River ecosystem

River ecosystems are flowing waters that drain the landscape, and include the
The food base of streams within riparian forests is mostly derived from the trees, but wider streams and those that lack a
River ecosystems are prime examples of lotic ecosystems. Lotic refers to flowing water, from the
The following unifying characteristics make the ecology of running waters unique among aquatic habitats: the flow is unidirectional, there is a state of continuous physical change, and there is a high degree of spatial and temporal heterogeneity at all scales (
Abiotic components (non-living)
The non-living components of an ecosystem are called abiotic components. E.g. stone, air, soil, etc.
Water flow

Unidirectional water flow is the key factor in lotic systems influencing their ecology. Streamflow can be continuous or intermittent, though. Streamflow is the result of the summative inputs from groundwater, precipitation, and overland flow. Water flow can vary between systems, ranging from torrential rapids to slow backwaters that almost seem like lentic systems. The speed or velocity of the water flow of the water column can also vary within a system and is subject to chaotic turbulence, though water velocity tends to be highest in the middle part of the stream channel (known as the thalveg). This turbulence results in divergences of flow from the mean downslope flow vector as typified by eddy currents. The mean flow rate vector is based on the variability of friction with the bottom or sides of the channel, sinuosity, obstructions, and the incline gradient.[5] In addition, the amount of water input into the system from direct precipitation, snowmelt, and/or groundwater can affect the flow rate. The amount of water in a stream is measured as discharge (volume per unit time). As water flows downstream, streams and rivers most often gain water volume, so at base flow (i.e., no storm input), smaller headwater streams have very low discharge, while larger rivers have much higher discharge. The "flow regime" of a river or stream includes the general patterns of discharge over annual or decadal time scales, and may capture seasonal changes in flow.[7][8]
While water flow is strongly determined by slope, flowing waters can alter the general shape or direction of the stream bed, a characteristic also known as
Light
Light is important to lotic systems, because it provides the energy necessary to drive
Temperature


Most lotic species are
Chemistry
Water chemistry in river ecosystems varies depending on which dissolved solutes and gases are present in the water column of the stream. Specifically river water can include, apart from the water itself,[citation needed]
- dissolved inorganic matter and major ions (calcium, sodium, magnesium, potassium, bicarbonate, sulphide, chloride)
- dissolved inorganic nutrients (nitrogen, phosphorus, silica)
- dissolved organic matter
- gases (nitrogen, nitrous oxide, carbon dioxide, oxygen)
- trace metals and pollutants
Dissolved ions and nutrients
Dissolved stream solutes can be considered either reactive or conservative. Reactive solutes are readily biologically assimilated by the
Dissolved gases
In terms of dissolved gases, oxygen is likely the most important chemical constituent of lotic systems, as all aerobic organisms require it for survival. It enters the water mostly via diffusion at the water-air interface. Oxygen's solubility in water decreases as water pH and temperature increases. Fast, turbulent streams expose more of the water's surface area to the air and tend to have low temperatures and thus more oxygen than slow, backwaters.[6] Oxygen is a byproduct of photosynthesis, so systems with a high abundance of aquatic algae and plants may also have high concentrations of oxygen during the day. These levels can decrease significantly during the night when primary producers switch to respiration. Oxygen can be limiting if circulation between the surface and deeper layers is poor, if the activity of lotic animals is very high, or if there is a large amount of organic decay occurring.[10]
Suspended matter
Rivers can also transport suspended inorganic and organic matter. These materials can include sediment[12] or terrestrially-derived organic matter that falls into the stream channel.[13] Often, organic matter is processed within the stream via mechanical fragmentation, consumption and grazing by invertebrates, and microbial decomposition.[14] Leaves and woody debris recognizable coarse particulate organic matter (CPOM) into particulate organic matter (POM), down to fine particulate organic matter. Woody and non-woody plants have different instream breakdown rates, with leafy plants or plant parts (e.g., flower petals) breaking down faster than woody logs or branches.[15]
Substrate
The inorganic
Biotic components (living)
The living components of an ecosystem are called the biotic components. Streams have numerous types of biotic organisms that live in them, including bacteria, primary producers, insects and other invertebrates, as well as fish and other vertebrates.
Biofilm
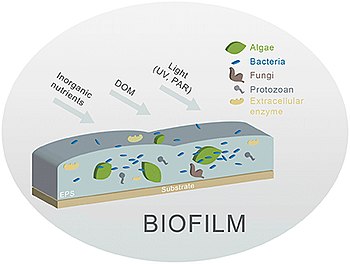
A
and add to the complexity of a streambed.The different biofilm components (algae and bacteria are the principal components) are embedded in an
Biofilms are one of the main

Microorganisms
Diatoms are one of the main dominant groups of periphytic algae in lotic systems and have been widely used as efficient indicators of water quality, because they respond quickly to environmental changes, especially organic pollution and eutrophication, with a broad spectrum of tolerances to conditions ranging, from oligotrophic to eutrophic.[19][23][24]
Primary producers
Algae, consisting of phytoplankton and periphyton, are the most significant sources of primary production in most streams and rivers.[6] Phytoplankton float freely in the water column and thus are unable to maintain populations in fast flowing streams. They can, however, develop sizeable populations in slow moving rivers and backwaters.[5] Periphyton are typically filamentous and tufted algae that can attach themselves to objects to avoid being washed away by fast currents. In places where flow rates are negligible or absent, periphyton may form a gelatinous, unanchored floating mat.[10]


Plants exhibit limited adaptations to fast flow and are most successful in reduced currents. More primitive plants, such as
Living in flowing water can be beneficial to plants and algae because the current is usually well aerated and it provides a continuous supply of nutrients.[10] These organisms are limited by flow, light, water chemistry, substrate, and grazing pressure.[6] Algae and plants are important to lotic systems as sources of energy, for forming microhabitats that shelter other fauna from predators and the current, and as a food resource.[11]
Insects and other invertebrates
Up to 90% of invertebrates in some lotic systems are insects. These species exhibit tremendous diversity and can be found occupying almost every available habitat, including the surfaces of stones, deep below the substratum in the hyporheic zone, adrift in the current, and in the surface film.[citation needed]
Insects have developed several strategies for living in the diverse flows of lotic systems. Some avoid high current areas, inhabiting the substratum or the sheltered side of rocks. Others have flat bodies to reduce the drag forces they experience from living in running water.[25] Some insects, like the giant water bug (Belostomatidae), avoid flood events by leaving the stream when they sense rainfall.[26] In addition to these behaviors and body shapes, insects have different life history adaptations to cope with the naturally-occurring physical harshness of stream environments.[27] Some insects time their life events based on when floods and droughts occur. For example, some mayflies synchronize when they emerge as flying adults with when snowmelt flooding usually occurs in Colorado streams. Other insects do not have a flying stage and spend their entire life cycle in the river.
Like most of the primary consumers, lotic invertebrates often rely heavily on the current to bring them food and oxygen.[28] Invertebrates are important as both consumers and prey items in lotic systems.[citation needed]
The common orders of insects that are found in river ecosystems include
Additional invertebrate
Fish and other vertebrates


Fish are probably the best-known inhabitants of lotic systems. The ability of a fish species to live in flowing waters depends upon the speed at which it can swim and the duration that its speed can be maintained. This ability can vary greatly between species and is tied to the habitat in which it can survive. Continuous swimming expends a tremendous amount of energy and, therefore, fishes spend only short periods in full current. Instead, individuals remain close to the bottom or the banks, behind obstacles, and sheltered from the current, swimming in the current only to feed or change locations.[1] Some species have adapted to living only on the system bottom, never venturing into the open water flow. These fishes are dorso-ventrally flattened to reduce flow resistance and often have eyes on top of their heads to observe what is happening above them. Some also have sensory barrels positioned under the head to assist in the testing of substratum.[11]
Lotic systems typically connect to each other, forming a path to the ocean (spring → stream → river → ocean), and many fishes have life cycles that require stages in both fresh and salt water.
Other vertebrate taxa that inhabit lotic systems include amphibians, such as salamanders, reptiles (e.g. snakes, turtles, crocodiles and alligators) various bird species, and mammals (e.g., otters, beavers, hippos, and river dolphins). With the exception of a few species, these vertebrates are not tied to water as fishes are, and spend part of their time in terrestrial habitats.[6] Many fish species are important as consumers and as prey species to the larger vertebrates mentioned above.[citation needed]
Trophic level dynamics
The concept of trophic levels are used in food webs to visualise the manner in which energy is transferred from one part of an ecosystem to another.[29] Trophic levels can be assigned numbers determining how far an organism is along the food chain.
- Level one: Producers, plant-like organisms that generate their own food using solar radiation, including algae, phytoplankton, mosses and lichens.
- Level two: Consumers, animal-like organism that get their energy from eating producers, such as zooplankton, small fish, and crustaceans.
- Level three: fungi.
All energy transactions within an
Top-down and bottom-up affect

A common issue with trophic level dynamics is how resources and production are regulated.[30] The usage and interaction between resources have a large impact on the structure of food webs as a whole. Temperature plays a role in food web interactions including top-down and bottom-up forces within ecological communities. Bottom-up regulations within a food web occur when a resource available at the base or bottom of the food web increases productivity, which then climbs the chain and influence the biomass availability to higher trophic organism.[30] Top-down regulations occur when a predator population increases. This limits the available prey population, which limits the availability of energy for lower trophic levels within the food chain. Many biotic and abiotic factors can influence top-down and bottom-up interactions.[31]
Trophic cascade
Another example of food web interactions are
Food chain
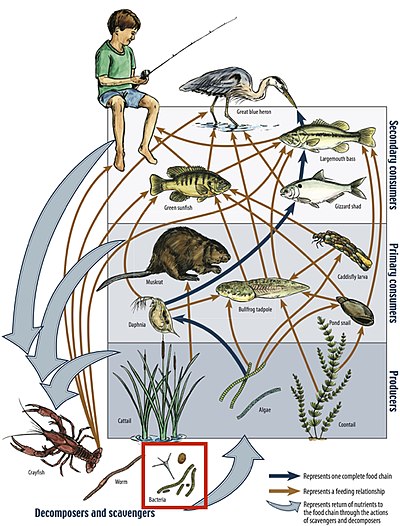
A food chain is a linear system of links that is part of a food web, and represents the order in which organisms are consumed from one trophic level to the next. Each link in a food chain is associated with a trophic level in the ecosystem. The numbered steps it takes for the initial source of energy starting from the bottom to reach the top of the food web is called the food chain length.[33] While food chain lengths can fluctuate, aquatic ecosystems start with primary producers that are consumed by primary consumers which are consumed by secondary consumers, and those in turn can be consumed by tertiary consumers so on and so forth until the top of the food chain has been reached.[citation needed]
Primary producers
Primary consumers
Secondary consumers
The
Food web complexity
Trophic relationships
Energy inputs

Energy sources can be
- Autochthonous (from the Latin "auto" = "self) energy sources are those derived from within the lotic system. During This process is discussed in more detail below.

- Allochthonous energy sources are those derived from outside the lotic system, that is, from the terrestrial environment. Leaves, twigs, fruits, etc. are typical forms of terrestrial CPOM that have entered the water by direct litter fall or lateral leaf blow.colonize the leaf, softening it as the mycelium of the fungus grows into it. The composition of the microbial community is influenced by the species of tree from which the leaves are shed (Rubbo and Kiesecker 2004). This combination of bacteria, fungi, and leaf are a food source for shredding invertebrates,[41] which leave only FPOM after consumption. These fine particles may be colonized by microbes again or serve as a food source for animals that consume FPOM. Organic matter can also enter the lotic system already in the FPOM stage by wind, surface runoff, bank erosion, or groundwater. Similarly, DOM can be introduced through canopy drip from rain or from surface flows.[6]
Invertebrates
Fish
Fish can also be placed into
Community patterns and diversity
Local species richness
Large rivers have comparatively more species than small streams. Many relate this pattern to the greater area and volume of larger systems, as well as an increase in habitat diversity. Some systems, however, show a poor fit between system size and
Resource partitioning
Although many alternate theories have been postulated for the ability of
Dietary segregation is the second-most common type of resource partitioning.
Persistence and succession
Over long time scales, there is a tendency for species composition in pristine systems to remain in a stable state.[42] This has been found for both invertebrate and fish species.[6] On shorter time scales, however, flow variability and unusual precipitation patterns decrease habitat stability and can all lead to declines in persistence levels. The ability to maintain this persistence over long time scales is related to the ability of lotic systems to return to the original community configuration relatively quickly after a disturbance (Townsend et al. 1987). This is one example of temporal succession, a site-specific change in a community involving changes in species composition over time. Another form of temporal succession might occur when a new habitat is opened up for colonization. In these cases, an entirely new community that is well adapted to the conditions found in this new area can establish itself.[6]
River continuum concept
The
According to the RCC, low ordered sites are small shaded streams where allochthonous inputs of CPOM are a necessary resource for consumers. As the river widens at mid-ordered sites, energy inputs should change. Ample sunlight should reach the bottom in these systems to support significant periphyton production. Additionally, the biological processing of CPOM (coarse particulate organic matter – larger than 1 mm) inputs at upstream sites is expected to result in the transport of large amounts of FPOM (fine particulate organic matter – smaller than 1 mm) to these downstream ecosystems. Plants should become more abundant at edges of the river with increasing river size, especially in lowland rivers where finer sediments have been deposited and facilitate rooting. The main channels likely have too much current and turbidity and a lack of substrate to support plants or periphyton. Phytoplankton should produce the only autochthonous inputs here, but photosynthetic rates will be limited due to turbidity and mixing. Thus, allochthonous inputs are expected to be the primary energy source for large rivers. This FPOM will come from both upstream sites via the decomposition process and through lateral inputs from floodplains.
Biota should change with this change in energy from the headwaters to the mouth of these systems. Namely, shredders should prosper in low-ordered systems and grazers in mid-ordered sites. Microbial decomposition should play the largest role in energy production for low-ordered sites and large rivers, while photosynthesis, in addition to degraded allochthonous inputs from upstream will be essential in mid-ordered systems. As mid-ordered sites will theoretically receive the largest variety of energy inputs, they might be expected to host the most biological diversity (Vannote et al. 1980).[5][6]
Just how well the RCC actually reflects patterns in natural systems is uncertain and its generality can be a handicap when applied to diverse and specific situations.
Disturbances such as congestion by dams or natural events such as shore flooding are not included in the RCC model.[44] Various researchers have since expanded the model to account for such irregularities. For example, J.V. Ward and J.A. Stanford came up with the Serial Discontinuity Concept in 1983, which addresses the impact of geomorphologic disorders such as congestion and integrated inflows. The same authors presented the Hyporheic Corridor concept in 1993, in which the vertical (in depth) and lateral (from shore to shore) structural complexity of the river were connected.[45] The flood pulse concept, developed by W. J. Junk in 1989, further modified by P. B. Bayley in 1990 and K. Tockner in 2000, takes into account the large amount of nutrients and organic material that makes its way into a river from the sediment of surrounding flooded land.[44]
Human impacts
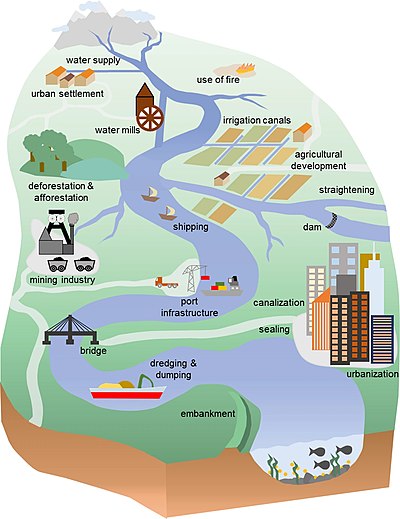
Humans exert a
However, a detectable human imprint on the environment extends back for thousands of years,
Pollution
River pollution can include but is not limited to: increasing sediment export, excess nutrients from fertilizer or urban runoff,
Pollutant sources of lotic systems are hard to control because they can derive, often in small amounts, over a very wide area and enter the system at many locations along its length. While direct pollution of lotic systems has been greatly reduced in the United States under the government's Clean Water Act, contaminants from diffuse non-point sources remain a large problem.[10] Agricultural fields often deliver large quantities of sediments, nutrients, and chemicals to nearby streams and rivers. Urban and residential areas can also add to this pollution when contaminants are accumulated on impervious surfaces such as roads and parking lots that then drain into the system. Elevated nutrient concentrations, especially nitrogen and phosphorus which are key components of fertilizers, can increase periphyton growth, which can be particularly dangerous in slow-moving streams.[10] Another pollutant, acid rain, forms from sulfur dioxide and nitrous oxide emitted from factories and power stations. These substances readily dissolve in atmospheric moisture and enter lotic systems through precipitation. This can lower the pH of these sites, affecting all trophic levels from algae to vertebrates.[11] Mean species richness and total species numbers within a system decrease with decreasing pH.[6]
Flow modification

Flow modification can occur as a result of dams, water regulation and extraction, channel modification, and the destruction of the river floodplain and adjacent riparian zones.[76]
Invasive species
See also
- Betty's Brain – software that "learns" about river ecosystems
- Flood pulse concept
- Lake ecosystem
- Rheophile
- Riparian zone
- River continuum concept
- River drainage system
- RIVPACS
- The Riverkeepers
- Upland and lowland rivers
References
- ^ a b c Angelier, E. 2003. Ecology of Streams and Rivers. Science Publishers, Inc., Enfield. Pp. 215.
- ^ "Biology Concepts & Connections Sixth Edition", Campbell, Neil A. (2009), page 2, 3 and G-9. Retrieved 2010-06-14.
- ISBN 0-412-74050-8.
- ISBN 978-0-521-51940-3.
- ^ a b c d e f g h i j k l m n o p Allan, J.D. 1995. Stream Ecology: structure and function of running waters. Chapman and Hall, London. Pp. 388.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z Giller, S. and B. Malmqvist. 1998. The Biology of Streams and Rivers. Oxford University Press, Oxford. Pp. 296.
- doi:10.1139/f89-228.
- JSTOR 1313099.
- ^ "Fluvial Processes in Geomorphology". store.doverpublications.com. Retrieved 2018-11-26.
- ^ a b c d e f g h i j k l m n o p q r Cushing, C.E. and J.D. Allan. 2001. Streams: their ecology and life. Academic Press, San Diego. Pp. 366.
- ^ a b c d e Brown 1987
- ISSN 2169-9003.
- S2CID 88167408.
- JSTOR 3546505. Retrieved 2018-11-27.
- ISSN 0066-4162.
- ^ S2CID 18984955..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - S2CID 6573778.
- PMID 22237539.
- ^ doi:10.1590/2175-7860201970041..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - S2CID 3473853.
- S2CID 28850938.
- .
- ISBN 978-3-319-31983-4.
- .
- S2CID 18882022.
- ^
Lytle, David A. (1999). "Use of Rainfall Cues by Abedus herberti (Hemiptera: Belostomatidae): A Mechanism for Avoiding Flash Floods" (PDF). Journal of Insect Behavior. 12 (1): 1–12. S2CID 25696846. Retrieved 26 November 2018.
- ^
Lytle, David A.; Poff, N. LeRoy (2004). "Adaptation to natural flow regimes". Trends in Ecology and Evolution. 19 (2): 94–100. PMID 16701235.
- JSTOR 2096788.
- ^ JSTOR 1930126.
- ^ PMID 21058543.
- ^ PMID 27663836.
- S2CID 3344238..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - .
- hdl:1808/8160.
- ^ .
- ^ PMID 15012327.
- ^ S2CID 24780727.
- ^ S2CID 2114852.
- ^ ISSN 1559-2731.
- S2CID 54609464.
- ^ U.S. EPA. "Classifications of Macroinvertebrates". Retrieved 3 July 2012.
- ^ Hildrew, A. G. and P. S. Giller. 1994. Patchiness, species interactions and disturbance in the stream benthos. In Aquatic Ecology: scale pattern and process. P. S. Giller, A. G. Hildrew, and D. G. Rafaelli (eds.). Blackwell, Oxford. Pp. 21–62.
- S2CID 40373623– via Canadian Science Publishing.
- ^ a b Junk J. W., P. B. Bayley, R. E. Sparks: "The flood pulse concept in river flood plain systems". Canadian Special Publications of Fisheries and Aquatic Sciences. 106. 1989.
- ^ Ward J. V., J. A. Stanford: The Serial Discontinuity Concept of River Ecosystems. T. D. Fontaine, S. M. Bartell: "Dynamics of Lotic Ecosystems". Science Publications, Ann Arbor Mich, 29–42. 1983.
- ISSN 2190-4707..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ISSN 0091-7613.
- doi:10.1130/G21108.1.
- S2CID 16218015.
- S2CID 131524600.
- S2CID 206642594.
- ^ S2CID 9743349.
- ^ doi:10.3390/quat1030021..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - .
- S2CID 133825138.
- .
- .
- S2CID 36804466.
- .
- S2CID 2501894.
- S2CID 128818837.
- S2CID 129727301.
- S2CID 131236197.
- .
- S2CID 55224176.
- ^ Wong, C.M., Williams, C.E., Collier, U., Schelle, P. and Pittock, J.(2007) World's top 10 rivers at risk World Wildlife Fund.
- S2CID 4422681.
- ^ Biello, David. "Fertilizer Runoff Overwhelms Streams and Rivers--Creating Vast "Dead Zones"". Scientific American. Retrieved 2018-11-27.
- ^ University, Michigan State. "Septic tanks aren't keeping poo out of rivers and lakes". MSUToday. Retrieved 2018-11-27.
- ^ "Growing Concern over Plastic Pollution in Rivers and Lakes". The Maritime Executive. Retrieved 2018-11-27.
- PMID 11944670.
- S2CID 90620297.
- ^ "U.S. rivers are becoming saltier – and it's not just from treating roads in winter". Retrieved 2018-11-27.
- PMID 27786199.
- ^ "Water Pollution: Everything You Need to Know". NRDC. Retrieved 2018-11-27.
- ^ "Altered water flow (hydrology)". NIWA. 2009-07-06. Retrieved 2018-11-27.
Further reading
- Brown, A. L. 1987. Freshwater Ecology. Heinimann Educational Books, London. P. 163.
- Carlisle, D. M. and M. D. Woodside. 2013. Ecological health in the nation's streams, United States Geological Survey. P. 6.
- Edington, J. M., Edington, M. A., and J. A. Dorman. 1984. Habitat partitioning amongst hydrophyschid larvae of a Malaysian stream. Entomologica 30: 123–129.
- Hynes, H. B. N. 1970. Ecology of Running Waters. Originally published in Toronto by University of Toronto Press, 555 p.
- Morin, P. J. 1999. Community Ecology. Blackwell Science, Oxford. P. 424.
- Power, M. E. (1990). "Effects of fish in river food webs" (PDF). Science. 250 (4982): 811–814. S2CID 24780727. Archived from the original(PDF) on 2013-05-24.
- Rubbo, M. J.; Kiesecker, J. M. (2004). "Leaf litter composition and community structure: translating regional species changes into local dynamics". Ecology. 85 (9): 2519–2525. doi:10.1890/03-0653.
- S2CID 43846597.
- Townsend, C. R.; Hildrew, A. G.; Schofield, K. (1987). "Persistence of stream invertebrate communities in relation to environmental variability". Animal Ecology. 56 (2): 597–613. JSTOR 5071.
- Vannote, R. L.; Minshall, G. W.; Cummins, K. W.; Sedell, J. R.; Cushing, C. E. (1980). "The river continuum concept". Canadian Journal of Fisheries and Aquatic Sciences. 37: 130–137. S2CID 40373623.
- Vinson, M. R.; Hawkins, C. P. (1998). "Biodiversity of stream insects: variation at local, basin, and regional scales". Annual Review of Entomology. 43: 271–293. PMID 15012391.
- Ward, J. V. 1992. Aquatic Insect Ecology: biology and habitat. Wiley, New York. P. 456.
- Watson, D. J.; Balon, E. K. (1984). "Ecomorphological analysis of fish taxocenes in rainforest streams of northern Borneo". Journal of Fish Biology. 25 (3): 371–384. .




