Rimexolone
 | |
| Clinical data | |
|---|---|
| Trade names | Vexol |
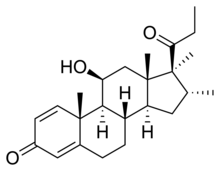
| Other names | Trimexolone; Org 6216; 11β-Hydroxy-16α,17α,21-trimethylpregna-1,4-dien-3,20-dione |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a606003 |
| Routes of administration | Eye drops |
| ATC code | |
| Pharmacokinetic data | |
| Elimination half-life | estimated 1–2 hours |
| Excretion | >80% faeces |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Rimexolone is a glucocorticoid steroid used to treat inflammation in the eye.[1] It is marketed as a 1% eye drop suspension under the trade name Vexol by Alcon Laboratories, but was discontinued in the US and other countries.[2][3]
Medical uses
Rimexolone is used to treat inflammation after
Contraindications
The substance is contraindicated in
because it only reduces the inflammation but does not act against such microorganisms.Side effects
The most common adverse effects are blurred vision,
oedema, headache, increased intraocular pressure and other side effects are seen in less than 1% of patients.[2][3]
Pharmacology
Pharmacodynamics
As a glucocorticoid, rimexolone acts as an agonist of the glucocorticoid receptor.
Pharmacokinetics
A small amount of rimexolone is absorbed into the systemic circulation. On hourly treatment with the eye drops for a week,
elimination half-life from the circulation is estimated at one to two hours; the substance is mainly (over 80%) excreted via the faeces.[2][3]
