Secretion

Secretion is the movement of material from one point to another, such as a secreted
In eukaryotic cells

Mechanism
In the Golgi apparatus, the glycosylation of the proteins is modified and further
Eventually, there is vesicle fusion with the cell membrane at porosomes, by a process called exocytosis, dumping its contents out of the cell's environment.[2]
Strict
Nonclassical secretion
There are many proteins like
At least four nonclassical (unconventional) protein secretion pathways have been described.[3] They include:
- direct protein translocation across the plasma membrane likely through membrane transport proteins
- blebbing
- lysosomal secretion
- release via exosomes derived from multivesicular bodies
In addition, proteins can be released from cells by mechanical or physiological wounding[4] and through non-lethal, transient oncotic pores in the plasma membrane induced by washing cells with serum-free media or buffers.[5]
In human tissues
Many
In gram-negative bacteria
Secretion is not unique to eukaryotes – it is also present in bacteria and archaea as well.
Type I secretion system (T1SS or TOSS)
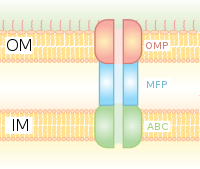
Type I secretion is a chaperone dependent secretion system employing the Hly and Tol gene clusters. The process begins as a leader sequence on the protein to be secreted is recognized by HlyA and binds HlyB on the membrane. This signal sequence is extremely specific for the ABC transporter. The HlyAB complex stimulates HlyD which begins to uncoil and reaches the outer membrane where TolC recognizes a terminal molecule or signal on HlyD. HlyD recruits TolC to the inner membrane and HlyA is excreted outside of the outer membrane via a long-tunnel protein channel.
Type I secretion system transports various molecules, from ions, drugs, to proteins of various sizes (20 – 900 kDa). The molecules secreted vary in size from the small Escherichia coli peptide colicin V, (10 kDa) to the Pseudomonas fluorescens cell adhesion protein LapA of 520 kDa.[7] The best characterized are the RTX toxins and the lipases. Type I secretion is also involved in export of non-proteinaceous substrates like cyclic β-glucans and polysaccharides.

Type II secretion system (T2SS)
Proteins secreted through the type II system, or main terminal branch of the general secretory pathway, depend on the Sec or Tat system for initial transport into the periplasm. Once there, they pass through the outer membrane via a multimeric (12–14 subunits) complex of pore forming secretin proteins. In addition to the secretin protein, 10–15 other inner and outer membrane proteins compose the full secretion apparatus, many with as yet unknown function. Gram-negative type IV pili use a modified version of the type II system for their biogenesis, and in some cases certain proteins are shared between a pilus complex and type II system within a single bacterial species.
Type III secretion system (T3SS or TTSS)
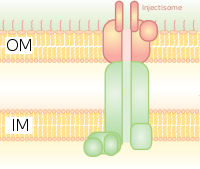
It is homologous to the basal body in bacterial flagella. It is like a molecular syringe through which a bacterium (e.g. certain types of Salmonella, Shigella, Yersinia, Vibrio) can inject proteins into eukaryotic cells. The low Ca2+ concentration in the cytosol opens the gate that regulates T3SS. One such mechanism to detect low calcium concentration has been illustrated by the lcrV (Low Calcium Response) antigen utilized by Yersinia pestis, which is used to detect low calcium concentrations and elicits T3SS attachment. The Hrp system in plant pathogens inject harpins and pathogen effector proteins through similar mechanisms into plants. This secretion system was first discovered in Yersinia pestis and showed that toxins could be injected directly from the bacterial cytoplasm into the cytoplasm of its host's cells rather than simply be secreted into the extracellular medium.[8]
Type IV secretion system (T4SS or TFSS)
| T4SS | |||||||||
|---|---|---|---|---|---|---|---|---|---|
TCDB 3.A.7 | | ||||||||
| OPM superfamily | 215 | ||||||||
| OPM protein | 3jqo | ||||||||
| |||||||||
It is homologous to
Protein members of this family are components of the type IV secretion system. They mediate
Function
The Type IV secretion system (T4SS) is the general mechanism by which bacterial cells secrete or take up macromolecules. Their precise mechanism remains unknown. T4SS is encoded on
Structure
As shown in the above figure, TraC, in particular consists of a three helix bundle and a loose globular appendage.[13]
Interactions
T4SS has two effector proteins: firstly, ATS-1, which stands for Anaplasma translocated substrate 1, and secondly AnkA, which stands for ankyrin repeat domain-containing protein A. Additionally, T4SS coupling proteins are VirD4, which bind to VirE2.[15]
Type V secretion system (T5SS)
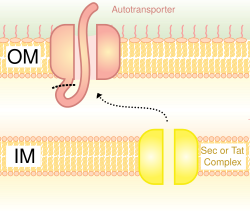
Also called the autotransporter system,
Type VI secretion system (T6SS)
Type VI secretion systems were originally identified in 2006 by the group of John Mekalanos at the Harvard Medical School (Boston, USA) in two bacterial pathogens, Vibrio cholerae and Pseudomonas aeruginosa.[18][19] These were identified when mutations in the Hcp and VrgG genes in Vibrio cholerae led to decreased virulence and pathogenicity. Since then, Type VI secretion systems have been found in a quarter of all proteobacterial genomes, including animal, plant, human pathogens, as well as soil, environmental or marine bacteria.[20][21] While most of the early studies of Type VI secretion focused on its role in the pathogenesis of higher organisms, more recent studies suggested a broader physiological role in defense against simple eukaryotic predators and its role in inter-bacteria interactions.[22][23] The Type VI secretion system gene clusters contain from 15 to more than 20 genes, two of which, Hcp and VgrG, have been shown to be nearly universally secreted substrates of the system. Structural analysis of these and other proteins in this system bear a striking resemblance to the tail spike of the T4 phage, and the activity of the system is thought to functionally resemble phage infection.[24]
Release of outer membrane vesicles
In addition to the use of the multiprotein complexes listed above, Gram-negative bacteria possess another method for release of material: the formation of
In gram-positive bacteria
In some Staphylococcus and Streptococcus species, the accessory secretory system handles the export of highly repetitive adhesion glycoproteins.
See also
- Bacterial effector protein
- Bacterial outer membrane vesicles
- Host–pathogen interaction
- Membrane vesicle trafficking
- Secretomics
- Secretory proteins
- Secretor status
References
- PMID 22659300.
- PMID 16563225.
- PMID 18590485.
- PMID 14570587.
- PMID 22008609.
- ISBN 978-1-904455-42-4.[page needed]
- PMID 24837291.
- ISBN 1-55581-171-X[page needed]
- S2CID 5721063.
- PMID 15035043.
- PMID 16153176.
- PMID 15546668.
- ^ PMID 14673074.
- PMID 12855161.
- PMID 20670295.
- S2CID 2708575.
- PMID 17482513.
- PMID 16432199.
- PMID 16763151.
- PMID 18289922.
- PMID 18617888.
- PMID 20961764.
- PMID 23542428.
- PMID 22746332.
- PMID 16291643.
- PMID 17163978.
- ^ Z. Esna Ashari, N. Dasgupta, K. Brayton & S. Broschat, “An optimal set of features for predicting type IV secretion system effector proteins for a subset of species based on a multi-level feature selection approach”, PLOS ONE Journal, 2018, 13, e0197041. (doi.org/10.1371/journal.pone.0197041.)
Further reading
- Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P, eds. (2002). "Search: Secretion". Molecular Biology of the Cell (4th ed.). New York: Garland Science. ISBN 978-0-8153-3218-3.
- White D (2000). The Physiology and Biochemistry of Prokaryotes (2nd ed.). Oxford University Press. ISBN 978-0-19-512579-5.
- Avon D. "Home page". Cells alive!.
External links
- Secretions at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- T5SS / Autotransporter illustration at Uni Münster
