Selenocysteine
| |
| |
| Names | |
|---|---|
| IUPAC name
Selenocysteine
| |
| Systematic IUPAC name
3-Selanyl-L-alanine (semisystematic name)
2-Amino-3-selanylpropanoic acid (fully systematic name) | |
| Other names
L-Selenocysteine; Selenium-cysteine
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.236.386 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3H7NO2Se | |
| Molar mass | 168.065 g·mol−1 |
| Properties | |
| Acidity (pKa) | 5.24,[2] 5.43[3] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Selenocysteine (symbol Sec or U,.
Selenocysteine is present in several
Selenocysteine was discovered in 1974[7] by biochemist Thressa Stadtman at the National Institutes of Health.[8]
Chemistry
Selenocysteine is the Se-analogue of cysteine. It is rarely encountered outside of living tissue (and is not available commercially) because it is very susceptible to air-oxidation. More common is the oxidized derivative
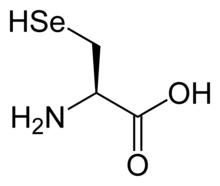
Structure
Selenocysteine has the same structure as cysteine, but with an atom of selenium taking the place of the usual sulfur. It has a selenol group. Like other natural proteinogenic amino acids, cysteine and selenocysteine have L chirality in the older D/L notation based on homology to D- and L-glyceraldehyde. In the newer R/S system of designating chirality, based on the atomic numbers of atoms near the asymmetric carbon, they have R chirality, because of the presence of sulfur or selenium as a second neighbor to the asymmetric carbon. The remaining chiral amino acids, having only lighter atoms in that position, have S chirality.)
Proteins which contain a selenocysteine residue are called selenoproteins. Most selenoproteins contain a single selenocysteine residue. Selenoproteins that exhibit catalytic activity are called selenoenzymes.[11]
Biology
Selenocysteine has a lower reduction potential than cysteine. These properties make it very suitable in proteins that are involved in antioxidant activity.[12]
Although it is found in the
Unlike the other amino acids, no free pool of selenocysteine exists in the cell. Its high reactivity would cause damage to cells.
The primary and secondary structure of selenocysteine-specific tRNA, tRNASec, differ from those of standard tRNAs in several respects, most notably in having an 8-base-pair (bacteria) or 10-base-pair (eukaryotes)[Archaea?] acceptor stem, a long variable region arm, and substitutions at several well-conserved base positions. The selenocysteine tRNAs are initially charged with serine by seryl-tRNA ligase, but the resulting Ser-tRNASec is not used for translation because it is not recognised by the normal translation elongation factor (EF-Tu in bacteria, eEF1A in eukaryotes).[Archaea?]
Rather, the tRNA-bound seryl residue is converted to a selenocysteine residue by the pyridoxal phosphate-containing enzyme selenocysteine synthase. In eukaryotes and archaea, two enzymes are required to convert tRNA-bound seryl residue into tRNA selenocysteinyl residue: PSTK (O-phosphoseryl-tRNA[Ser]Sec kinase) and selenocysteine synthase.[20][21] Finally, the resulting Sec-tRNASec is specifically bound to an alternative translational elongation factor (SelB or mSelB (or eEFSec)), which delivers it in a targeted manner to the ribosomes translating mRNAs for selenoproteins. The specificity of this delivery mechanism is brought about by the presence of an extra protein domain (in bacteria, SelB) or an extra subunit (SBP2 for eukaryotic mSelB/eEFSec)[Archaea?] which bind to the corresponding RNA secondary structures formed by the SECIS elements in selenoprotein mRNAs.
Selenocysteine is decomposed by the enzyme selenocysteine lyase into L-alanine and selenide.[22]
As of 2021[update], 136 human proteins (in 37 families) are known to contain selenocysteine (selenoproteins).[23]
Selenocysteine derivatives γ-glutamyl-Se-methylselenocysteine and
Applications
Biotechnological applications of selenocysteine include use of 73Se-labeled Sec (half-life of 73Se = 7.2 hours) in
See also
- Pyrrolysine, another amino acid not in the basic set of 20.
- Selenomethionine, another selenium-containing amino acid, which is randomly substituted for methionine.
References
- ^ Merck Index, 12th Edition, 8584
- PMID 6076213.
- ^ PMID 27748600.
- ^ "Nomenclature and Symbolism for Amino Acids and Peptides". IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. Archived from the original on 9 October 2008. Retrieved 5 March 2018.
- .
- ^ PMID 15967579.
- ^ "Stadtman Pioneer of Selenium Biochemistry - Office of NIH History and Stetten Museum". history.nih.gov. Retrieved 2023-04-06.
- S2CID 84982102.
- PMID 26090162.
- PMID 26949981.
- S2CID 32351033.
- S2CID 11002236.
- PMID 15069108.
- PMID 1838215.
- S2CID 976337.
- PMID 20488192.
- ISBN 978-0-387-89381-5.
- PMID 8344267.
- PMID 7959166.
- PMID 17194211.
- PMID 17142313.
- PMID 24987004.
- ISBN 978-0-85404-190-9.
Further reading
- Zinoni F, Birkmann A, Stadtman TC, Böck A (July 1986). "Nucleotide sequence and expression of the selenocysteine-containing polypeptide of formate dehydrogenase (formate-hydrogen-lyase-linked) from Escherichia coli". Proceedings of the National Academy of Sciences of the United States of America. 83 (13): 4650–4. PMID 2941757.
- Zinoni F, Birkmann A, Leinfelder W, Böck A (May 1987). "Cotranslational insertion of selenocysteine into formate dehydrogenase from Escherichia coli directed by a UGA codon". Proceedings of the National Academy of Sciences of the United States of America. 84 (10): 3156–60. PMID 3033637.
- Cone JE, Del Río RM, Davis JN, Stadtman TC (August 1976). "Chemical characterization of the selenoprotein component of clostridial glycine reductase: identification of selenocysteine as the organoselenium moiety". Proceedings of the National Academy of Sciences of the United States of America. 73 (8): 2659–63. PMID 1066676.
- Fenyö D, Beavis RC (February 2016). "Selenocysteine: Wherefore Art Thou?". Journal of Proteome Research. 15 (2): 677–8. PMID 26680273.
External links
- For the first time a Uruguayan scientist finds selenocysteine in fungi Archived 2019-02-22 at the Wayback Machine (in Spanish)

