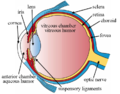
Retina
| Retina | |
|---|---|
 Right human eye cross-sectional view; eyes vary significantly among animals. | |
| Details | |
| Pronunciation | UK: /ˈrɛtɪnə/, US: /ˈrɛtənə/, pl. retinae /-ni/ |
| Part of | Eye |
| System | Visual system |
| Artery | Central retinal artery |
| Identifiers | |
| Latin | rēte, tunica interna bulbi |
| MeSH | D012160 |
| TA98 | A15.2.04.002 |
| TA2 | 6776 |
| FMA | 58301 |
| Anatomical terminology | |
The retina (from
The neural retina consists of several layers of
Light striking the retina initiates a cascade of chemical and electrical events that ultimately trigger nerve impulses that are sent to various visual centres of the brain through the fibres of the optic nerve. Neural signals from the rods and cones undergo processing by other neurons, whose output takes the form of action potentials in retinal ganglion cells whose axons form the optic nerve.[1]
In vertebrate
Structure
Inverted versus non-inverted retina
The vertebrate retina is inverted in the sense that the light-sensing cells are in the back of the retina, so that light has to pass through layers of neurons and capillaries before it reaches the photosensitive sections of the rods and cones.[5] The ganglion cells, whose axons form the optic nerve, are at the front of the retina; therefore, the optic nerve must cross through the retina en route to the brain. No photoreceptors are in this region, giving rise to the blind spot.[6] In contrast, in the cephalopod retina, the photoreceptors are in front, with processing neurons and capillaries behind them. Because of this, cephalopods do not have a blind spot.
Although the overlying neural tissue is partly transparent, and the accompanying
The cephalopods have a non-inverted retina, which is comparable in resolving power to the eyes of many vertebrates. Squid eyes do not have an analog of the vertebrate retinal pigment epithelium (RPE). Although their photoreceptors contain a protein, retinochrome, that recycles retinal and replicates one of the functions of the vertebrate RPE, cephalopod photoreceptors are likely not maintained as well as in vertebrates, and that as a result, the useful lifetime of photoreceptors in invertebrates is much shorter than in vertebrates.[10] Having easily replaced stalk eyes (some lobsters) or retinae (some spiders, such as Deinopis[11]) rarely occurs.
The cephalopod retina does not originate as an outgrowth of the brain, as the vertebrate one does. This difference suggests that vertebrate and cephalopod eyes are not homologous, but have evolved separately. From an evolutionary perspective, a more complex structure such as the inverted retina can generally come about as a consequence of two alternate processes - an advantageous "good" compromise between competing functional limitations, or as a historical maladaptive relic of the convoluted path of organ evolution and transformation. Vision is an important adaptation in higher vertebrates.
A third view of the "inverted" vertebrate eye is that it combines two benefits - the maintenance of the photoreceptors mentioned above, and the reduction in light intensity necessary to avoid blinding the photoreceptors, which are based on the extremely sensitive eyes of the ancestors of modern hagfish (fish that live in very deep, dark water).[12]
A recent study on the evolutionary purpose for the inverted retina structure from the APS (American Physical Society)[13] says that "The directional of glial cells helps increase the clarity of human vision. But we also noticed something rather curious: the colours that best passed through the glial cells were green to red, which the eye needs most for daytime vision. The eye usually receives too much blue—and thus has fewer blue-sensitive cones.
Further computer simulations showed that green and red are concentrated five to ten times more by the glial cells, and into their respective cones, than blue light. Instead, excess blue light gets scattered to the surrounding rods. This optimization is such that color vision during the day is enhanced, while night-time vision suffers very little".
Retinal layers
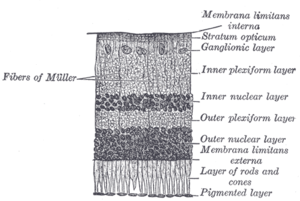


The vertebrate retina has 10 distinct layers.[15] From closest to farthest from the vitreous body:
- Müller cells
- Nerve fibre layer – axons of the ganglion cellbodies (a thin layer of Müller cell footplates exists between this layer and the inner limiting membrane)
- amacrine cells[2]
- Inner plexiform layer – contains the synapse between the bipolar cell axons and the dendrites of the ganglion and amacrine cells[2]
- Outer plexiform layer – projections of rods and cones ending in the rod spherule and cone pedicle, respectively, these make synapses with dendrites of bipolar cells and horizontal cells.[2] In the macular region, this is known as the Fiber layer of Henle.
- Outer nuclear layer – cell bodies of rods and cones
- External limiting membrane – layer that separates the inner segment portions of the photoreceptors from their cell nuclei
- Inner segment / outer segment layer – inner segments and outer segments of rods and cones, the outer segments contain a highly specialized light-sensing apparatus.[16][17]
- Retinal pigment epithelium – single layer of cuboidal epithelial cells (with extrusions not shown in diagram). This layer is closest to the choroid, and provides nourishment and supportive functions to the neural retina, The black pigment melanin in the pigment layer prevents light reflection throughout the globe of the eyeball; this is extremely important for clear vision.[18][19][20]
These layers can be grouped into four main processing stages—photoreception; transmission to
The
Additional structures, not directly associated with vision, are found as outgrowths of the retina in some vertebrate groups. In
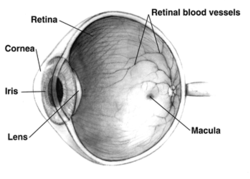
In adult humans, the entire retina is about 72% of a sphere about 22 mm in diameter. The entire retina contains about 7 million cones and 75 to 150 million rods. The optic disc, a part of the retina sometimes called "the blind spot" because it lacks photoreceptors, is located at the optic papilla, where the optic-nerve fibres leave the eye. It appears as an oval white area of 3 mm2. Temporal (in the direction of the temples) to this disc is the macula, at whose centre is the fovea, a pit that is responsible for sharp central vision, but is actually less sensitive to light because of its lack of rods. Human and non-human primates possess one fovea, as opposed to certain bird species, such as hawks, that are bifoviate, and dogs and cats, that possess no fovea, but a central band known as the visual streak.[citation needed] Around the fovea extends the central retina for about 6 mm and then the peripheral retina. The farthest edge of the retina is defined by the ora serrata. The distance from one ora to the other (or macula), the most sensitive area along the horizontal meridian, is about 32 mm.[clarification needed]
In section, the retina is no more than 0.5 mm thick. It has three layers of nerve cells and two of synapses, including the unique ribbon synapse. The optic nerve carries the ganglion-cell axons to the brain, and the blood vessels that supply the retina. The ganglion cells lie innermost in the eye while the photoreceptive cells lie beyond. Because of this counter-intuitive arrangement, light must first pass through and around the ganglion cells and through the thickness of the retina, (including its capillary vessels, not shown) before reaching the rods and cones. Light is absorbed by the retinal pigment epithelium or the choroid (both of which are opaque).
The
Between the ganglion-cell layer and the rods and cones are two layers of neuropils, where synaptic contacts are made. The neuropil layers are the outer plexiform layer and the inner plexiform layer. In the outer neuropil layer, the rods and cones connect to the vertically running bipolar cells, and the horizontally oriented horizontal cells connect to ganglion cells.
The central retina predominantly contains cones, while the peripheral retina predominantly contains rods. In total, the retina has about seven million cones and a hundred million rods. At the centre of the macula is the foveal pit where the cones are narrow and long, and arranged in a hexagonal mosaic, the most dense, in contradistinction to the much fatter cones located more peripherally in the retina.[23] At the foveal pit, the other retinal layers are displaced, before building up along the foveal slope until the rim of the fovea, or parafovea, is reached, which is the thickest portion of the retina. The macula has a yellow pigmentation, from screening pigments, and is known as the macula lutea. The area directly surrounding the fovea has the highest density of rods converging on single bipolar cells. Since its cones have a much lesser convergence of signals, the fovea allows for the sharpest vision the eye can attain.[2]
Though the rod and cones are a
Layers imagable with optical coherence tomography
Using optical coherence tomography (OCT), 18 layers can be identified in the retina. The layers and anatomical correlation are:[24][25][26]



From innermost to outermost, the layers identifiable by OCT are as follows:
| # | OCT Layer / Conventional Label | Anatomical Correlate | Reflectivity
|
Specific
anatomical boundaries? |
Additional
references |
|---|---|---|---|---|---|
| 1 | Posterior cortical vitreous | Posterior cortical vitreous | Hyper-reflective | Yes | [25] |
| 2 | Preretinal space | In eyes where the vitreous has fully or partially detached from the retina, this is the space created between the posterior cortical vitreous face and the internal limiting membrane of the retina. | Hypo-reflective | [25] | |
| 3 | Internal limiting membrane (ILM) | Formed by Müller cell endfeet
(unclear if it can be observed on OCT) |
Hyper-reflective | No | [25] |
Nerve fiber layer (NFL)
|
axons travelling towards the optic nerve
| ||||
| 4 | Ganglion cell layer (GCL) | amacrine cells )
|
Hypo-reflective | [25] | |
| 5 | Inner plexiform layer (IPL) | Synapses between ganglion cells
|
Hyper-reflective | [25] | |
| 6 | Inner nuclear layer (INL) | a) amacrine cell bodies
b) Müller cell nuclei
|
Hypo-reflective | [25] | |
| 7 | Outer plexiform layer (OPL) | horizontal cells
|
Hyper-reflective | [25] | |
| 8 | (Inner half) Henle's nerve fiber layer (HL) | Photoreceptor axons
(obliquely orientated fibres; not present in mid-peripheral or peripheral retina) |
Hypo-reflective | No | [25] |
| (Outer half) Outer nuclear layer (ONL) | The photoreceptor cell bodies | ||||
| 9 | External limiting membrane (ELM) | Made of Müller cells and photoreceptor inner segments
|
Hyper-reflective | [25] | |
| 10 | Myoid zone (MZ) | The innermost portion of the photoreceptor inner segment (IS) containing:
|
Hypo-reflective | No | [27][28] |
| 11 | Ellipsoid zone (EZ) | The outermost portion of the mitochondria
|
Very Hyper-reflective | No | [24][29][27][25][30][31] |
| IS/OS junction or Photoreceptor integrity line (PIL) | The photoreceptor connecting cilia which bridge the inner and outer segments of the photoreceptor cells. | ||||
| 12 | Photoreceptor outer segments (OS) | The photoreceptor outer segments (OS) which contain disks filled with opsin, the molecule that absorbs photons. | Hypo-reflective | [32][25] | |
| 13 | Interdigitation zone (IZ) | Apices of the RPE cells which encase part of the cone OSs.
Poorly distinguishable from RPE. Previously: "cone outer segment tips line" (COST) |
Hyper-reflective | No | |
| 14 | RPE/Bruch's complex | RPE phagosome zone | Very Hyper-reflective | No | [24][25] |
| RPE melanosome zone | Hypo-reflective | ||||
| RPE mitochondria zone + Junction between the RPE & Bruch's membrane | Very Hyper-reflective | ||||
| 15 | Choriocapillaris
|
Thin layer of moderate reflectivity in inner choroid | No | [25] | |
| 16 | Sattler's layer | Thick layer of round or ovalshaped hyperreflective profiles, with hyporeflective cores in mid-choroid | [25] | ||
| 17 | Haller's layer | Thick layer of oval-shaped hyperreflective profiles, with hyporeflective cores in outer choroid | [25] | ||
| 18 | Choroidal-scleral juncture | Zone at the outer choroid with a marked change in texture, in which large circular or ovoid profiles abut a
homogenous region of variable reflectivity |
[25] | ||
Development
Retinal development begins with the establishment of the eye fields mediated by the
In addition to guiding cell fate determination, cues exist in the retina to determine the dorsal-ventral (D-V) and nasal-temporal (N-T) axes. The D-V axis is established by a ventral to dorsal gradient of
Blood supply
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|
The retina is stratified into distinct layers, each containing specific cell types or cellular compartments[38] that have metabolisms with different nutritional requirements.[39] To satisfy these requirements, the ophthalmic artery bifurcates and supplies the retina via two distinct vascular networks: the choroidal network, which supplies the choroid and the outer retina, and the retinal network, which supplies the retina's inner layer.[40]
Although the inverted retina of vertebrates appears counter-intuitive, it is necessary for the proper functioning of the retina. The photoreceptor layer must be embedded in the retinal pigment epithelium (RPE), which performs at least seven vital functions,[41] one of the most obvious being to supply oxygen and other necessary nutrients needed for the photoreceptors to function.
Energy requirements
The energy requirements of the retina are even greater than that of the brain.[4] This is due to the additional energy needed to continuously renew the photoreceptor outer segments, of which 10% are shed daily.[4] Energy demands are greatest during dark adaptation when its sensitivity is most enhanced.[42] The choroid supplies about 75% of these nutrients to the retina and the retinal vasculature only 25%.[5]
When light strikes 11-cis-retinal (in the disks in the rods and cones), 11-cis-retinal changes to all-trans-retinal which then triggers changes in the opsins. Now, the outer segments do not regenerate the retinal back into the cis- form once it has been changed by light. Instead the retinal is pumped out to the surrounding RPE where it is regenerated and transported back into the outer segments of the photoreceptors. This recycling function of the RPE protects the photoreceptors against photo-oxidative damage[43][44] and allows the photoreceptor cells to have decades-long useful lives.
In birds
The bird retina is devoid of blood vessels, perhaps to give unobscured passage of light for forming images, thus giving better resolution. It is, therefore, a considered view that the bird retina depends for nutrition and oxygen supply on a specialized organ, called the "pecten" or pecten oculi, located on the blind spot or optic disk. This organ is extremely rich in blood vessels and is thought to supply nutrition and oxygen to the bird retina by diffusion through the vitreous body. The pecten is highly rich in alkaline phosphatase activity and polarized cells in its bridge portion – both befitting its secretory role.[45] Pecten cells are packed with dark melanin granules, which have been theorized to keep this organ warm with the absorption of stray light falling on the pecten. This is considered to enhance metabolic rate of the pecten, thereby exporting more nutritive molecules to meet the stringent energy requirements of the retina during long periods of exposure to light.[46]
Biometric identification and diagnosis of disease
The bifurcations and other physical characteristics of the inner retinal vascular network are known to vary among individuals,[47] and these individual variances have been used for biometric identification and for early detection of the onset of disease. The mapping of vascular bifurcations is one of the basic steps in biometric identification.[48] Results of such analyses of retinal blood vessel structure can be evaluated against the ground truth data[49] of vascular bifurcations of retinal fundus images that are obtained from the DRIVE dataset.[50] In addition, the classes of vessels of the DRIVE dataset have also been identified,[51] and an automated method for accurate extraction of these bifurcations is also available.[52] Changes in retinal blood circulation are seen with aging[53] and exposure to air pollution,[54] and may indicate cardiovascular diseases such as hypertension and atherosclerosis.[55][56][57] Determining the equivalent width of arterioles and venules near the optic disc is also a widely used technique to identify cardiovascular risks.[58]
Function
The retina translates an optical image into neural impulses starting with the patterned excitation of the colour-sensitive pigments of its rods and cones, the retina's photoreceptor cells. The excitation is processed by the neural system and various parts of the brain working in parallel to form a representation of the external environment in the brain.
The cones respond to bright light and mediate high-resolution colour vision during daylight illumination (also called photopic vision). The rod responses are saturated at daylight levels and do not contribute to pattern vision. However, rods do respond to dim light and mediate lower-resolution, monochromatic vision under very low levels of illumination (called scotopic vision). The illumination in most office settings falls between these two levels and is called mesopic vision. At mesopic light levels, both the rods and cones are actively contributing pattern information. What contribution the rod information makes to pattern vision under these circumstances is unclear.
The response of cones to various wavelengths of light is called their spectral sensitivity. In normal human vision, the spectral sensitivity of a cone falls into one of three subtypes, often called blue, green, and red, but more accurately known as short, medium, and long wavelength-sensitive cone subtypes. It is a lack of one or more of the cone subtypes that causes individuals to have deficiencies in colour vision or various kinds of
In the photoreceptors, exposure to light hyperpolarizes the membrane in a series of graded shifts. The outer cell segment contains a
When thus excited by light, the photoceptor sends a proportional response
In the retinal ganglion cells there are two types of response, depending on the receptive field of the cell. The receptive fields of retinal ganglion cells comprise a central, approximately circular area, where light has one effect on the firing of the cell, and an annular surround, where light has the opposite effect. In ON cells, an increment in light intensity in the centre of the receptive field causes the firing rate to increase. In OFF cells, it makes it decrease. In a linear model, this response profile is well described by a difference of Gaussians and is the basis for edge detection algorithms. Beyond this simple difference, ganglion cells are also differentiated by chromatic sensitivity and the type of spatial summation. Cells showing linear spatial summation are termed X cells (also called parvocellular, P, or midget ganglion cells), and those showing non-linear summation are Y cells (also called magnocellular, M, or parasol retinal ganglion cells), although the correspondence between X and Y cells (in the cat retina) and P and M cells (in the primate retina) is not as simple as it once seemed.
In the transfer of visual signals to the brain, the
Although there are more than 130 million retinal receptors, there are only approximately 1.2 million fibres (axons) in the optic nerve. So, a large amount of pre-processing is performed within the retina. The fovea produces the most accurate information. Despite occupying about 0.01% of the visual field (less than 2° of visual angle), about 10% of axons in the optic nerve are devoted to the fovea. The resolution limit of the fovea has been determined to be around 10,000 points. The information capacity is estimated at 500,000 bits per second (for more information on bits, see information theory) without colour or around 600,000 bits per second including colour.[59]
Spatial encoding

When the retina sends neural impulses representing an image to the brain, it spatially encodes (compresses) those impulses to fit the limited capacity of the optic nerve. Compression is necessary because there are 100 times more photoreceptor cells than ganglion cells. This is done by "decorrelation", which is carried out by the "centre–surround structures", which are implemented by the bipolar and ganglion cells.
There are two types of centre–surround structures in the retina – on-centres and off-centres. On-centres have a positively weighted centre and a negatively weighted surround. Off-centres are just the opposite. Positive weighting is more commonly known as excitatory, and negative weighting as inhibitory.
These centre–surround structures are not physical apparent, in the sense that one cannot see them by staining samples of tissue and examining the retina's anatomy. The centre–surround structures are logical (i.e., mathematically abstract) in the sense that they depend on the connection strengths between bipolar and ganglion cells. It is believed that the connection strength between cells is caused by the number and types of ion channels embedded in the synapses between the bipolar and ganglion cells.
The centre–surround structures are mathematically equivalent to the edge detection algorithms used by computer programmers to extract or enhance the edges in a digital photograph. Thus, the retina performs operations on the image-representing impulses to enhance the edges of objects within its visual field. For example, in a picture of a dog, a cat and a car, it is the edges of these objects that contain the most information. In order for higher functions in the brain (or in a computer for that matter) to extract and classify objects such as a dog and a cat, the retina is the first step to separating out the various objects within the scene.
As an example, the following matrix is at the heart of a computer algorithm that implements edge detection. This matrix is the computer equivalent to the centre–surround structure. In this example, each box (element) within this matrix would be connected to one photoreceptor. The photoreceptor in the centre is the current receptor being processed. The centre photoreceptor is multiplied by the +1 weight factor. The surrounding photoreceptors are the "nearest neighbors" to the centre and are multiplied by the −1/8 value. The sum of all nine of these elements is finally calculated. This summation is repeated for every photoreceptor in the image by shifting left to the end of a row and then down to the next line.
| -1/8 | -1/8 | -1/8 |
| -1/8 | +1 | -1/8 |
| -1/8 | -1/8 | -1/8 |
The total sum of this matrix is zero, if all the inputs from the nine photoreceptors are of the same value. The zero result indicates the image was uniform (non-changing) within this small patch. Negative or positive sums mean the image was varying (changing) within this small patch of nine photoreceptors.
The above matrix is only an approximation to what really happens inside the retina. The differences are:
- The above example is called "balanced". The term balanced means that the sum of the negative weights is equal to the sum of the positive weights so that they cancel out perfectly. Retinal ganglion cells are almost never perfectly balanced.
- The table is square while the centre–surround structures in the retina are circular.
- Neurons operate on axons. Computers operate on a single floating-point number that is essentially constant from each input pixel. (The computer pixel is basically the equivalent of a biological photoreceptor.)
- The retina performs all these calculations in parallel while the computer operates on each pixel one at a time. The retina performs no repeated summations and shifting as would a computer.
- Finally, the horizontal and amacrine cellsplay a significant role in this process, but that is not represented here.
Here is an example of an input image and how edge detection would modify it.
Once the image is spatially encoded by the centre–surround structures, the signal is sent out along the optic nerve (via the axons of the ganglion cells) through the
Simplified signal flow: Photoreceptors → Bipolar → Ganglion → Chiasm → LGN → V1 cortex
Clinical significance
There are many inherited and acquired diseases or disorders that may affect the retina. Some of them include:
- Retinitis pigmentosa is a group of genetic diseases that affect the retina and cause the loss of night vision and peripheral vision.
- Macular degeneration describes a group of diseases characterized by loss of central vision because of death or impairment of the cells in the macula.
- in the retina.
- In laser photocoagulation and pars plana vitrectomy.
- Both diabetes mellitus can cause damage to the tiny blood vessels that supply the retina, leading to hypertensive retinopathy and diabetic retinopathy.
- Retinoblastoma is a cancer of the retina.
- Retinal diseases in sudden acquired retinal degeneration.
- Lipaemia retinalis is a white appearance of the retina, and can occur by lipid deposition in lipoprotein lipase deficiency.
- Retinal Detachment. The neural retina occasionally detaches from the pigment epithelium. In some instances, the cause of such detachment is injury to the eyeball that allows fluid or blood to collect between the neural retina and the pigment epithelium. Detachment is occasionally caused by contracture of fine collagenous fibrils in the vitreous humor, which pull areas of the retina toward the interior of the globe.[23]
- Night blindness: Night blindness occurs in any person with severe vitamin A deficiency. The reason for this is that without vitamin A, the amounts of retinal and rhodopsin that can be formed are severely depressed. This condition is called night blindness because the amount of light available at night is too little to permit adequate vision in vitamin A–deficient persons.[18]
In addition, the retina has been described as a "window" into the brain and body, given that abnormalities detected through an examination of the retina can discover both neurological and systemic diseases.[61]
Diagnosis
A number of different instruments are available for the diagnosis of diseases and disorders affecting the retina. Ophthalmoscopy and fundus photography have long been used to examine the retina. Recently, adaptive optics has been used to image individual rods and cones in the living human retina, and a company based in Scotland has engineered technology that allows physicians to observe the complete retina without any discomfort to patients.[62]
The
Treatment
Treatment depends upon the nature of the disease or disorder.
Common treatment modalities
The following are commonly modalities of management for retinal disease:
- anti-VEGF or corticosteroidagents
- Vitreoretinal surgery
- Use of nutritional supplements
- Modification of systemic risk factors for retinal disease
Uncommon treatment modalities
Retinal gene therapy
Several clinical trials have already reported positive results using rAAV to treat
The unique architecture of the retina and its relatively immune-privileged environment help this process.
This strategy is effective against a number of retinal diseases that have been studied, including neovascular diseases that are features of
Organ transplantation
History
Around 300
Between 1011 and 1021 CE,
In 1604, Johannes Kepler worked out the optics of the eye and decided that the retina must be where sight begins. He left it up to other scientists to reconcile the inverted retinal image with our perception of the world as upright.[79]
In 1894, Santiago Ramón y Cajal published the first major characterization of retinal neurons in Retina der Wirbelthiere (The Retina of Vertebrates).[80]
George Wald, Haldan Keffer Hartline, and Ragnar Granit won the 1967 Nobel Prize in Physiology or Medicine for their scientific research on the retina.[81]
A recent University of Pennsylvania study calculated that the approximate bandwidth of human retinas is 8.75 megabits per second, whereas a guinea pig's retinal transfer rate is 875 kilobits per second.[82]
In 2012,
Additional images
-
The structures of the eye labeled
-
Another view of the eye and the structures of the eye labeled
-
Illustration of image as 'seen' by the retina independent of optic nerve and striate cortex processing
See also
- Adeno associated virus and gene therapy of the human retina
- Charles Schepens – "the father of modern retinal surgery"
- Evolution of the eye
- Duplex retina
- Retinal scan
- Retinal vein occlusion
- List of xanthoma variants associated with hyperlipoproteinemia subtypes
- Rhodopsin
- Persistence of vision
References
- ISBN 978-1-58112-468-2.
- ^ a b c d e f g "Sensory Reception: Human Vision: Structure and function of the Human Eye" vol. 27, Encyclopædia Britannica, 1987
- ^ "Penn Researchers Calculate How Much the Eye Tells the Brain" (Press release). PENN Medicine. 26 July 2006. Archived from the original on 11 March 2013. Retrieved 22 April 2022.
- ^ PMID 33935647.
- ^ PMID 21413391. Retrieved 1 January 2018.
- ^ Kolb, Helga. "Photoreceptors". Webvision. Retrieved 11 January 2018.
- PMID 17485670.
- .
- ^ S2CID 5045660.
- PMID 235007.
- ^ "How spiders see the world – Australian Museum". www.australian.museum. Archived from the original on 12 September 2017. Retrieved 5 December 2017.
- PMID 21356325.
- PMID 20482021.
- ^ Foundations of Vision Archived 3 December 2013 at the Wayback Machine, Brian A. Wandell
- ^ "The Retinal Tunic". Virginia–Maryland Regional College of Veterinary Medicine. Archived from the original on 18 May 2007.
- PMID 27260426.
- PMID 22074925.
- ^ a b Guyton and Hall Physiology. p. 612.
- PMID 21091424.
- S2CID 1351539.
- ISBN 978-0-19-515956-1.
- ISBN 978-0-03-910284-5.
- ^ a b Guyton and Hall Physiology. p. 609.
- ^ PMID 29037595.
- ^ PMID 24755005.
- OCLC 964379175.
- ^ ISBN 978-3642120404.
- .
- ^ "Outer Retinal Layers as Predictors of Vision Loss". Review of Ophthalmology.
- ^ "The ABCs of OCT". Review of Optometry.
- PMID 19465337.
- ^ Boston, Marco A. Bonini Filho, MD, and Andre J. Witkin, MD. "Outer Retinal Layers as Predictors of Vision Loss". Retrieved 7 April 2018.
{{cite news}}: CS1 maint: multiple names: authors list (link) - ^ PMID 23071378.
- PMID 7892602.
- S2CID 15038502.
- PMID 15036211.
- ^ PMID 31399471.
- OCLC 745905738.
- S2CID 21312546.
- ^ Kiel, Jeffrey W. Anatomy. Morgan & Claypool Life Sciences. Archived from the original on 5 December 2017. Retrieved 17 April 2017.
- ^ Strauss, Olaf. "The retinal pigment epithelium". Webvision. Retrieved 1 January 2018.
- PMID 37081065.
- ^ "LIGHT-INDUCED DAMAGE to the RETINA". photobiology.info. Retrieved 23 February 2023.
- ^ "Diagrammatic representation of disc shedding and phagosome retrieval into the pigment epithelial cell". Archived from the original on 21 September 2012. Retrieved 22 April 2022.
- from the original on 9 October 2014.
- from the original on 14 July 2015.
- PMID 7288393.
- ISBN 978-3-642-23671-6. Archived(PDF) from the original on 9 August 2017.
- ^ "Retinal fundus images – Ground truth of vascular bifurcations and crossovers". University of Groningen. Retrieved 20 April 2018.
- ^ "DRIVE: Digital Retinal Images for Vessel Extraction". Image Sciences Institute, Utrecht University. Archived from the original on 6 August 2020. Retrieved 20 April 2018.
- S2CID 7705121.
- S2CID 14654500.
- PMID 21152417.
- PMID 23777785.
- PMID 7155524.
- PMID 12149100.
- S2CID 7434103.
- PMID 15177969.
- OCLC 962009228.
- OCLC 62034580.
- PMID 34687632.
- ^ Seeing into the Future Archived 12 February 2012 at the Wayback Machine Ingenia, March 2007
- PMID 27682886.
- PMID 30635610.
- PMID 15960597.
- ^ PMID 19583479.
- ^ PMID 19953081.
- ^ PMID 18441370.
- ^ PMID 19854499.
- (PDF) from the original on 11 August 2017.
- PMID 18774912.
- PMID 21606598.
- PMID 12756418.
- S2CID 9926758.
- S2CID 30202286.
- ^ PMID 15454954.
- PMID 19984605.
- ^ Sabra, A. I. (Ed.). (1011–1021/1989). The optics of Ibn Al-Haytham: Books I-III: On direct vision (A. I. Sabra, Trans.). The Warburg Institute.
- PMID 4567856. Retrieved 23 February 2023.
- ^ "Santiago Ramón y Cajal – Biographical". www.nobelprize.org. Archived from the original on 6 October 2015. Retrieved 20 October 2015.
- ^ "Nobelprize.org". nobelprize.org. Archived from the original on 30 June 2017. Retrieved 5 December 2017.
- ^ Reilly, Michael. "Calculating the speed of sight". New Scientist. Archived from the original on 31 May 2015. Retrieved 5 December 2017.
- S2CID 4415311.
- PMID 18329018.
- ^ "About: EyeWire". Archived from the original on 13 February 2012. Retrieved 26 March 2012.
- ^ "Retina << EyeWire". Archived from the original on 24 March 2012. Retrieved 27 March 2012.
- ^ "EyeWire". Archived from the original on 24 April 2012. Retrieved 27 March 2012.
Further reading
- S. Ramón y Cajal, Histologie du Système Nerveux de l'Homme et des Vertébrés, Maloine, Paris, 1911.
- Rodieck RW (1965). "Quantitative analysis of cat retinal ganglion cell response to visual stimuli". Vision Res. 5 (11): 583–601. PMID 5862581.
- Wandell, Brian A. (1995). Foundations of vision. Sunderland, Mass: Sinauer Associates. ISBN 978-0-87893-853-7.
- Wässle H, Boycott BB (1991). "Functional architecture of the mammalian retina". Physiol. Rev. 71 (2): 447–480. PMID 2006220.
- Schulz HL, Goetz T, Kaschkoetoe J, Weber BH (2004). "The Retinome – Defining a reference transcriptome of the adult mammalian retina/retinal pigment epithelium". BMC Genomics (about a PMID 15283859.
- Dowling, John (2007). "Retina". .
External links
- Histology of the Eye, edited by William Krause, Dept. Pathology and Anatomical science, University of Missouri School of Medicine
- Eye, Brain, and Vision – online book – by David Hubel
- Kolb, H., Fernandez, E., & Nelson, R. (2003). Webvision: The neural organization of the vertebrate retina. Salt Lake City, Utah: John Moran Eye Center, University of Utah. Retrieved 22 July 2014.
- Retinal layers image. NeuroScience 2nd Ed at United States National Library of Medicine
- Jeremy Nathans's Seminars: "The Vertebrate Retina: Structure, Function, and Evolution"
- Retina – Cell Centered Database
- Histology image: 07901loa – Histology Learning System at Boston University
- MedlinePlus Encyclopedia: 002291