney Cancer". Cancer Research Institute. Archived from the original on 24 December 2017. Retrieved 19 March 2018.
- ^ PMID 16339096.[permanent dead link]
- ^ "FDA approves pembrolizumab for adjuvant treatment of renal cell carcinoma". FDA. 17 November 2021.
- S2CID 12091991.
- PMID 17408850.
- ^ PMID 18313224.
- ^ S2CID 205529087.
- PMID 23083849.
- PMID 22832091.
- PMID 23317510.
- ^ Kidney Cancer (Adult) – Renal Cell Carcinoma Archived 2016-04-05 at the Wayback Machine American Cancer Society. Retrieved on 2010-09-10
- ISSN 0368-2811.
- ^ [1] Archived 2015-12-05 at the Wayback Machine Renal Cancer Causes, Symptoms, Treatment. eMedicine Health. Retrieved on 2010-09-10
- PMID 10647646.
- PMID 14752067.
- PMID 10561319.
- PMID 12655523.
- ISBN 978-1-62703-061-8.
- ^ a b c d e Delahunt, Brett (March 8, 2009). "History of Renal Neoplasia" (PDF). United States and Canadian Academy of Pathology 2009 Annual Meeting. Archived from the original (PDF) on March 29, 2020. Retrieved April 27, 2013.
- ^ Delahunt, Brett; Thornton A (1996). "Renal cell carcinoma. A historical perspective". J Urol Pathol. 4: 31–49.
- PMID 15848734.
- .
- PMID 14861941.
- S2CID 4224899.
- PMID 19015066.
