Endoplasmic reticulum
Animal cell diagram | |
|---|---|
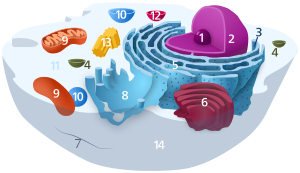 Components of a typical animal cell:
|
The endoplasmic reticulum (ER) is a part of a transportation system of the
The two types of ER share many of the same proteins and engage in certain common activities such as the synthesis of certain lipids and cholesterol. Different types of cells contain different ratios of the two types of ER depending on the activities of the cell. RER is found mainly toward the nucleus of cell and SER towards the cell membrane or plasma membrane of cell.
The outer (
The ER was observed by
Structure

The general structure of the endoplasmic reticulum is a network of membranes called
Rough endoplasmic reticulum
The surface of the rough endoplasmic reticulum (often abbreviated RER or rough ER; also called granular endoplasmic reticulum) is studded with protein-manufacturing
The membrane of the rough endoplasmic reticulum is in the form of large double-membrane sheets that are located near, and continuous with, the outer layer of the
The rough endoplasmic reticulum is key in multiple functions:
- Manufacture of mannose-6-phosphate marker added in the cis-Golgi network.[18]
- Manufacture of secreted proteins, either secreted constitutively with no tag or secreted in a regulatory manner involving clathrin and paired basic amino acids in the signal peptide.
- SNAREproteins are key in the fusion event.
- Initial glycosylation as assembly continues. This is N-linked (O-linking occurs in the Golgi).
Smooth endoplasmic reticulum

In most cells the smooth endoplasmic reticulum (abbreviated SER) is scarce. Instead there are areas where the ER is partly smooth and partly rough, this area is called the transitional ER. The transitional ER gets its name because it contains ER exit sites. These are areas where the transport vesicles that contain lipids and proteins made in the ER, detach from the ER and start moving to the
Sarcoplasmic reticulum

The sarcoplasmic reticulum (SR), from the Greek σάρξ sarx ("flesh"), is smooth ER found in
Functions
The endoplasmic reticulum serves many general functions, including the folding of protein molecules in sacs called
Protein transport
Secretory proteins, mostly
The endoplasmic reticulum is also part of a protein sorting pathway. It is, in essence, the transportation system of the eukaryotic cell. The majority of its resident proteins are retained within it through a retention motif. This motif is composed of four amino acids at the end of the protein sequence. The most common retention sequences are KDEL for lumen located proteins and KKXX for transmembrane protein.[31] However, variations of KDEL and KKXX do occur, and other sequences can also give rise to endoplasmic reticulum retention. It is not known whether such variation can lead to sub-ER localizations. There are three KDEL (1, 2 and 3) receptors in mammalian cells, and they have a very high degree of sequence identity. The functional differences between these receptors remain to be established.[32]
Bioenergetics regulation of ER ATP supply by a CaATiER mechanism

The endoplasmic reticulum does not harbor an ATP-regeneration machinery, and therefore requires ATP import from mitochondria. The imported ATP is vital for the ER to carry out its house keeping cellular functions, such as for protein folding and trafficking.[33]
The ER ATP transporter, SLC35B1/AXER, was recently cloned and characterized,[34] and the mitochondria supply ATP to the ER through a Ca2+-antagonized transport into the ER (CaATiER) mechanism.[35] The CaATiER mechanism shows sensitivity to cytosolic Ca2+ ranging from high nM to low μM range, with the Ca2+-sensing element yet to be identified and validated.[citation needed]
Clinical significance
Increased and supraphysiological ER stress in pancreatic β cells disrupts normal insulin secretion, leading to hyperinsulinemia[36] and consequently peripheral insulin resistance associated with obesity in humans.[37] Human clinical trials also suggested a causal link between obesity-induced increase in insulin secretion and peripheral insulin resistance.[38]
Abnormalities in
The
References
- ^ "Endoplasmic Reticulum (Rough and Smooth)". British Society of Cell Biology. Archived from the original on 24 November 2015. Retrieved 21 November 2015.
- OCLC 493441682.
- PMID 14283576.
- PMID 19871454.
- PMID 13052830.
- ^ ISBN 978-0-8153-3218-3. Archivedfrom the original on 2017-10-03.
- ISBN 978-0-87893-106-4.
- ^ "reticulum". The Free Dictionary.
- S2CID 19078317.
- ISBN 978-0-7167-4366-8.
- PMID 10931837.
- ^ S2CID 16107069.
- PMID 23870120.
- PMID 25396396.
- McGraw-Hill Encyclopedia of Science and Technology. Retrieved September 13, 2006, from Answers.com Web site: "Answers - the Most Trusted Place for Answering Life's Questions". Answers.com. Archivedfrom the original on 2006-11-16. Retrieved 2006-09-13.
- PMID 15350976.
- PMID 16806880.
- PMID 30237200.
- PMID 28119445.
- S2CID 34302.
- PMID 29290583.
- ^ "Functions of Smooth ER". University of Minnesota Duluth.
- PMID 12370264.
- S2CID 4316039.
- ISBN 9780080919317.
- ISBN 978-0321909077.
- PMID 16200199.
- S2CID 25858120.
- S2CID 22517395.
- PMID 1363623.
- PMID 12631711.
- PMID 18086916.
- PMID 1740446.
- PMID 30154480.
- PMID 31498082.
- PMID 34163039.
- PMID 32651241.
- PMID 34905513.
- PMID 21389082.
- PMID 18775308.
- ^ Walter, Peter. "Peter Walter's Short Talk: Unfolding the UPR". iBiology.
- S2CID 25570626.
External links
- Lipid and protein composition of Endoplasmic reticulum in OPM database
- Animations of the various cell functions referenced here Archived 2008-04-22 at the Wayback Machine
