Root

In
Function
The major functions of roots are absorption of water, plant nutrition and anchoring of the plant body to the ground.[2]
Anatomy
Root morphology is divided into four zones: the root cap, the
When dissected, the arrangement of the cells in a root is root hair, epidermis, epiblem, cortex, endodermis, pericycle and, lastly, the vascular tissue in the centre of a root to transport the water absorbed by the root to other places of the plant.[clarification needed]
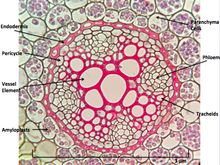
Perhaps the most striking characteristic of roots that distinguishes them from other plant organs such as stem-branches and leaves is that roots have an endogenous[5] origin, i.e., they originate and develop from an inner layer of the mother axis, such as pericycle.[6] In contrast, stem-branches and leaves are exogenous, i.e., they start to develop from the cortex, an outer layer.
In response to the concentration of nutrients, roots also synthesise

Root system architecture (RSA)

Definition
In its simplest form, the term root system architecture (RSA) refers to the spatial configuration of a plant's root system. This system can be extremely complex and is dependent upon multiple factors such as the species of the plant itself, the composition of the soil and the availability of nutrients.[8] Root architecture plays the important role of providing a secure supply of nutrients and water as well as anchorage and support.
The configuration of root systems serves to structurally support the plant, compete with other plants and for uptake of nutrients from the soil.[9] Roots grow to specific conditions, which, if changed, can impede a plant's growth. For example, a root system that has developed in dry soil may not be as efficient in flooded soil, yet plants are able to adapt to other changes in the environment, such as seasonal changes.[9]
Terms and components
The main terms used to classify the architecture of a root system are:[10]
| Branch magnitude | Number of links (exterior or interior) |
| Topology | Pattern of branching ( Radial )
|
| Link length | Distance between branches |
| Root angle | Radial angle of a lateral root's base around the parent root's circumference, the angle of a lateral root from its parent root, and the angle an entire system spreads. |
| Link radius | Diameter of root |
All components of the root architecture are regulated through a complex interaction between genetic responses and responses due to environmental stimuli. These developmental stimuli are categorised as intrinsic, the genetic and nutritional influences, or extrinsic, the environmental influences and are interpreted by
Extrinsic factors affecting root architecture include gravity, light exposure, water and oxygen, as well as the availability or lack of nitrogen, phosphorus, sulphur, aluminium and sodium chloride. The main hormones (intrinsic stimuli) and respective pathways responsible for root architecture development include:
| Auxin | Lateral root formation, maintenance of apical dominance and adventitious root formation. |
Cytokinins |
Cytokinins regulate root apical meristem size and promote lateral root elongation. |
| Ethylene | Promotes crown root formation. |
Gibberellins |
Together with ethylene, they promote crown primordia growth and elongation. Together with auxin, they promote root elongation. Gibberellins also inhibit lateral root primordia initiation. |
Growth

Early root growth is one of the functions of the apical meristem located near the tip of the root. The meristem cells more or less continuously divide, producing more meristem, root cap cells (these are sacrificed to protect the meristem), and undifferentiated root cells. The latter become the primary tissues of the root, first undergoing elongation, a process that pushes the root tip forward in the growing medium. Gradually these cells differentiate and mature into specialized cells of the root tissues.[12]
Growth from apical meristems is known as primary growth, which encompasses all elongation. Secondary growth encompasses all growth in diameter, a major component of
In plants with secondary growth, the vascular cambium, originating between the xylem and the phloem, forms a
At this point, the cork cambium begins to form the periderm, consisting of protective cork cells. The walls of cork cells contains suberin thickenings, which is an extra cellular complex biopolymer.[13] The suberin thickenings functions by providing a physical barrier, protection against pathogens and by preventing water loss from the surrounding tissues. In addition, it also aids the process of wound healing in plants.[14] It is also postulated that suberin could be a component of the apoplastic barrier (present at the outer cell layers of roots) which prevents toxic compounds from entering the root and reduces radial oxygen loss (ROL) from the aerenchyma during waterlogging.[15] In roots, the cork cambium originates in the pericycle, a component of the vascular cylinder.[15]
The vascular cambium produces new layers of secondary xylem annually.[citation needed] The xylem vessels are dead at maturity (in some) but are responsible for most water transport through the vascular tissue in stems and roots.

Tree roots usually grow to three times the diameter of the branch spread, only half of which lie underneath the trunk and canopy. The roots from one side of a tree usually supply nutrients to the foliage on the same side. Some families however, such as Sapindaceae (the maple family), show no correlation between root location and where the root supplies nutrients on the plant.[16]
Regulation
There is a correlation of roots using the process of plant perception to sense their physical environment to grow,[17] including the sensing of light,[18] and physical barriers. Plants also sense gravity and respond through auxin pathways,[19] resulting in gravitropism. Over time, roots can crack foundations, snap water lines, and lift sidewalks. Research has shown that roots have ability to recognize 'self' and 'non-self' roots in same soil environment.[20]
The correct environment of
Gravitropism directs roots to grow downward at germination, the growth mechanism of plants that also causes the shoot to grow upward.[22] Different types of roots such as primary, seminal, lateral and crown are maintained at different gravitropic setpoint angles i.e. the direction in which they grow. Recent research show that root angle in cereal crops such as barley and wheat is regulated by a novel gene called Enhanced Gravitropism 1 (EGT1).[23]
Research indicates that plant roots growing in search of productive nutrition can sense and avoid soil compaction through diffusion of the gas ethylene.[24]

Shade avoidance response
In order to avoid shade, plants utilize a shade avoidance response. When a plant is under dense vegetation, the presence of other vegetation nearby will cause the plant to avoid lateral growth and experience an increase in upward shoot, as well as downward root growth. In order to escape shade, plants adjust their root architecture, most notably by decreasing the length and amount of lateral roots emerging from the primary root. Experimentation of mutant variants of Arabidopsis thaliana found that plants sense the Red to Far Red light ratio that enters the plant through photoreceptors known as phytochromes.[25] Nearby plant leaves will absorb red light and reflect far-red light, which will cause the ratio red to far red light to lower. The phytochrome PhyA that senses this Red to Far Red light ratio is localized in both the root system as well as the shoot system of plants, but through knockout mutant experimentation, it was found that root localized PhyA does not sense the light ratio, whether directly or axially, that leads to changes in the lateral root architecture.[25] Research instead found that shoot localized PhyA is the phytochrome responsible for causing these architectural changes of the lateral root. Research has also found that phytochrome completes these architectural changes through the manipulation of auxin distribution in the root of the plant.[25] When a low enough Red to Far Red ratio is sensed by PhyA, the phyA in the shoot will be mostly in its active form.[26] In this form, PhyA stabilize the transcription factor HY5 causing it to no longer be degraded as it is when phyA is in its inactive form. This stabilized transcription factor is then able to be transported to the roots of the plant through the phloem, where it proceeds to induce its own transcription as a way to amplify its signal. In the roots of the plant HY5 functions to inhibit an auxin response factor known as ARF19, a response factor responsible for the translation of PIN3 and LAX3, two well known auxin transporting proteins.[26] Thus, through manipulation of ARF19, the level and activity of auxin transporters PIN3 and LAX3 is inhibited.[26] Once inhibited, auxin levels will be low in areas where lateral root emergence normally occurs, resulting in a failure for the plant to have the emergence of the lateral root primordium through the root pericycle. With this complex manipulation of Auxin transport in the roots, lateral root emergence will be inhibited in the roots and the root will instead elongate downwards, promoting vertical plant growth in an attempt to avoid shade.[25][26]
Research of Arabidopsis has led to the discovery of how this auxin mediated root response works. In an attempt to discover the role that phytochrome plays in lateral root development, Salisbury et al. (2007) worked with Arabidopsis thaliana grown on agar plates. Salisbury et al. used wild type plants along with varying protein knockout and gene knockout Arabidopsis mutants to observe the results these mutations had on the root architecture, protein presence, and gene expression. To do this, Salisbury et al. used GFP fluorescence along with other forms of both macro and microscopic imagery to observe any changes various mutations caused. From these research, Salisbury et al. were able to theorize that shoot located phytochromes alter auxin levels in roots, controlling lateral root development and overall root architecture.[25] In the experiments of van Gelderen et al. (2018), they wanted to see if and how it is that the shoot of A. thaliana alters and affects root development and root architecture. To do this, they took Arabidopsis plants, grew them in agar gel, and exposed the roots and shoots to separate sources of light. From here, they altered the different wavelengths of light the shoot and root of the plants were receiving and recorded the lateral root density, amount of lateral roots, and the general architecture of the lateral roots. To identify the function of specific photoreceptors, proteins, genes, and hormones, they utilized various Arabidopsis knockout mutants and observed the resulting changes in lateral roots architecture. Through their observations and various experiments, van Gelderen et al. were able to develop a mechanism for how root detection of Red to Far-red light ratios alter lateral root development.[26]
Types
A true root system consists of a primary root and secondary roots (or
- the diffuse root system: the primary root is not dominant; the whole root system is fibrous and branches in all directions. Most common in monocots. The main function of the fibrous root is to anchor the plant.
Specialized








The roots, or parts of roots, of many plant species have become specialized to serve adaptive purposes besides the two primary functions[clarification needed], described in the introduction.
- Adventitious roots arise out-of-sequence from the more usual root formation of branches of a primary root, and instead originate from the stem, branches, leaves, or old woody roots. They commonly occur in ivy (Hedera), strawberry (Fragaria) and willow (Salix). Most aerial roots and stilt roots are adventitious. In some conifers adventitious roots can form the largest part of the root system. Adventitious root formation is enhanced in many plant species during (partial) submergence, to increase gas exchange and storage of gases like oxygen.[27] Distinct types of adventitious roots can be classified and are dependent on morphology, growth dynamics and function.[28][29]
- Aerating roots (or knee root or knee or pneumatophores): roots rising above the ground, especially above water such as in some mangrove genera (Avicennia, Sonneratia). In some plants like Avicennia the erect roots have a large number of breathing pores for exchange of gases.
- Aerial roots: roots entirely above the ground, such as in ivy (Hedera) or in epiphytic orchids. Many aerial roots are used to receive water and nutrient intake directly from the air – from fogs, dew or humidity in the air.[30] Some rely on leaf systems to gather rain or humidity and even store it in scales or pockets. Other aerial roots, such as mangrove aerial roots, are used for aeration and not for water absorption. Other aerial roots are used mainly for structure, functioning as prop roots, as in maize or anchor roots or as the trunk in strangler fig. In some Epiphytes – plants living above the surface on other plants, aerial roots serve for reaching to water sources or reaching the surface, and then functioning as regular surface roots.[30]
- Canopy roots/arboreal roots: roots that form when tree branches support mats of epiphytes and detritus, which hold water and nutrients in the canopy. They grow out into these mats, likely to utilize the available nutrients and moisture.[31]
- Contractile roots: roots that pull bulbs or corms of dandelion, deeper in the soil through expanding radially and contracting longitudinally. They have a wrinkled surface.[32]
- Coarse roots: roots that have undergone secondary thickening and have a woody structure. These roots have some ability to absorb water and nutrients, but their main function is transport and to provide a structure to connect the smaller diameter, fine roots to the rest of the plant.
- Dimorphic root systems: roots with two distinctive forms for two separate functions
- Fine roots: typically primary roots <2 mm diameter that have the function of water and nutrient uptake. They are often heavily branched and support mycorrhizas. These roots may be short lived, but are replaced by the plant in an ongoing process of root 'turnover'.
- Haustorial roots: roots of parasitic plants that can absorb water and nutrients from another plant, such as in dodder.
- Propagative roots: roots that form adventitious buds that develop into aboveground shoots, termed Canada thistle, cherryand many others.
- Proteoid roots or cluster roots: dense clusters of rootlets of limited growth that develop under low phosphate or low iron conditions in Proteaceae and some plants from the following families Betulaceae, Casuarinaceae, Elaeagnaceae, Moraceae, Fabaceae and Myricaceae.
- Stilt roots: adventitious support roots, common among mangroves. They grow down from lateral branches, branching in the soil.
- Storage roots: roots modified for storage of food or water, such as beets. They include some taprootsand tuberous roots.
- Structural roots: large roots that have undergone considerable secondary thickening and provide mechanical support to woody plants and trees.
- Surface roots: roots that proliferate close below the soil surface, exploiting water and easily available nutrients. Where conditions are close to optimum in the surface layers of soil, the growth of surface roots is encouraged and they commonly become the dominant roots.
- Tuberous roots: fleshy and enlarged lateral roots for food or water storage, e.g. sweet potato. A type of storage root distinct from taproot.
- Photosynthetic roots: roots that are green and photosynthesize, providing sugar to the plant. They are similar to phylloclades. Several orchids have these, such as Dendrophylax and Taeniophyllum.
- Root nodules: roots that harbor nitrogen-fixing soil bacteria. These are often very short and rounded. Root nodules are found in virtually all legumes.
- Coralloid roots: similar to root nodules, these provide nitrogen to the plant. They are often larger than nodules, branched, and located at or near the soil surface, and harbor nitrogen-fixing cyanobacteria. They are only found in cycads.
Depths

The distribution of vascular plant roots within soil depends on plant form, the spatial and temporal availability of water and nutrients, and the physical properties of the soil. The deepest roots are generally found in deserts and temperate coniferous forests; the shallowest in tundra, boreal forest and temperate grasslands. The deepest observed living root, at least 60 metres (200 ft) below the ground surface, was observed during the excavation of an open-pit mine in Arizona, US. Some roots can grow as deep as the tree is high. The majority of roots on most plants are however found relatively close to the surface where nutrient availability and aeration are more favourable for growth. Rooting depth may be physically restricted by rock or compacted soil close below the surface, or by anaerobic soil conditions.
Records

| Species | Location | Maximum rooting depth (m) | References[33][34] |
|---|---|---|---|
| Boscia albitrunca | Kalahari desert | 68 | Jennings (1974) |
| Juniperus monosperma | Colorado Plateau | 61 | Cannon (1960) |
| Eucalyptus sp. | Australian forest | 61 | Jennings (1971) |
Acacia erioloba
|
Kalahari desert | 60 | Jennings (1974) |
| Prosopis juliflora | Arizona desert | 53.3 | Phillips (1963) |
Evolutionary history
The fossil record of roots—or rather, infilled voids where roots rotted after death—spans back to the late
Environmental interactions

Light has been shown to have some impact on roots, but its not been studied as much as the effect of light on other plant systems. Early research in the 1930s found that light decreased the effectiveness of
Certain plants, namely
Soil temperature is a factor that effects
Plant interactions
Plants can interact with one another in their environment through their root systems. Studies have demonstrated that plant-plant interaction occurs among root systems via the soil as a medium. Researchers have tested whether plants growing in ambient conditions would change their behavior if a nearby plant was exposed to drought conditions.[41] Since nearby plants showed no changes in stomatal aperture researchers believe the drought signal spread through the roots and soil, not through the air as a volatile chemical signal.[42]
Soil interactions
Soil microbiota can suppress both disease and beneficial root symbionts (mycorrhizal fungi are easier to establish in sterile soil). Inoculation with soil bacteria can increase internode extension, yield and quicken flowering. The migration of bacteria along the root varies with natural soil conditions. For example, research has found that the root systems of wheat seeds inoculated with
Grass root systems are beneficial at reducing
Scientists have observed significant diversity of the microbial cover of roots at around 10 percent of three week old root segments covered. On younger roots there was even low coverage, but even on 3-month-old roots the coverage was only around 37%. Before the 1970s, scientists believed that the majority of the root surface was covered by microorganisms.[4]
Nutrient absorption
Researchers studying
Researchers found that partial deficiencies of K or P did not change the
Economic importance

The term
Tree roots can heave and destroy concrete sidewalks and crush or clog buried pipes.[46] The aerial roots of strangler fig have damaged ancient Mayan temples in Central America and the temple of Angkor Wat in Cambodia.
Trees stabilize soil on a slope prone to
Roots can also protect the environment by holding the soil to reduce soil erosion. This is especially important in areas such as

See also
- Absorption of water
- Cypress knee
- Drought rhizogenesis
- Fibrous root system
- Mycorrhiza – root symbiosis in which individual hyphae extending from the mycelium of a fungus colonize the roots of a host plant.
- Mycorrhizal network
- Plant physiology
- Rhizosphere – region of soil around the root influenced by root secretions and microorganisms present
- Root cutting
- Rooting powder
- Stolon
- Tanada effect
- Taproot
References
- ISBN 978-1-83947-180-3.
- ^ "Plant parts=Roots". University of Illinois Extension.
- ISBN 978-0-8493-4925-6.
- ^ a b c "Backyard Gardener: Understanding Plant Roots". University of Arizona Cooperative Extension.
- ^ Gangulee HC, Das KS, Datta CT, Sen S. College Botany. Vol. 1. Kolkata: New Central Book Agency.
- ^ Dutta AC, Dutta TC. BOTANY For Degree Students (6th ed.). Oxford University Press.
- ISBN 978-1847925206.
- PMID 16021787.
- ^ S2CID 24181646.
- ^ Fitter AH (1991). "The ecological significance of root system architecture: an economic approach". In Atkinson D (ed.). Plant Root Growth: An Ecological Perspective. Blackwell. pp. 229–243.
- PMID 11706172.
- ISBN 978-1-285-41534-5. Archivedfrom the original on 2018-01-21. Retrieved 2017-04-24.
- ^ "Suberin – an overview | ScienceDirect Topics". www.sciencedirect.com. Retrieved 2021-08-31.
- ^ "Suberin Form & Function – Mark Bernards – Western University". www.uwo.ca. Retrieved 2021-08-31.
- ^ PMID 23785371.
- S2CID 29919924– via Springer.
- PMID 17360695.
- ^ UV-B light sensing mechanism discovered in plant roots, San Francisco State University, December 8, 2008
- PMID 10205161.
- PMID 18811732.
- S2CID 128600212.
- PMID 11541950.
- S2CID 251104211.
- S2CID 231606782.
- ^ PMID 17419844.
- ^ PMID 29321188.
- PMID 27063366.
- S2CID 254769152.
- S2CID 257438007.
- ^ S2CID 85888916.
- S2CID 778003.
- ^ Pütz N (2002). "Contractile roots". In Waisel Y., Eshel A., Kafkafi U. (eds.). Plant roots: The hidden half (3rd ed.). New York: Marcel Dekker. pp. 975–987.
- S2CID 2092130.
- .
- ^ Retallack GJ (1986). "The fossil record of soils" (PDF). In Wright VP (ed.). Paleosols: their Recognition and Interpretation. Oxford: Blackwell. pp. 1–57. Archived (PDF) from the original on 2017-01-07.
- .
- ISBN 978-1-4398-4649-0.
- hdl:2115/44841.
- ^ Postgate, J. (1998). Nitrogen Fixation (3rd ed.). Cambridge, UK: Cambridge University Press.
- ISBN 978-0-8493-5054-2.
- OCLC 1041421612.
- PMID 22408186.
- .
- ^ Plant Roots and their Environment. Elsevier. 1988. p. 17.
- ^ Plant Roots and their Environment. Elsevier. 1988. p. 25.
- The Los Angeles Times. Retrieved 2023-03-30.
Further reading
- Baldocchi DD, Xu L (October 2007). "What limits evaporation from Mediterranean oak woodlands–The supply of moisture in the soil, physiological control by plants or the demand by the atmosphere?". Advances in Water Resources. 30 (10): 2113–22. .
- Brundrett, M. C. (2002). "Coevolution of roots and mycorrhizas of land plants". New Phytologist. 154 (2): 275–304. PMID 33873429.
- Clark L (2004). "Primary Root Structure and Development – lecture notes" (PDF). Archived from the original (PDF) on 3 January 2006.
- Coutts MP (1987). "Developmental processes in tree root systems". Canadian Journal of Forest Research. 17 (8): 761–767. doi:10.1139/x87-122.
- Raven JA, Edwards D (2001). "Roots: evolutionary origins and biogeochemical significance". Journal of Experimental Botany. 52 (Suppl 1): 381–401. PMID 11326045.
- Schenk HJ, Jackson RB (2002). "The global biogeography of roots". Ecological Monographs. 72 (3): 311–328. JSTOR 3100092.
- Sutton RF, Tinus RW (1983). "Root and root system terminology". Forest Science Monograph. 24: 137.
- Phillips WS (1963). "Depth of roots in soil". Ecology. 44 (2): 424. JSTOR 1932198.
- Caldwell MM, Dawson TE, Richards JH (1998). "Hydraulic lift: consequences of water efflux from the roots of plants". Oecologia. 113 (2): 151–161. S2CID 24181646.
