Selenium dioxide

| |
| |

| |
| Names | |
|---|---|
| Other names
Selenium(IV) oxide
Selenous anhydride | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.028.358 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 3283 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| SeO2 | |
| Molar mass | 110.96 g/mol |
| Appearance | White crystals, turn slightly pink with trace decomposition[1] |
| Odor | rotten radishes |
| Density | 3.954 g/cm3, solid |
| Melting point | 340 °C (644 °F; 613 K) (sealed tube) |
| Boiling point | 350 °C (662 °F; 623 K) subl. |
| 38.4 g/100 mL (20 °C) 39.5 g/100 ml (25 °C) 82.5 g/100 mL (65 °C) | |
| Solubility | soluble in benzene |
| Solubility in ethanol | 6.7 g/100 mL (15 °C) |
| Solubility in acetone | 4.4 g/100 mL (15 °C) |
| Solubility in acetic acid | 1.11 g/100 mL (14 °C) |
| Solubility in methanol | 10.16 g/100 mL (12 °C) |
| Vapor pressure | 1.65 kPa (70 °C) |
| Acidity (pKa) | 2.62; 8.32 |
| −27.2·10−6 cm3/mol | |
Refractive index (nD)
|
> 1.76 |
| Structure | |
| see text | |
| trigonal (Se) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Toxic by ingestion and inhalation[2] |
| GHS labelling: | |
  
| |
| Danger | |
| H301, H331, H373, H410 | |
| P260, P261, P264, P270, P271, P273, P301+P310, P304+P340, P311, P314, P321, P330, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LCLo (lowest published)
|
5890 mg/m3 (rabbit, 20 min) 6590 mg/m3 (goat, 10 min) 6590 mg/m3 (sheep, 10 min)[3] |
| Safety data sheet (SDS) | ICSC 0946 |
| Related compounds | |
Other anions
|
Selenium disulfide |
Other cations
|
Sulfur dioxide Tellurium dioxide |
| Selenium trioxide | |
Related compounds
|
Selenous acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Selenium dioxide is the chemical compound with the formula SeO2. This colorless solid is one of the most frequently encountered compounds of selenium. It is used in making specialized glasses as well as a reagent in organic chemistry.[4]
Properties
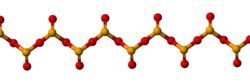
Solid SeO2 is a one-dimensional
The solid sublimes readily. At very low concentrations the vapour has a revolting odour, resembling decayed horseradishes. At higher concentrations the vapour has an odour resembling horseradish sauce and can burn the nose and throat on inhalation. Whereas SO2 tends to be molecular and SeO2 is a one-dimensional chain, TeO2 is a cross-linked polymer.[5]
SeO2 is considered an acidic oxide: it dissolves in water to form selenous acid.[6] Often the terms selenous acid and selenium dioxide are used interchangeably. It reacts with base to form selenite salts containing the SeO2−
3 anion. For example, reaction with sodium hydroxide produces sodium selenite:
- SeO2 + 2 NaOH → Na2SeO3 + H2O
Preparation
Selenium dioxide is prepared by oxidation of selenium by burning in air or by reaction with nitric acid or hydrogen peroxide, but perhaps the most convenient preparation is by the dehydration of selenous acid.
- 2 H2O2 + Se → SeO2 + 2 H2O
- 3 Se + 4 HNO3 + H2O → 3 H2SeO3 + 4 NO
- H2SeO3 ⇌ SeO2 + H2O
Occurrence
The natural form of selenium dioxide, downeyite, is a very rare mineral. It is only found at a small number of burning coal banks, where it forms around vents created from escaping gasses.[8]
Uses
Organic synthesis
SeO2 is an important reagent in

This can be described more generally as;
- R2C=CR'-CHR"2 + [O] → R2C=CR'-C(OH)R"2
where R, R', R" may be
Selenium dioxide can also be used to synthesize 1,2,3-selenadiazoles from acylated hydrazone derivatives.[12]
As a colorant
Selenium dioxide imparts a red colour to glass. It is used in small quantities to counteract the colour due to iron impurities and so to create (apparently) colourless glass. In larger quantities, it gives a deep ruby red colour.
Selenium dioxide is the active ingredient in some cold-bluing solutions.
It was also used as a toner in
Safety
Selenium is an
References
- ^ "Safety data sheet: Selenium dioxide" (PDF). integrachem.com. 27 March 2015. Retrieved 2022-12-02.
- ^ "Selenium dioxide safety and hazards". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-12-02.
- ^ "Selenium compounds (as Se)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ISBN 978-0-470-84289-8.
- ^ ISBN 9780854043668
- ^ ISBN 0-12-352651-5
- ISSN 0022-2852.
- ^ Finkelman, Robert B.; Mrose, Mary E. (1977). "Downeyite, the first verified natural occurrence of SeO2" (PDF). American Mineralogist. 62: 316–320.
- ^ Ronzio, A. R.; Waugh, T. D. (1955). "Glyoxal Bisulfite". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 438. - ^ a b Hach, C. C. Banks, C. V.; Diehl, H. (1963). "1,2-Cyclohexanedione Dioxime". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 4, p. 229. - doi:10.15227/orgsyn.056.0025).
{{cite journal}}: CS1 maint: multiple names: authors list (link - .

