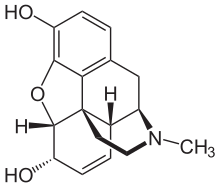
Morphine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈmɔːrfiːn/ |
| Trade names | Statex, MS Contin, Oramorph, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682133 |
| License data | |
| Pregnancy category |
|
intrathecal | |
| Drug class | Opioid |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 20–40% (by mouth), 36–71% (rectally),[9] 100% (IV/IM) |
| Protein binding | 30–40% |
| Metabolism | Liver: UGT2B7 |
| Metabolites | • Morphine-3-glucuronide (90%) • Morphine-6-glucuronide (10%) |
| Onset of action | 5 minutes (IV), 15 minutes (IM),[10] 20 minutes (PO)[11] |
| Elimination half-life | 2–3 hours |
| Duration of action | 3–7 hours[12][13] |
| Excretion | Kidney 90%, bile duct 10% |
| Identifiers | |
| |
JSmol) | |
| Solubility in water | HCl & sulf.: 60 |
| |
| |
| (verify) | |
Morphine, formerly also called morphia, is a strong
Potentially serious
Morphine was first isolated between 1803 and 1805 by German pharmacist
The primary source of morphine is isolation from
Medical uses
Pain
Morphine is used primarily to treat both acute and chronic severe pain. Its duration of analgesia is about three to seven hours.[12][13] Side-effects of nausea and constipation are rarely severe enough to warrant stopping treatment.
It is used for pain due to
Morphine has also traditionally been used in the treatment of
A 2016 Cochrane review concluded that morphine is effective in relieving cancer pain.[30]
Shortness of breath
Morphine is beneficial in reducing the symptom of shortness of breath due to both cancer and noncancer causes.[31][32] In the setting of breathlessness at rest or on minimal exertion from conditions such as advanced cancer or end-stage cardiorespiratory diseases, regular, low-dose sustained-release morphine significantly reduces breathlessness safely, with its benefits maintained over time.[33][34]
Opioid use disorder
Morphine is also available as a slow-release formulation for
-
Two capsules (5 mg & 10 mg) of morphine sulfate extended-release
-
1 milliliter ampoule containing 10 mg of morphine
Contraindications
Relative contraindications to morphine include:
- respiratory depression when appropriate equipment is not available.[12]
- Although it has previously been thought that morphine was contraindicated in acute pancreatitis, a review of the literature shows no evidence for this.[36]
Adverse effects
- Common and short term
- Itchiness[37]
- Nausea[37]
- Vomiting[37]
- Constipation[37]
- Drowsiness[37]
- Dry mouth[37]
- Respiratory depression[12]
- Itching
- Other
- Dizziness
- Decreased sex drive
- Loss of appetite
- Impaired sexual function
- Decreased testosterone levels
- Depression
- Immunodeficiency
- Opioid-induced abnormal pain sensitivity
- Irregular menstruation
- Increased risk of falls
- Slowed breathing
Constipation
Like
Hormone imbalance
Clinical studies consistently conclude that morphine, like other opioids, often causes
Effects on human performance
Most reviews conclude that opioids produce minimal impairment of human performance on tests of sensory, motor, or attentional abilities. However, recent studies have been able to show some impairments caused by morphine, which is not surprising, given that morphine is a central nervous system depressant. Morphine has resulted in impaired functioning on critical flicker frequency (a measure of overall CNS arousal) and impaired performance on the Maddox wing test (a measure of the deviation of the visual axes of the eyes). Few studies have investigated the effects of morphine on motor abilities; a high dose of morphine can impair finger tapping and the ability to maintain a low constant level of isometric force (i.e. fine motor control is impaired),[42] though no studies have shown a correlation between morphine and gross motor abilities.
In terms of
In chronic opioid users, such as those on Chronic Opioid Analgesic Therapy (COAT) for managing severe,
Reinforcement disorders
Addiction

Morphine is a highly
Tolerance
Several hypotheses are given about how tolerance develops, including opioid receptor
Dependence and withdrawal
This section needs additional citations for verification. (November 2019) |
Cessation of dosing with morphine creates the prototypical opioid withdrawal syndrome, which, unlike that of
Acute morphine withdrawal, along with that of any other opioid, proceeds through a number of stages. Other opioids differ in the intensity and length of each, and weak opioids and mixed agonist-antagonists may have acute withdrawal syndromes that do not reach the highest level. As commonly cited[by whom?], they are:
- Stage I, 6 h to 14 h after last dose: Drug craving, anxiety, irritability, perspiration, and mild to moderate dysphoria
- Stage II, 14 h to 18 h after last dose: Yawning, heavy perspiration, mild depression, lacrimation, crying, headaches, runny nose, dysphoria, also intensification of the above symptoms, "yen sleep" (a waking trance-like state)
- Stage III, 16 h to 24 h after last dose: Rhinorrhea (runny nose) and increase in other of the above, dilated pupils, piloerection (goose bumps – a purported origin of the phrase, 'cold turkey,' but in fact the phrase originated outside of drug treatment),[49] muscle twitches, hot flashes, cold flashes, aching bones and muscles, loss of appetite, and the beginning of intestinal cramping
- Stage IV, 24 h to 36 h after last dose: Increase in all of the above including severe cramping and involuntary leg movements ("kicking the habit" also called restless leg syndrome), loose stool, insomnia, elevation of blood pressure, moderate elevation in body temperature, increase in frequency of breathing and tidal volume, tachycardia(elevated pulse), restlessness, nausea
- Stage V, 36 h to 72 h after last dose: Increase in the above, fetal position, vomiting, free and frequent liquid diarrhea, which sometimes can accelerate the time of passage of food from mouth to out of system, weight loss of 2 kg to 5 kg per 24 h, increased white cell count, and other blood changes
- Stage VI, after completion of above: Recovery of appetite and normal bowel function, beginning of transition to post acute and chronic symptoms that are mainly psychological, but may also include increased sensitivity to pain, hypertension, colitis or other gastrointestinal afflictions related to motility, and problems with weight control in either direction
In advanced stages of withdrawal, ultrasonographic evidence of pancreatitis has been demonstrated in some patients and is presumably attributed to spasm of the pancreatic sphincter of Oddi.[50]
The withdrawal symptoms associated with morphine addiction are usually experienced shortly before the time of the next scheduled dose, sometimes within as early as a few hours (usually 6 h to 12 h) after the last administration. Early symptoms include watery eyes, insomnia, diarrhea, runny nose, yawning, dysphoria, sweating, and in some cases a strong drug craving. Severe headache, restlessness, irritability, loss of appetite, body aches, severe abdominal pain, nausea and vomiting, tremors, and even stronger and more intense drug craving appear as the syndrome progresses. Severe depression and vomiting are very common. During the acute withdrawal period, systolic and diastolic blood pressures increase, usually beyond premorphine levels, and heart rate increases,[51] which have potential to cause a heart attack, blood clot, or stroke.
Chills or cold flashes with goose bumps ("cold turkey") alternating with flushing (hot flashes), kicking movements of the legs ("kicking the habit"[46]) and excessive sweating are also characteristic symptoms.[52] Severe pains in the bones and muscles of the back and extremities occur, as do muscle spasms. At any point during this process, a suitable narcotic can be administered that will dramatically reverse the withdrawal symptoms. Major withdrawal symptoms peak between 48 h and 96 h after the last dose and subside after about 8 to 12 days. Sudden withdrawal by heavily dependent users who are in poor health is very rarely fatal. Morphine withdrawal is considered less dangerous than alcohol, barbiturate, or benzodiazepine withdrawal.[53][54]
The psychological dependence associated with morphine addiction is complex and protracted. Long after the physical need for morphine has passed, the addict will usually continue to think and talk about the use of morphine (or other drugs) and feel strange or overwhelmed coping with daily activities without being under the influence of morphine. Psychological withdrawal from morphine is usually a very long and painful process. Addicts often experience severe depression, anxiety, insomnia, mood swings, amnesia (forgetfulness), low self-esteem, confusion, paranoia, and other psychological disorders. Without intervention, the syndrome will run its course, and most of the overt physical symptoms will disappear within 7 to 10 days including psychological dependence. A high probability of relapse exists after morphine withdrawal when neither the physical environment nor the behavioral motivators that contributed to the abuse have been altered. Testimony to morphine's addictive and reinforcing nature is its relapse rate. Abusers of morphine (and heroin) have one of the highest relapse rates among all drug users, ranging up to 98% in the estimation of some medical experts.[55]
Toxicity
| Properties of Morphine | |||||
|---|---|---|---|---|---|
| Molar mass[56] | 285.338 g/mol | ||||
Acidity (pKa)[56]
|
| ||||
| Solubility[56] | 0.15 g/L at 20 °C | ||||
| Melting point[56] | 255 °C | ||||
| Boiling point[56] | 190 °C sublimes | ||||
A large
Pharmacology
Pharmacodynamics
| Compound | Ki ) |
Ratio | Ref | ||
|---|---|---|---|---|---|
MOR |
DOR |
KOR |
MOR:DOR:KOR | ||
| Morphine | 1.8 nM | 90 nM | 317 nM | 1:50:176 | [59] |
| (−)-Morphine | 1.24 nM | 145 nM | 23.4 nM | 1:117:19 | [60] |
| (+)-Morphine | >10 μM | >100 μM | >300 μM | ND | [60] |
| Compound | Route | Dose |
|---|---|---|
| Codeine | PO | 200 mg |
| Hydrocodone | PO | 20–30 mg |
| Hydromorphone | PO | 7.5 mg |
| Hydromorphone | IV | 1.5 mg |
| Morphine | PO | 30 mg |
| Morphine | IV | 10 mg |
| Oxycodone | PO | 20 mg |
| Oxycodone | IV | 10 mg |
| Oxymorphone | PO | 10 mg |
| Oxymorphone | IV | 1 mg |
Due to its long history and established use as a pain medication, this compound has become the benchmark to which all other opioids are compared.
Morphine is a
The effects of morphine can be countered with

Gene expression
Studies have shown that morphine can alter the expression of a number of
Effects on the immune system
Morphine has long been known to act on receptors expressed on cells of the
The first step of determining that morphine may affect the immune system was to establish that the opiate receptors known to be expressed on cells of the central nervous system are also expressed on cells of the immune system. One study successfully showed that
This regulation of cytokines appear to occur via the
Further studies on the effects of morphine on the immune system have shown that morphine influences the production of
Pharmacokinetics
Absorption and metabolism
Morphine can be taken
Extended-release
There are
Detection in body fluids
Morphine and its major metabolites, morphine-3-glucuronide and morphine-6-glucuronide, can be detected in blood, plasma, hair, and urine using an
Ingestion of codeine or food containing poppy seeds can cause false positives.[85]
A 1999 review estimated that relatively low doses of heroin (which metabolizes immediately into morphine) are detectable by standard urine tests for 1–1.5 days after use.
Chirality and biological activity
Morphine is a pentacyclic 3°amine (alkaloid) with 5 stereogenic centers and exists in 32 stereoisomeric forms. But the desired analgesic activity resides exclusively in the natural product, the (-)-enantiomer with the configuration (5R,6S,9R,13S,14R).[88][89]
Natural occurrence

Morphine is the most abundant opiate found in
In the brain of mammals, morphine is detectable in trace steady-state concentrations.
Human biosynthesis
This section needs expansion with: a more standard presentation, without a scheme-in-text, and with description of key enzymes, points of pathway regulation, etc. You can help by adding to it. (October 2016) |
Morphine is an
- L-tyrosine → L-DOPA → 3,4-dihydroxyphenylacetaldehyde(DOPAL)
- Dopamine + DOPAL → (S)-norlaudanosoline →→→ (S)-reticuline → 1,2-dehydroreticulinium → (R)-reticuline → salutaridine → salutaridinol → thebaine → neopinone → codeinone → codeine → morphine
The intermediate (S)-norlaudanosoline (also known as tetrahydropapaveroline) is synthesized through addition of DOPAL and dopamine.[14] CYP2D6, a cytochrome P450 isoenzyme is involved in two steps along the biosynthetic pathway, catalyzing both the biosynthesis of dopamine from tyramine and of morphine from codeine.[14][95]
Urinary concentrations of endogenous codeine and morphine have been found to significantly increase in individuals taking L-DOPA for the treatment of Parkinson's disease.[14]
Biosynthesis in the opium poppy
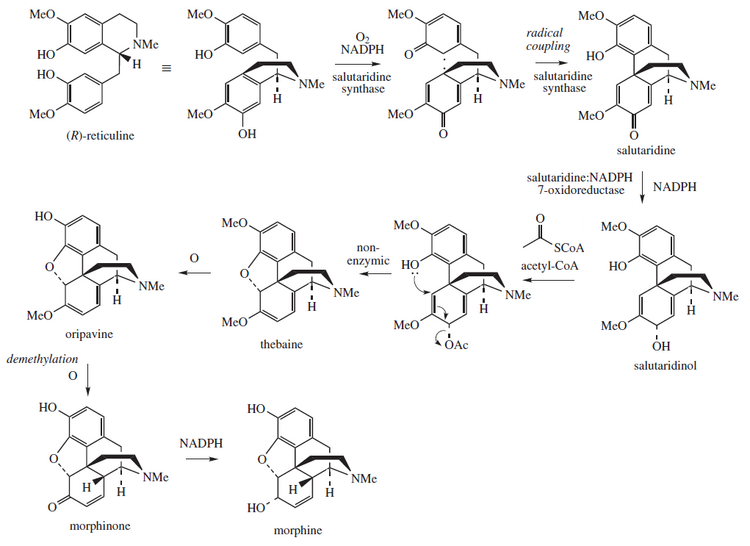
Morphine is biosynthesized in the
Chemistry
This section has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|
Elements of the morphine structure have been used to create completely synthetic drugs such as the
Structure description



Morphine is a benzylisoquinoline alkaloid with two additional ring closures.[101] As Jack DeRuiter of the Department of Drug Discovery and Development (formerly, Pharmacal Sciences), Harrison School of Pharmacy, Auburn University stated in his Fall 2000 course notes for that earlier department's "Principles of Drug Action 2" course, "Examination of the morphine molecule reveals the following structural features important to its pharmacological profile...
- A rigid
pentacyclic structure consisting of a benzene ring (A), two partially unsaturated cyclohexane rings (B and C), a piperidine ring (D) and a tetrahydrofuran ring (E). Rings A, B and C are the phenanthrenering system. This ring system has little conformational flexibility...- Two hydroxyl functional groups: a C3-
allylic[hydroxyl group],- An ether linkage between E4 and E5,
- Unsaturation between C7 and C8,
- A basic, [tertiary]-amine function at position 17, [and]
- [Five] centers of chirality (C5, C6, C9, C13 and C14) with morphine exhibiting a high degree of stereoselectivity of analgesic action."
better source needed][needs update]
Morphine and most of its derivatives do not exhibit optical isomerism, although some more distant relatives like the morphinan series (levorphanol, dextorphan and the racemic parent chemical racemorphan) do,[103] and as noted above stereoselectivity in vivo is an important issue.[citation needed]
Uses and derivatives
Most of the licit morphine produced is used to make codeine by methylation.[104] It is also a precursor for many drugs including heroin (3,6-diacetylmorphine), hydromorphone (dihydromorphinone), and oxymorphone (14-hydroxydihydromorphinone).[105] Most semi-synthetic opioids, both of the morphine and codeine subgroups, are created by modifying one or more of the following:[citation needed]
- Halogenating or making other modifications at positions 1 or 2 on the morphine carbon skeleton.
- The methyl group that makes morphine into codeine can be removed or added back, or replaced with another functional group like ethyl and others to make codeine analogues of morphine-derived drugs and vice versa. Codeine analogues of morphine-based drugs often serve as prodrugs of the stronger drug, as in codeine and morphine, hydrocodone and hydromorphone, oxycodone and oxymorphone, nicocodeine and nicomorphine, dihydrocodeine and dihydromorphine, etc.
- Saturating, opening, or other changes to the bond between positions 7 and 8, as well as adding, removing, or modifying functional groups to these positions; saturating, reducing, eliminating, or otherwise modifying the 7–8 bond and attaching a functional group at 14 yields hydromorphinol; the oxidation of the hydroxyl group to a carbonyl and changing the 7–8 bond to single from double changes codeine into oxycodone.
- Attachment, removal or modification of functional groups to positions 3 or 6 (dihydrocodeine and related, hydrocodone, nicomorphine); in the case of moving the methyl functional group from position 3 to 6, codeine becomes heterocodeine, which is 72 times stronger, and therefore six times stronger than morphine
- Attachment of functional groups or other modification at position 14 (oxymorphone, oxycodone, naloxone)
- Modifications at positions 2, 4, 5 or 17, usually along with other changes to the molecule elsewhere on the morphine skeleton. Often this is done with drugs produced by catalytic reduction, hydrogenation, oxidation, or the like, producing strong derivatives of morphine and codeine.
Many morphine derivatives can also be manufactured using
As a result of the extensive study and use of this molecule, more than 250 morphine derivatives (also counting codeine and related drugs) have been developed since the last quarter of the 19th century.[citation needed] These drugs range from 25% the analgesic strength of codeine (or slightly more than 2% of the strength of morphine) to several thousand times the strength of morphine, to powerful opioid antagonists, including naloxone (Narcan), naltrexone (Trexan), diprenorphine (M5050, the reversing agent for the Immobilon dart) and nalorphine (Nalline).[citation needed] Some opioid agonist-antagonists, partial agonists, and inverse agonists are also derived from morphine.[citation needed] The receptor-activation profile of the semi-synthetic morphine derivatives varies widely and some, like apomorphine are devoid of narcotic effects.[citation needed]
Chemical salts of Morphine
This section needs additional citations for verification. (March 2021) |
Both morphine and its hydrated form are sparingly soluble in water.[106] For this reason, pharmaceutical companies produce sulfate and hydrochloride salts of the drug, both of which are over 300 times more water-soluble than their parent molecule.[clarification needed][citation needed] Whereas the pH of a saturated morphine hydrate solution is 8.5, the salts are acidic.[citation needed] Since they derive from a strong acid but weak base, they are both at about pH = 5;[clarification needed][citation needed] as a consequence, the morphine salts are mixed with small amounts of NaOH to make them suitable for injection.[citation needed]
A number of salts of morphine are used, with the most common in current clinical use being the hydrochloride, sulfate, tartrate, and citrate;[citation needed] less commonly methobromide, hydrobromide, hydroiodide, lactate, chloride, and bitartrate and the others listed below.[citation needed] Morphine diacetate (heroin) is not a salt, but rather a further derivative,[citation needed] see above.[107]
Morphine meconate is a major form of the alkaloid in the poppy, as is morphine pectinate, nitrate, sulfate, and some others.[
The salts listed by the
| Select salts of morphine with their Controlled Substances Act (CSA) schedule, Administrative Controlled Substances Code Number (ACSCN), and free base conversion ratio.[clarification needed][citation needed] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Production

In the
The poppy straw methods predominate in Continental Europe and the British Commonwealth, with the latex method in most common use in India. The latex method can involve either vertical or horizontal slicing of the unripe pods with a two-to five-bladed knife with a guard developed specifically for this purpose to the depth of a fraction of a millimetre and scoring of the pods can be done up to five times. An alternative latex method sometimes used in China in the past is to cut off the poppy heads, run a large needle through them, and collect the dried latex 24 to 48 hours later.[citation needed]
In India, opium harvested by licensed poppy farmers is dehydrated to uniform levels of hydration at government processing centers, and then sold to pharmaceutical companies that extract morphine from the opium. However, in Turkey and Tasmania, morphine is obtained by harvesting and processing the fully mature dry seed pods with attached stalks, called poppy straw. In Turkey, a water extraction process is used, while in Tasmania, a solvent extraction process is used.[citation needed]
Opium poppy contains at least 50 different alkaloids, but most of them are of very low concentration. Morphine is the principal alkaloid in raw opium and constitutes roughly 8–19% of opium by dry weight (depending on growing conditions).[80] Some purpose-developed strains of poppy now produce opium that is up to 26% morphine by weight.[citation needed] A rough rule of thumb to determine the morphine content of pulverised dried poppy straw is to divide the percentage expected for the strain or crop via the latex method by eight or an empirically determined factor, which is often in the range of 5 to 15.[citation needed] The Norman strain of P. somniferum, also developed in Tasmania, produces down to 0.04% morphine but with much higher amounts of thebaine and oripavine, which can be used to synthesise semi-synthetic opioids as well as other drugs like stimulants, emetics, opioid antagonists, anticholinergics, and smooth-muscle agents.[citation needed]
In the 1950s and 1960s, Hungary supplied nearly 60% of Europe's total medication-purpose morphine production. To this day, poppy farming is legal in Hungary, but poppy farms are limited by law to 2 acres (8,100 m2). It is also legal to sell dried poppy in flower shops for use in floral arrangements.
It was announced in 1973 that a team at the National Institutes of Health in the United States had developed a method for total synthesis of morphine, codeine, and thebaine using coal tar as a starting material. A shortage in codeine-hydrocodone class cough suppressants (all of which can be made from morphine in one or more steps, as well as from codeine or thebaine) was the initial reason for the research.
Most morphine produced for pharmaceutical use around the world is actually converted into codeine as the concentration of the latter in both raw opium and poppy straw is much lower than that of morphine; in most countries, the usage of codeine (both as end-product and precursor) is at least equal or greater than that of morphine on a weight basis.
Chemical synthesis
The first
GMO synthesis
Research
Thebaine has been produced by
Precursor to other opioids
Pharmaceutical
This section needs expansion with: sourced content that brielfy summarises how morphine is used, industrially and globally, to produce other compounds with utility in medicine or research. You can help by adding to it. (February 2020) |
Morphine is a precursor in the manufacture in a number of opioids such as dihydromorphine, hydromorphone, hydrocodone, and oxycodone as well as codeine, which itself has a large family of semi-synthetic derivatives.[124]
Illicit
Illicit morphine is produced, though rarely, from codeine found in over-the-counter cough and pain medicines.[citation needed] Another illicit source is morphine extracted from extended-release morphine products.[125] Chemical reactions can then be used to convert morphine, dihydromorphine, and hydrocodone into heroin or other opioids [e.g., diacetyldihydromorphine (Paralaudin), and thebacon].[citation needed] Other clandestine conversions—of morphine, into ketones of the hydromorphone class, or other derivatives like dihydromorphine (Paramorfan), desomorphine (Permonid), metopon, etc., and of codeine into hydrocodone (Dicodid), dihydrocodeine (Paracodin), etc. —require greater expertise, and types and quantities of chemicals and equipment that are more difficult to source, and so are more rarely used, illicitly (but cases have been recorded).[citation needed]
History

The earliest known reference to morphine can be traced back to Theophrastus in the 3rd century BC, however, possible references to morphine may go as far back as 2100 BC as Sumerian clay tablets which records lists of medical prescriptions include opium-based cures.[126]
An opium-based elixir has been ascribed to alchemists of Byzantine times, but the specific formula was lost during the Ottoman conquest of Constantinople (Istanbul).[127] Around 1522, Paracelsus made reference to an opium-based elixir that he called laudanum from the Latin word laudāre, meaning "to praise". He described it as a potent painkiller, but recommended that it be used sparingly. The recipe given differs substantially from that of modern-day laudanum.[128]
Morphine was discovered as the first active alkaloid extracted from the opium poppy plant in December 1804 in
The drug was first marketed to the general public by Sertürner and Company in 1817 as a pain medication, and also as a treatment for opium and alcohol addiction. It was first used as a poison in 1822 when Edme Castaing of France was convicted of murdering a patient.[133] Commercial production began in Darmstadt, Germany, in 1827 by the pharmacy that became the pharmaceutical company Merck, with morphine sales being a large part of their early growth.[134][135] In the 1850s, Alexander Wood reported that he had injected morphine into his wife Rebecca as an experiment; the myth goes that this killed her because of respiratory depression,[130] but she outlived her husband by ten years.[136]
Later it was found that morphine was more addictive than either alcohol or opium, and its extensive use during the American Civil War allegedly resulted in over 400,000[137] people with the "soldier's disease" of morphine addiction.[138] This idea has been a subject of controversy, as there have been suggestions that such a disease was in fact a fabrication; the first documented use of the phrase "soldier's disease" was in 1915.[139][140]
Diacetylmorphine (better known as heroin) was synthesized from morphine in 1874 and brought to market by Bayer in 1898. Heroin is approximately 1.5 to 2 times more potent than morphine weight for weight. Due to the lipid solubility of diacetylmorphine, it can cross the blood–brain barrier faster than morphine, subsequently increasing the reinforcing component of addiction.[141] Using a variety of subjective and objective measures, one study estimated the relative potency of heroin to morphine administered intravenously to post-addicts to be 1.80–2.66 mg of morphine sulfate to 1 mg of diamorphine hydrochloride (heroin).[45]

Morphine became a controlled substance in the US under the Harrison Narcotics Tax Act of 1914, and possession without a prescription in the US is a criminal offense. Morphine was the most commonly abused narcotic analgesic in the world until heroin was synthesized and came into use. In general, until the synthesis of
The structural formula of morphine was determined by 1925 by
In 2003, there was discovery of endogenous morphine occurring naturally in the human body. Thirty years of speculation were made on this subject because there was a receptor that, it appeared, reacted only to morphine: the μ3-opioid receptor in human tissue.[146] Human cells that form in reaction to cancerous neuroblastoma cells have been found to contain trace amounts of endogenous morphine.[94]
Society and culture
Legal status
- In Schedule 8drug under the variously titled State and Territory Poisons Acts.
- In Canada, morphine is classified as a Schedule I drug under the Controlled Drugs and Substances Act.
- In France, morphine is in the strictest schedule of controlled substances, based upon the December 1970 French controlled substances law.
- In Germany, morphine is a verkehrsfähiges und verschreibungsfähiges Betäubungsmittel listed under Anlage III (the equivalent of CSA Schedule II) of the Betäubungsmittelgesetz.[147]
- In Switzerland, morphine is scheduled similarly to Germany's legal classification of the drug.
- In Japan, morphine is classified as a narcotic under the Narcotics and Psychotropics Control Act (麻薬及び向精神薬取締法, mayaku oyobi kōseishinyaku torishimarihō).
- In the Netherlands, morphine is classified as a List 1 drug under the Opium Law.
- In New Zealand, morphine is classified as a Class B drug under the Misuse of Drugs Act 1975.[148]
- In the Misuse of Drugs Regulations 2001.
- In the tonnes of production for sale, and 27.3 tonnes of production as an intermediate, or chemical precursor, for conversion into other drugs.[149] Morphine produced for use in extremely dilute formulations is excluded from the manufacturing quota.[citation needed]
- Internationally (UN), morphine is a Schedule I drug under the Single Convention on Narcotic Drugs.[150]
Non-medical use

The euphoria, comprehensive alleviation of distress and therefore all aspects of suffering, promotion of sociability and empathy, "body high", and
Animal and human studies and clinical experience back up the contention that morphine is one of the most euphoric drugs known, and via all but the IV route heroin and morphine cannot be distinguished according to studies because heroin is a prodrug for the delivery of systemic morphine. Chemical changes to the morphine molecule yield other euphorigenics such as dihydromorphine, hydromorphone (Dilaudid, Hydal), and oxymorphone (Numorphan, Opana), as well as the latter three's methylated equivalents dihydrocodeine, hydrocodone, and oxycodone, respectively; in addition to heroin, there are dipropanoylmorphine, diacetyldihydromorphine, and other members of the 3,6 morphine diester category like nicomorphine and other similar semi-synthetic opiates like desomorphine, hydromorphinol, etc. used clinically in many countries of the world but also produced illicitly in rare instances.[medical citation needed]
In general, non-medical use of morphine entails taking more than prescribed or outside of medical supervision, injecting oral formulations, mixing it with unapproved potentiators such as alcohol, cocaine, and the like, or defeating the extended-release mechanism by chewing the tablets or turning into a powder for snorting or preparing injectables. The latter method can be as time-consuming and involved as traditional methods of smoking opium. This and the fact that the liver destroys a large percentage of the drug on the first pass impacts the demand side of the equation for clandestine re-sellers, as many customers are not needle users and may have been disappointed with ingesting the drug orally. As morphine is generally as hard or harder to divert than oxycodone in a lot of cases, morphine in any form is uncommon on the street, although ampoules and phials of morphine injection, pure pharmaceutical morphine powder, and soluble multi-purpose tablets are very popular where available.[medical citation needed]
Morphine is also available in a paste that is used in the production of heroin, which can be smoked by itself or turned to a soluble salt and injected; the same goes for the penultimate products of the Kompot (Polish Heroin) and black tar processes. Poppy straw as well as opium can yield morphine of purity levels ranging from poppy tea to near-pharmaceutical-grade morphine by itself or with all of the more than 50 other alkaloids. It also is the active narcotic ingredient in opium and all of its forms, derivatives, and analogues as well as forming from breakdown of heroin and otherwise present in many batches of illicit heroin as the result of incomplete acetylation.[medical citation needed]
Names
Morphine is marketed under many different brand names in various parts of the world.[1] It was formerly called Morphia in British English.[152]
Informal names for morphine include: Cube Juice, Dope, Dreamer, Emsel, First Line, God's Drug, Hard Stuff, Hocus, Hows, Lydia, Lydic, M, Miss Emma, Mister Blue, Monkey, Morf, Morph, Morphide, Morphie, Morpho, Mother, MS, Ms. Emma, Mud, New Jack Swing (if mixed with heroin), Sister, Tab, Unkie, Unkie White, and Stuff.[153]
MS Contin tablets are known as misties, and the 100 mg extended-release tablets as greys and blockbusters. The "
Access in developing countries
Although morphine is cheap, people in poorer countries often do not have access to it. According to a 2005 estimate by the
Experts in pain management attribute the under-distribution of morphine to an unwarranted fear of the drug's potential for addiction and abuse. While morphine is clearly addictive, Western doctors believe it is worthwhile to use the drug and then wean the patient off when the treatment is over.[156]
References
- ^ a b c "International listings for Morphine". Drugs.com. Archived from the original on 14 June 2015. Retrieved 2 June 2015.
- ^ a b "Morphine Use During Pregnancy". Drugs.com. 14 October 2019. Retrieved 21 August 2020.
- ISBN 978-1-4557-0150-6.
- ^ "Morphine Product information". Health Canada. 9 August 2005. Retrieved 4 April 2024.
- ^ )
- ^ "Sevredol Summary of Product Characteristics (SmPC)". (emc). 13 February 2024. Retrieved 20 February 2024.
- ^ "MS Contin- morphine sulfate tablet". DailyMed. 27 December 2023. Retrieved 20 February 2024.
- ^ "Modified-released oral opioids". European Medicines Agency. 18 November 2010. Retrieved 20 February 2024.
- PMID 3387374.
- ISBN 978-0-521-43379-2. Archivedfrom the original on 8 September 2017.
- ISBN 978-0-19-959510-5. Archivedfrom the original on 8 September 2017.
- ^ a b c d e f g h i j k l m n o p "Morphine sulfate". The American Society of Health-System Pharmacists. Archived from the original on 2 May 2015. Retrieved 1 June 2015.
- ^ ISBN 978-1-58255-784-7. Archivedfrom the original on 8 September 2017.
- ^ PMID 22578954. Archived from the original(PDF) on 24 August 2016. Retrieved 10 October 2016.
Positive evolutionary pressure has apparently preserved the ability to synthesize chemically authentic morphine, albeit in homeopathic concentrations, throughout animal phyla.
- ^ ISBN 978-0-674-02990-3. Archivedfrom the original on 8 September 2017.
- ^ ISBN 978-3-7643-8335-0.
- ^ ISBN 978-1-4833-2188-2. Archivedfrom the original on 8 September 2017.
- ISBN 978-1-4522-6601-5. Archivedfrom the original on 8 September 2017.
- ISBN 978-92-1-048119-9. Archivedfrom the original on 8 September 2017.
- ^ ISBN 978-92-1-048157-1. Archived(PDF) from the original on 2 June 2015.
- ^ ISBN 978-1-4381-0211-5.
- ISBN 978-1-4200-0346-8.
- ISBN 978-0-8036-4086-3.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Morphine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "First Generic Drug Approvals 2023". U.S. Food and Drug Administration (FDA). 30 May 2023. Archived from the original on 30 June 2023. Retrieved 30 June 2023.
- PMID 15976786.
- ^ Sosnowski MA. "BestBets: Does the application of opiates, during an attack of Acute Cardiogenic Pulmonary Oedma, reduce patients' mortality and morbidity?". BestBets. Best Evidence Topics. Archived from the original on 16 June 2010. Retrieved 6 December 2008.
- PMID 27105021.
- PMID 20890149.
- PMID 20722311.
- PMID 22336677.
- S2CID 26739450.
- ISBN 978-0-642-82459-2. Archived from the original(PDF) on 10 October 2012.
- S2CID 13209026.
- ^ PMID 16717269.
- PMID 15082884.
- PMID 1755865.
- PMID 23414717.
- PMID 19193821.
- PMID 1755931.
- S2CID 2126631.
- PMID 10760625.
- ^ PMID 13767429.
- ^ a b National Institute on Drug Abuse (NIDA) (April 2013). "Heroin". DrugFacts. U.S. National Institutes of Health. Archived from the original on 30 November 2005. Retrieved 29 April 2008.
- S2CID 21651602.
- PMID 18076994.
- ^ "Why do We Quit 'Cold Turkey'?". Archived from the original on 21 November 2016. Retrieved 21 November 2016.
- ^ "Opiate Withdrawal Stages". Archived from the original on 5 June 2014. Retrieved 13 June 2014.
- PMID 10096766.
- ^ "Morphine (and Heroin)". Drugs and Human Performance Fact Sheets. U.S. National Traffic Safety Administration. Archived from the original on 3 October 2006. Retrieved 17 May 2007.
- ^ "Narcotics". DEA Briefs & Background, Drugs and Drug Abuse, Drug Descriptions. U.S. Drug Enforcement Administration. Archived from the original on 14 January 2012.
{{cite web}}: CS1 maint: unfit URL (link) - ISBN 978-1-59403-087-1.
- ISBN 978-0-911910-00-1.
- ^ ISBN 978-0-8493-0485-9.
- ^ Duldner Jr JE (2 March 2009). "Morphine overdose". MedlinePlus. U.S. National Library of Medicine. Archived from the original on 24 May 2016.
- PMID 22784117.
- ISSN 0171-2004.
- ^ PMID 7562497.
- ISBN 978-1-4496-1073-9.
- ISBN 978-0-323-11374-8.
- ISBN 978-0-7295-8162-2.
- ISBN 978-1-4377-1679-5.
- PMID 22816017.
- PMID 23019498.
- ^ a b "MS-Contin (Morphine Sulfate Controlled-Release) Drug Information: Clinical Pharmacology". Prescribing Information. RxList. Archived from the original on 15 May 2007.
- PMID 23646826.
- S2CID 10033780.
- PMID 8747752.
- S2CID 18200635.
- S2CID 19669811.
- S2CID 9296949.
- S2CID 39489581.
- PMID 17380193.
- PMID 17908329.
- PMID 18443637.
- S2CID 20887610.
- ^ from the original on 1 December 2008.
- ^ a b Jenkins AJ (2008) Pharmacokinetics of specific drugs. In Karch SB (Ed), Pharmacokinetics and pharmacodynamics of abused drugs. CRC Press: Boca Raton.
- ^ a b c "Morphine, slow release (By mouth)". University of Maryland Medical Center. Archived from the original on 22 December 2015.
- S2CID 5603973. Last reviewed on 18 November 2015
- ^ a b "Dosing & Uses". Medscape. Archived from the original on 31 October 2015. Retrieved 21 December 2015.
- ^ a b "EndLink: An Internet-based End of Life Care Education Program – Morphine Dosing" (PDF). Northwestern University. Archived (PDF) from the original on 4 March 2016.
- ISBN 978-0-9626523-7-0.
- S2CID 43808583.
- S2CID 385874.
- OCLC 27642721.
- ^ "Morphine – Chiralpedia". 18 July 2022. Retrieved 28 August 2022.
- ISBN 978-1-56024-923-8.
- PMID 925935.
- ISBN 978-0-387-98543-5.
- ^ "μ receptor". IUPHAR/BPS Guide to PHARMACOLOGY. International Union of Basic and Clinical Pharmacology. 15 March 2017. Archived from the original on 7 November 2017. Retrieved 28 December 2017.
Morphine occurs endogenously
- ^ PMID 15383669.
Without doubt, human cells can produce the alkaloid morphine.
- PMID 24374199.
Additionally, CYP2D is involved in the synthesis of endogenous morphine from various precursors, including L-3,4-dihydroxyphenylalanine (L-DOPA), reticulin, tetrahydropapaveroline (THP), and tyramine (Kulkarni, 2001; Mantione et al., 2008; Zhu, 2008).
- (PDF) from the original on 19 June 2012.
- ^ Le Page M (18 May 2015). "Home-brew heroin: soon anyone will be able to make illegal drugs". New Scientist. Archived from the original on 13 April 2016.
- ^ Service RF (25 June 2015). "Final step in sugar-to-morphine conversion deciphered". Science. Archived from the original on 21 August 2015.
- PMID 26272907.
- ^ "Yeast-Based Opioid Production Completed". 13 August 2015. Archived from the original on 7 September 2015. Retrieved 15 August 2015.
- ^ DeRuiter J (Fall 2000). "Narcotic analgesics: morphine and "peripherally modified" morphine analogs" (PDF). Principles of Drug Action 2. Auburn University. Archived (PDF) from the original on 11 January 2012.
- PMID 13999272.
- ^ "UNODC - Bulletin on Narcotics - 1958 Issue 3 - 005". United Nations : Office on Drugs and Crime. Retrieved 11 February 2022.
- PMID 31643200, retrieved 14 November 2023
- OCLC 1136560730.
- ^ Heroin (morphine diacetate) is a Schedule I controlled substance, so it is not used clinically in the United States;[citation needed] it is a sanctioned medication in the United Kingdom and in Canada and some countries in Continental Europe, its use being particularly common (nearly to the degree of the hydrochloride salt)[clarification needed] in the United Kingdom.[citation needed]
- ^ Morphine valerate was one ingredient of a medication available for both oral and parenteral administration popular many years ago in Europe and elsewhere called Trivalin—not to be confused with the current, unrelated herbal preparation of the same name—which also included the valerates of caffeine and cocaine.[citation needed] A version containing codeine valerate as a fourth ingredient is distributed under the name Tetravalin.[citation needed]
- .
- .
- .
- .
- .
- .
- .
- .
- S2CID 32048193.
- PMID 12465957.
- PMID 17078705.
- PMID 18561354.
- PMID 19624126.
- ^ Freemantle M (20 June 2005). "The Top Pharmaceuticals That Changed The World-Morphine". Chemical and Engineering News.
- ^ "Genetically modified E. coli pump out morphine precursor: Bacteria yield 300 times more opiates than yeast". ScienceDaily.
- ^ "Narcotics (Opioids) | DEA". www.dea.gov. Retrieved 21 January 2021.
- PMID 2249204.
- PMID 17152761.
- .
- ^ Sigerist HE (1941). "Laudanum in the Works of Paracelsus" (PDF). Bull. Hist. Med. 9: 530–544. Retrieved 5 September 2018.
- ^ "Friedrich Sertürner (Untitled letter to the editor)" [Journal of Pharmacy for Physicians, Apothecaries, and Chemists]. Journal der Pharmacie für Aerzte, Apotheker und Chemisten (in German). 13: 229–243, see especially "III. Säure im Opium" (acid in opium), pp. 234–235, and "I. Nachtrag zur Charakteristik der Säure im Opium" (Addendum on the characteristics of the acid in opium), pp. 236–241. 1805. Archived from the original on 17 August 2016.
- ^ PMID 20010421.
- ^ Sertürner coined the term morphium in: Sertuerner (1817) "Ueber das Morphium, eine neue salzfähige Grundlage, und die Mekonsäure, als Hauptbestandtheile des Opiums" (On morphine, a new salifiable [i.e., precipitable], fundamental substance, and meconic acid, as principal components of opium), Annalen der Physik, 55 : 56–89. It was Gay-Lussac, a French chemist and editor of Annales de Chimie et de Physique, who coined the word morphine in a French translation of Sertuener's original German article: Sertuener (1817) "Analyse de l'opium: De la morphine et de l'acide méconique, considérés comme parties essentielles de l'opium" (Analysis of opium: On morphine and on meconic acid, considered as essential constituents of opium), Annales de Chimie et de Physique, 2nd series, 5 : 21–42. From p. 22: " ... car il a pris pour cette substance, que j'appelle morphine (morphium), ce qui n'en était qu'une combinaison avec l'acide de l'opium." ( ... for he [i.e., French chemist and pharmacist Charles Derosne (1780–1846)] took as that substance [i.e., the active ingredient in opium], which I call "morphine" (or morphium), what was only a compound of it with acid of opium.)
- ^ Offit P (March–April 2017). "God's Own Medicine". Skeptical Inquirer. 41 (2): 44.
- ^ Annual Register. J. Dodsley. 1824. p. 1. Retrieved 1 September 2015.
Edme.
- OCLC 966360188.
- OCLC 1162209949.
- ISBN 978-0-393-32545-4.
- ^ Vassallo SA (July 2004). "Lewis H. Wright Memorial Lecture". ASA Newsletter. 68 (7): 9–10. Archived from the original on 2 February 2014.
- ^ "Opiate Narcotics". The Report of the Canadian Government Commission of Inquiry into the Non-Medical Use of Drugs. Canadian Government Commission. Archived from the original on 4 April 2007.
- ^ Mandel J. "Mythical Roots of US Drug Policy – Soldier's Disease and Addicts in the Civil War". Archived from the original on 5 April 2007.
- ^ "Soldiers Disease A Historical Hoax?". iPromote Media Inc. 2006. Archived from the original on 27 September 2007.
- S2CID 17860947.
- ^ "Morphine Easy Home Cure". Overland Monthly. 35 (205): 14. 1900. Archived from the original on 1 February 2014.
- ^ Gulland JM, Robinson R (1925). "Constitution of codeine and thebaine". Memoirs and Proceedings of the Literary and Philosophical Society of Manchester. 69: 79–86.
- ^ Dickman S (3 October 2003). "Marshall D. Gates, Chemist to First Synthesize Morphine, Dies". Press Release. University of Rochester. Archived from the original on 1 December 2010.
- PMID 3314338.
- PMID 16301642.
- ^ "Anlage III (zu § 1 Abs. 1) verkehrsfähige und verschreibungsfähige Betäubungsmittel" [Appendix III (to §1 paragraph 1) marketable and prescription narcotics]. Bundesamt für Justiz (Federal Office of Justice) (in German). Archived from the original on 28 April 2014.
- ^ "Misuse of Drugs Act 1975, sch 2". 13 January 2022. Archived from the original on 27 January 2021.
- ^ 82 FR 51293
- ^ "List of narcotic drugs under international control" (PDF). Yellow List (PDF) (50th ed.): 5. March 2011. Archived (PDF) from the original on 22 December 2014.
- S2CID 236695938, retrieved 11 January 2024
- ^ "Morphia". Lexico Dictionaries | English. Archived from the original on 4 August 2020. Retrieved 14 September 2019.
- ISBN 978-0-313-31807-8.
- ^ Milani B. Scholten (3rd ed.). Switzerland: World Health Organization. pp. 1–22. Archived from the original on 5 June 2018. Retrieved 17 January 2018.
- ^ Human Rights Watch (2 June 2011). "Global State of Pain Treatment; Access to Palliative Care as a Human Right". Human Rights Watch. Retrieved 27 January 2020.
- ^ McNeil Jr DG (10 September 2007). "Drugs Banned, Many of World's Poor Suffer in Pain". The New York Times. Archived from the original on 12 May 2023. Retrieved 11 September 2007.
External links
- "Morphine Injection". MedlinePlus.
- "Morphine Rectal". MedlinePlus.


