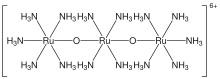
Ruthenium red
| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.228.922 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Cl6H42N14O2Ru3 | |
| Molar mass | 786.34 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The inorganic dye ammoniated mucopolysaccharides.
Ruthenium red (RR) has also been used as a pharmacological tool to study specific cellular mechanisms. Selectivity is a significant issue in such studies as RR is known to interact with many proteins.myosin light-chain phosphatase, and Ca2+ binding proteins such as calmodulin. Ruthenium red displays nanomolar potency against several of its binding partners (e.g. TRPV4, ryanodine receptors,...). For example, it is a potent inhibitor of intracellular calcium release by ryanodine receptors (Kd ~20 nM).[6] As a TRPA1 blocker, it assists in reducing the airway inflammation caused by pepper spray.
RR has been used on plant material since 1890 for staining
pectins, mucilages, and gums. RR is a stereoselective stain for pectic acid, insofar as the staining site occurs between each monomer unit and the next adjacent neighbor.[7]
References
- PMID 21671873.
- PMID 22711817.
- PMID 23345406.
- PMID 24336289.
- PMID 17074387.
- S2CID 86436194. Archived from the originalon 2008-04-23. Retrieved 2006-10-22.
- ^ Mariani Colombo P, Rascio N. "Ruthenium red staining for electron microscopy of plant material". Journal of Ultrastructure Research Volume 60, Issue 2, August 1977, Pages 135–139
