Robinson annulation
| Robinson annulation | |
|---|---|
| Named after | Robert Robinson
|
| Reaction type | Ring forming reaction |
| Identifiers | |
| Organic Chemistry Portal | robinson-annulation |
| RSC ontology ID | RXNO:0000380 |
The Robinson annulation is a
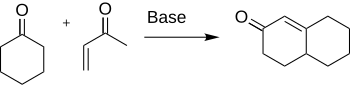
Formation of
The initial paper on the Robinson annulation was published by
Robinson and Rapson found in 1935 that the interaction between cyclohexanone and α,β-unsaturated ketone afforded the desired cyclohexenone. It remains one of the key methods for the construction of six membered ring compounds. Since it is so widely used, there are many aspects of the reaction that have been investigated such as variations of the substrates and reaction conditions as discussed in the scope and variations section.[4] Robert Robinson won the Nobel Prize for Chemistry in 1947 for his contribution to the study of alkaloids.[5]
Reaction mechanism
In the Michael reaction, the
In order to avoid a reaction between the original enolate and the cyclohexenone product, the initial Michael adduct is often isolated first and then cyclized to give the desired octalone in a separate step.[6]
Stereochemistry
Studies have been completed on the formation of the

It has been postulated that the difference in the formation of these transition states and their corresponding products is due to solvent interactions. Scanio found that changing the solvent of the reaction from dioxane to DMSO gives different stereochemistry in step D above. This suggests that the presence of protic or aprotic solvents gives rise to different transition states.[8]
Mechanistic classification
Scope and variations
Reaction conditions
Although the Robinson annulation is generally conducted under basic conditions, reactions have been conducted under a variety of conditions. Heathcock and Ellis report similar results to the base-catalyzed method using sulfuric acid.[2] The Michael reaction can occur under neutral conditions through an enamine. A Mannich base can be heated in the presence of the ketone to produce the Michael adduct.[6] Successful preparation of compounds using the Robinson annulation methods have been reported.[9]
The Michael acceptor
A typical

Wichterle reaction
The Wichterle reaction is a variant of the Robinson annulation that replaces

Hauser annulation
The reaction sequence in the related Hauser annulation is a

Asymmetric Robinson annulation
Asymmetric synthesis of Robinson annulation products most often involve the use of a


Wang, et al. reported the one-pot synthesis of chiral thiochromenes by such an organocatalytic Robinson annulation.[15]
Applications to synthesis
The
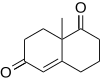
F. Dean Toste and co-workers
Enantioselective route to platensimycin

Scientists at Merck discovered platensimycin, a novel antibiotic lead compound with potential medicinal applications as seen in the adjacent picture.[17]
Initial synthesis gave a racemic form of the compound using an intramolecular etherification reaction of the alcohol motifs and the double bond. Yamamoto and coworkers report the use of an alternative intramolecular Robinson annulation to provide a straightforward enantioselective synthesis of tetracyclic core of platensimycin. The key Robinson annulation step was reported to be accomplished in one pot using L-proline for chiral control. The reaction conditions can be seen below.[18]

References
- ^ .
- ^ .
- .
- ISBN 978-0-471-57022-6.
- ISBN 978-0-495-11258-7.
- ^ .
- .
- .
- doi:10.15227/orgsyn.063.0037. Archived from the original(PDF) on 24 April 2012.
- ISBN 978-0471007593.
- ISBN 978-0-470-63885-9.
- .
- .
- ^ .
- ^ Wang, W.; Li, H.; Wang, J.; Zu, L., J. Am. Chem. Soc. 2006; 128, 10354.
- PMID 17729226.
- S2CID 4329677. Retrieved 9 September 2021.
- PMID 17630748.
