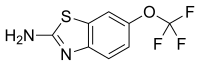
Riluzole
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Rilutek, Tiglutik, Exservan, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a696013 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Elimination half-life | 9–15 hours[1] |
| Excretion | Urine (90%)[1] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Riluzole is a
Medical use
Amyotrophic lateral sclerosis
Riluzole was approved in the United States for the treatment of ALS by the U.S. Food and Drug Administration (FDA) in 1995.[3] A Cochrane Library review states a 9% gain in the probability of surviving one year.[2]
Adverse effects
- Very common (>10% frequency):lung function
- Common (1–10% frequency):aminotransferases
- Uncommon (0.1-1% frequency):[5] pancreatitis; interstitial lung disease
- Rare (<0.1% frequency):angiooedema, anaphylactoid reaction)
Overdose
Symptoms of overdose include: neurological and psychiatric symptoms, acute toxic encephalopathy with stupor, coma and methemoglobinemia.[1] Severe methemoglobinemia may be rapidly reversible after treatment with methylene blue.[1]
Contraindications
Contraindications for riluzole include: known prior hypersensitivity to riluzole or any of the excipients inside the preparations, liver disease, pregnancy or lactation.[1]
Interactions
CYP1A2 substrates, inhibitors and inducers would probably interact with riluzole, due its dependency on this cytochrome for metabolism.[1]
Mechanism of action
Riluzole preferentially blocks TTX-sensitive sodium channels, which are associated with damaged neurons.[6][7] Riluzole has also been reported to directly inhibit the kainate and NMDA receptors.[8] The drug has also been shown to postsynaptically potentiate GABAA receptors via an allosteric binding site.[9] However, the action of riluzole on glutamate receptors has been controversial, as no binding of the drug to any known sites has been shown for them.[10][11] In addition, as its antiglutamatergic action is still detectable in the presence of sodium channel blockers, it is also uncertain whether or not it acts via this way. Rather, its ability to stimulate glutamate uptake seems to mediate many of its effects.[12][13] In addition to its role in accelerating glutamate clearance from the synapse, riluzole may also prevent glutamate release from presynaptic terminals.[14] Since CK1δ plays a key role in TDP-43 proteinopathy, a pathological hallmark of ALS, this could help to better decipher drug mechanism of action.
Synthesis
Riluzole can be prepared beginning with the reaction of 4-(trifluoromethoxy)aniline with potassium thiocyanate followed by reaction with bromine, forming the thiazole ring.[15][16][17]
Society and culture
Legal status
Riluzole was approved for medical use in the European Union in October 1996.[18]
Research
A number of case studies and randomized controlled trials have indicated that riluzole, which is neuroprotective and a glutamate modulator, may have use in mood and anxiety disorders.[19][20][21] However, it failed in trials of Huntington's disease and Parkinson's disease.[21]
Riluzole has been investigated in rodent models for its potential ability to protect against
A
References
- ^ a b c d e f g h i "PRODUCT INFORMATION RILUTEK® (riluzole) Tablets" (PDF). TGA eBusiness Services. sanofi-aventis australia pty ltd. January 6, 2009. Retrieved February 18, 2014.
- ^ PMID 22419278.
- PMID 31644225.
- ^ "Rilutek (riluzole) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved February 18, 2014.
- ^ ISBN 978-0-9805790-9-3.
- PMID 9262334.
- PMID 20236142.
- PMID 7685290.
- S2CID 24194421.
- S2CID 208788906.
- S2CID 5887788.
- S2CID 23849619.
- PMID 12629173.
- S2CID 35667296.
- ^ Yagupol'skii LM, Gandel'sman LZ (1963). "Missing". Zh. Obshch. Khim. 33: 2301.
- ^ EP 50551, Mizoule J, "Medicament containing 2-amino-6-trifluoro-methoxy benzothiazole", published 1982-04-28, assigned to Pharmindustrie; US 4370338, Mizoule J, issued 1983, assigned to Pharmindustrie
- ^ U.S. patent 4,826,860
- ^ "Rilutek EPAR". European Medicines Agency (EMA). Retrieved October 1, 2020.
- PMID 37463744.
- PMID 20807069.
- ^ PMID 18721116.
- PMID 31360245.
- S2CID 29393000.
- ^ "BHV 0223 – AdisInsight". Adisinsight.springer.com. Retrieved May 20, 2016.
- ISSN 2041-5990.
- ^ Terry M (August 16, 2018). "Biohaven Reports Positive Early Clinical Trial Results for Anxiety Drug". BioSpace. Retrieved November 5, 2023.
- PMID 36368186.
- PMID 35780243.

