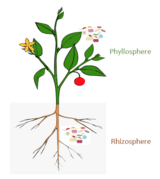
Rhizosphere
This article needs attention from an expert in Plants. See the talk page for details. (September 2020) |
The rhizosphere is the narrow region of soil or substrate that is directly influenced by root secretions and associated soil microorganisms known as the root microbiome.[2] Soil pores in the rhizosphere can contain many bacteria and other microorganisms that feed on sloughed-off plant cells, termed rhizodeposition,[3] and the proteins and sugars released by roots, termed root exudates.[4] This symbiosis leads to more complex interactions, influencing plant growth and competition for resources. Much of the nutrient cycling and disease suppression by antibiotics required by plants occurs immediately adjacent to roots due to root exudates and metabolic products of symbiotic and pathogenic communities of microorganisms.[5] The rhizosphere also provides space to produce allelochemicals to control neighbours and relatives.[6]
The rhizoplane refers to the root surface including its associated soil particles which closely interact with each other.[7] The plant-soil feedback loop and other physical factors occurring at the plant-root soil interface are important selective pressures in communities and growth in the rhizosphere and rhizoplane.[8]
Background
The term "rhizosphere" was used first in 1904 by the German plant physiologist Lorenz Hiltner to describe how plant roots interface with the surrounding soil.[9][10] The prefix rhiza- comes from the Greek, meaning "root". Hiltner postulated the rhizosphere was a region surrounding the plant roots and populated with microorganisms under some degree of control by chemicals released from the plant roots.[9][8]
Chemical interactions
| Part of a series on |
| Microbiomes |
|---|
 |
Chemical availability
Plant roots may exude 20–40% of the sugars and organic acids—photosynthetically fixed carbon.[11] Plant root exudates, such as organic acids, change the chemical structure and the biological communities of the rhizosphere in comparison with the bulk soil or parent soil. Concentrations of organic acids and saccharides affect the ability of the biological communities to shuttle phosphorus, nitrogen,[12][13] potassium, and water to the root cap,[4] and the total availability of iron to the plant and to its neighbors.[14] The ability of the plant's root and its associated soil microorganisms to provide specific transport proteins affects the availability of iron and other minerals for it and its neighbors. This can affect the composition of the community and its fitness.
Root exudates come in the form of chemicals released into the rhizosphere by cells in the roots and cell waste referred to as "rhizodeposition." This rhizodeposition comes in various forms of organic carbon and nitrogen that provide for the communities around plant roots and dramatically affect the chemistry surrounding the roots.
Allelochemicals
Although it goes beyond the rhizosphere area, it is notable that some plants secrete
Ecology of the rhizosphere

Rhizodeposition allows for the growth of communities of microorganisms directly surrounding and inside plant roots. This leads to complex interactions between species, including mutualism, predation/parasitism, and competition.
Predation
Predation is considered to be top-down because these interactions decrease the population, but the closeness of the interactions of species directly affects the availability of resources causing the population to also be affected by bottom-up controls.[24] Without soil fauna, microbes that directly prey upon competitors of plants and plant mutualists, interactions within the rhizosphere would be antagonistic toward the plants. Soil fauna provide the top-down component of the rhizosphere while also allowing for the bottom-up increase in nutrients from rhizodeposition and inorganic nitrogen. The complexity of these interactions has also been shown through experiments with common soil fauna, such as nematodes and protists. Predation by bacterial-feeding nematodes was shown to influence nitrogen availability and plant growth.[25] There was also an increase in the populations of bacteria to which nematodes were added. Predation upon Pseudomonas by amoeba shows predators are able to upregulate toxins produced by prey without direct interaction using supernatant.[26] The ability of predators to control the expression and production of biocontrol agents in prey without direct contact is related to the evolution of prey species to signals of high predator density and nutrient availability.
The food web in the rhizosphere can be considered as three different channels with two different sources of energy: the detritus-dependent channels are fungi and bacterial species, and the root energy-dependent channel consists of nematodes, symbiotic species, and some arthropods.[24] This food web is constantly in flux since the amount of detritus available and the rate of root sloughing changes as roots grow and age. This bacterial channel is considered to be a faster channel because of the ability of species to focus on more accessible resources in the rhizosphere and have faster regeneration times compared with the fungal channel. All three of these channels are also interrelated to the roots that form the base of the rhizosphere ecosystem and the predators, such as the nematodes and protists, that prey upon many of the same species of microflora.
Competition
The competition between plants due to released exudates is dependent upon geometrical properties, which determine the capacity of interception of exudates from any point in relation to the plants’ roots, physicochemical properties, which determine the capacity of each root to take up exudates in the area.[27] Geometrical properties are the density of roots, root diameter, and distribution of the roots. Physicochemical properties are exudation rate, decay rate of exudates, and the properties of the environment that affect diffusion. These properties define the rhizosphere of roots and the likelihood that plants can directly compete with neighbors.
Plants and soil microflora indirectly compete against one another by tying up limiting resources, such as carbon and nitrogen, into their biomass.[28] This competition can occur at varying rates due to the ratio of carbon to nitrogen in detritus and the ongoing mineralization of nitrogen in the soil. Mycorrhizae and heterotrophic soil microorganisms compete for both carbon and nitrogen, depending upon which is limiting at the time, which itself heavily depends on the species, scavenging abilities, and the environmental conditions affecting nitrogen input. Plants are less successful at the uptake of organic nitrogen, such as amino acids, than the soil microflora that exist in the rhizosphere.[29] This informs other mutualistic relationships formed by plants in relation to nitrogen uptake.
Competition over other resources, such as oxygen in limited environments, is directly affected by the spatial and temporal locations of species and the rhizosphere. In methanotrophs, proximity to higher density roots and the surface are important and help to determine where they are dominant over heterotrophs in rice paddies.[30]
The weak connection between the various channels of energy is important in the regulation of the populations of both predator and prey and the availability of resources to the biome. Strong connections between resource-consumer and consumer-consumer create coupled systems of oscillators, which are then determined by the nature of the available resources.[31] These systems can then be considered cyclical, quasi-periodic, or chaotic.
Mutualism
Plants
Symbiotic
In exchange for the resources and shelter that plants and roots provide, fungi and bacteria control pathogenic microbes.[36] The fungi that perform such activities also serve close relationships with species of plants in the form of mycorrhizal fungi, which are diverse in how they relate to plants. Arbuscular mycorrhizal fungi and the bacteria that make the rhizosphere their home also form close relationships in order to be more competitive.[37] which plays into the bigger cycles of nutrients that impact the ecosystem, such as biogeochemical pathways.[14]
Community structure
The rhizosphere has been referred to as an information super highway because of the proximity of data points, which include roots and organisms in the soil, and the methods for transferring data using exudates and communities.
Microbial consortium

Although various studies have shown that single microorganisms can exert beneficial effects on plants, it is increasingly evident that when a
The diagram on the right illustrates that rhizosphere microorganisms like plant-growth-promoting bacteria (PGPB),


Communication

Communication is often the basis of
Prokaryotes and eukaryotes have interacted for millions of years, evolving and refining over time their communication systems. As proposed by Hauser in 1996,
In a particular environment, individuals can communicate and interact with multiple partners, and the nature of interaction can determine variable costs and benefits to the partner, as a biological market.[45] A large number of signals can be exchanged involving the plant itself, insects, fungi, and microbes. This all takes place in a high-density environmental niche. Usually, communication is the result of chemical responses of cells to signatory molecules coming from other cells. These signals affect both the metabolism and transcription of genes activating several regulatory mechanisms.[41]

Frequently in the rhizosphere, more than two organisms (and species) can take part in the communication, resulting in a complex network of interactions and cross-talks which influence the fitness of all participating partners. Thus, this environment can be considered a hot spot for numerous inter-kingdom signals exchange, which involves plant-associated microbial communities (rhizobiome). The microbial community's composition is mainly shaped and recruited by hundreds of
It stands to reason that the plants play a fundamental role in the rhizosphere scene.[48] Indeed, because of the chemical signals conveyed by nutrient-rich exudates released by the plant roots, a large variety of microbes can first colonize the rhizosphere and then gradually penetrate the root and the overall plant tissue (endophytes).[49] Otherwise, they can colonize the host plant establishing a lasting and beneficial symbiotic relationship.[50] To date, numerous investigations on root exudates composition have been performed.[46][51][52][41]
The most known plant-microbe dialogue on the rhizosphere scene, which determines direct and indirect advantages to the partners, was properly addressed as early as 1904 when Hiltner described the symbiotic interaction among legumes and rhizobia.[10] This symbiosis is a highly specific process in which the genetic and chemical communication signals are strictly plant-bacterium specific. In this mutualistic interaction, rhizobia positively influence the host's growth thanks to the nitrogen fixation process and at the same time can benefit from the nutrients provided by the plant.[41]
This symbiosis has been extensively studied in recent decades, and many studies on the communication and the signaling between the two partners at different steps of the symbiosis (from root infection to nodule development) have been elucidated.[53][54] However, the knowledge about the earlier steps of rhizosphere colonization, namely the opening line at the root surface, remains poorly characterized. Nonetheless, increasing data have shown the importance of intraspecies and multispecies communications among rhizospheric biotic components for the improvement of rhizobia–legumes interaction. In addition, it has been shown that rhizobia are part of the rhizosphere of a wide variety of non-legume plants where they can play a role as plant growth promoting components, recovering a central role in plant core microbiome.[55][41]
Methods
The following are some methods commonly used or of interest in rhizosphere research. Many of these methods include both field testing of the root systems and in lab testing using simulated environments to perform experiments, such as pH determination.[56]
- High-throughput screening
- High-Throughput Sequencing: 16S rRNA Amplicon, Metagenomics, Metatranscriptomics
- Culture Depend Approaches
- Root Imaging
- Isotopic labeling
- Enzyme Assays
- Mini rhizotron camera
- Various methods used to determine water movement in the rhizosphere e.g. microelectrodes and agar techniques for pH and microsampling of rhizosphere materials[56]
- Pyrolysis–field ionization mass spectrometry allows for spectrometry of agricultural fields to find fulvic and humic acids and the extraction residues (humins) in certain studies and expanded to general organic compounds in other recent work.[57][19]

See also
- Plant to plant communication via mycorrhizal networks
- Soil biomantle
- Soil respiration
- Rhizobacteria
- Root mucilage
References
- ^ PMID 33841353..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ "Microbial Health of the Rhizosphere". Archived from the original on March 12, 2007. Retrieved 5 May 2006.
- – via Research Gate.
- ^ PMID 12746510.
- ^ Ingham, Elaine R. "The Soil Food Web". USDA-NRCS. Retrieved 3 July 2006.
- ^ .
- S2CID 35099987.
- ^ a b McNear Jr., David H. (2013). "The Rhizosphere - roots, soil and everything in between". Nature Education. 4 (3): 1.
- ^ a b Hiltner, L. (1904) "Ueber neuere Erfahrungen und Probleme auf dem Gebiete derBodenbakteriologie und unter besonderer BerUcksichtigung der Grundungung und Brache. Arb Deut Landw Gesell, 98: 57-78.
- ^ S2CID 4419735.
- ^ Canarini A, Kaiser C, Merchant A, Richter A and Wanek W (2019) Root Exudation of Primary Metabolites: Mechanisms and Their Roles in Plant Responses to Environmental Stimuli. Frontiers in Plant Science 10:157. doi: 10.3389/fpls.2019.00157
- S2CID 26813067.
- S2CID 8562338.
- ^ S2CID 6840457.
- PMID 16219069.
- S2CID 96925936.
- .
- ISSN 2452-2198.
- ^ PMID 18618895.
- PMID 16623597.
- S2CID 219905513..
 Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Modified material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - S2CID 42795586.
- S2CID 131260721.
- ^ .
- JSTOR 1942528.
- PMID 20525866.
- S2CID 23413577.
- PMID 21238010.
- .
- PMID 11472935.
- S2CID 4420271.
- PMID 16787107.
- PMID 16822096.
- PMID 22493242.
- .
- ^ .
- S2CID 39961232.
- PMID 14729216.
- PMID 30812698.
- ^ doi:10.3390/agronomy11020219..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ .
- OCLC 54460090.
- S2CID 5014169.
- ^ Zahavi, Amotz (2008). "The handicap principle and signalling in collaborative systems". In Hughes, David P.; d'Ettorre, Patrizia (eds.). Sociobiology of Communication. Oxford University Press. pp. 1–10.
- PMID 26041807.
- ^ S2CID 17890501.
- PMID 27393937.
- ISBN 9780123948083.
- PMID 18789693.
- PMID 16269768.
- PMID 23373698.
- PMID 26832945.
- PMID 21838550.
- S2CID 7849557.
- PMID 26032777.
- ^ S2CID 24345065.
- S2CID 23547420.
- ISBN 978-3-540-22220-0.
Further reading
- "The Soil Habitat". University of Western Australia. Archived from the original on 20 August 2006. Retrieved 3 July 2006.
- Andrew Wylie (2006). "Digging in the Dirt: Is the Study of the Rhizosphere Ripe for a Systems Biology Approach?". Science Creative Quarterly. Retrieved 24 April 2017.
- McNear Jr., D. H. (2013). "The Rhizosphere – Roots, Soil and Everything In Between". Nature Education Knowledge. 4 (3): 1. Retrieved 24 April 2017.
