Self-assembled monolayer
| Part of a series of articles on |
| Molecular self-assembly |
|---|
 |
Self-assembled monolayers (SAM) of organic molecules are molecular assemblies formed spontaneously on surfaces by
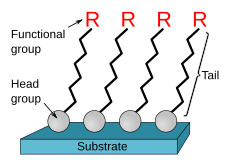
SAMs are created by the chemisorption of "head groups" onto a substrate from either the vapor or liquid phase[7][8] followed by a slow organization of "tail groups".[9] Initially, at small molecular density on the surface, adsorbate molecules form either a disordered mass of molecules or form an ordered two-dimensional "lying down phase",[7] and at higher molecular coverage, over a period of minutes to hours, begin to form three-dimensional crystalline or semicrystalline structures on the substrate surface.[10] The "head groups" assemble together on the substrate, while the tail groups assemble far from the substrate. Areas of close-packed molecules nucleate and grow until the surface of the substrate is covered in a single monolayer.
Adsorbate molecules adsorb readily because they lower the surface free-energy of the substrate
Types
Selecting the type of head group depends on the application of the SAM.
Preparation
Metal substrates for use in SAMs can be produced through physical vapor deposition techniques, electrodeposition or electroless deposition.[1] Thiol or selenium SAMs produced by adsorption from solution are typically made by immersing a substrate into a dilute solution of alkane thiol in ethanol, though many different solvents can be used[1] besides use of pure liquids.[16] While SAMs are often allowed to form over 12 to 72 hours at room temperature,[10][19] SAMs of alkanethiolates form within minutes.[20][21] Special attention is essential in some cases, such as that of dithiol SAMs to avoid problems due to oxidation or photoinduced processes, which can affect terminal groups and lead to disorder and multilayer formation.[22][23] In this case appropriate choice of solvents, their degassing by inert gasses and preparation in the absence of light is crucial[22][23] and allows formation of "standing up" SAMs with free –SH groups. Self-assembled monolayers can also be adsorbed from the vapor phase.[8][24] In some cases when obtaining an ordered assembly is difficult or when different density phases need to be obtained substitutional self-assembly is used. Here one first forms the SAM of a given type of molecules, which give rise to ordered assembly and then a second assembly phase is performed (e.g. by immersion into a different solution). This method has also been used to give information on relative binding strengths of SAMs with different head groups and more generally on self-assembly characteristics.[18][25]
Characterization
The thicknesses of SAMs can be measured using
Defects
Defects due to both external and intrinsic factors may appear. External factors include the cleanliness of the substrate, method of preparation, and purity of the adsorbates.[1][10] SAMs intrinsically form defects due to the thermodynamics of formation, e.g. thiol SAMs on gold typically exhibit etch pits (monatomic vacancy islands) likely due to extraction of adatoms from the substrate and formation of adatom-adsorbate moieties. Recently, a new type of fluorosurfactants have found that can form nearly perfect monolayer on gold substrate due to the increase of mobility of gold surface atoms.[30][31][32]
Nanoparticle properties
The structure of SAMs is also dependent on the curvature of the substrate. SAMs on nanoparticles, including
Kinetics
There is evidence that SAM formation occurs in two steps: an initial fast step of adsorption and a second slower step of monolayer organization. Adsorption occurs at the liquid–liquid, liquid–vapor, and liquid-solid interfaces. The transport of molecules to the surface occurs due to a combination of diffusion and convective transport. According to the Langmuir or Avrami kinetic model the rate of deposition onto the surface is proportional to the free space of the surface.[7]
Where θ is the proportional amount of area deposited and k is the rate constant. Although this model is robust it is only used for approximations because it fails to take into account intermediate processes.
Once the molecules are at the surface the self-organization occurs in three phases:[7]
- 1. A low-density phase with random dispersion of molecules on the surface.
- 2. An intermediate-density phase with conformational disordered molecules or molecules lying flat on the surface.
- 3. A high-density phase with close-packed order and molecules standing normal to the substrate's surface.
The phase transitions in which a SAM forms depends on the temperature of the environment relative to the triple point temperature, the temperature in which the tip of the low-density phase intersects with the intermediate-phase region. At temperatures below the triple point the growth goes from phase 1 to phase 2 where many islands form with the final SAM structure, but are surrounded by random molecules. Similar to nucleation in metals, as these islands grow larger they intersect forming boundaries until they end up in phase 3, as seen below.[7]
At temperatures above the triple point the growth is more complex and can take two paths. In the first path the heads of the SAM organize to their near final locations with the tail groups loosely formed on top. Then as they transit to phase 3, the tail groups become ordered and straighten out. In the second path the molecules start in a lying down position along the surface. These then form into islands of ordered SAMs, where they grow into phase 3, as seen below.[7]
The nature in which the tail groups organize themselves into a straight ordered monolayer is dependent on the inter-molecular attraction, or
Many of the SAM properties, such as thickness, are determined in the first few minutes. However, it may take hours for defects to be eliminated via annealing and for final SAM properties to be determined.[7][10] The exact kinetics of SAM formation depends on the adsorbate, solvent and substrate properties. In general, however, the kinetics are dependent on both preparations conditions and material properties of the solvent, adsorbate and substrate.[7] Specifically, kinetics for adsorption from a liquid solution are dependent on:[1]
- Temperature – room-temperature preparation improves kinetics and reduces defects.
- Concentration of adsorbate in the solution – low concentrations require longer immersion times[1][10] and often create highly crystalline domains.[10]
- Purity of the adsorbate – impurities can affect the final physical properties of the SAM
- Dirt or contamination on the substrate – imperfections can cause defects in the SAM
The final structure of the SAM is also dependent on the chain length and the structure of both the adsorbate and the substrate. Steric hindrance and metal substrate properties, for example, can affect the packing density of the film,[1][10] while chain length affects SAM thickness.[12] Longer chain length also increases the thermodynamic stability.[1]
Patterning
1. Locally attract
This first strategy involves locally depositing
- Micro-contact printing or soft lithography is analogous to printing ink with a rubber stamp. The SAM molecules are inked onto a pre-shaped elastomeric stamp with a solvent and transferred to the substrate surface by stamping. The SAM solution is applied to the entire stamp but only areas that make contact with the surface allow transfer of the SAMs. The transfer of the SAMs is a complex diffusion process that depends on the type of molecule, concentration, duration of contact, and pressure applied. Typical stamps use PDMS because its elastomeric properties, E = 1.8 MPa, allow it to fit the contour of micro surfaces and its low surface energy, γ = 21.6 dyn/cm². This is a parallel process and can thus place nanoscale objects over a large area in a short time.[1]
- atomic force microscope to transfer molecules on the tip to a substrate. Initially the tip is dipped into a reservoir with an ink. The ink on the tip evaporates and leaves the desired molecules attached to the tip. When the tip is brought into contact with the surface a water meniscus forms between the tip and the surface resulting in the diffusion of molecules from the tip to the surface. These tips can have radii in the tens of nanometers, and thus SAM molecules can be very precisely deposited onto a specific location of the surface. This process was discovered by Chad Mirkin and co-workers at Northwestern University.[34]
2. Locally remove
The locally remove strategy begins with covering the entire surface with a SAM. Then individual SAM molecules are removed from locations where the deposition of
- The scanning tunneling microscope can remove SAM molecules in many different ways. The first is to remove them mechanically by dragging the tip across the substrate surface. This is not the most desired technique as these tips are expensive and dragging them causes a lot of wear and reduction of the tip quality. The second way is to degrade or desorb the SAM molecules by shooting them with an electron beam. The scanning tunneling microscope can also remove SAMs by field desorption and field enhanced surface diffusion.[33]
- Atomic force microscope
- The most common use of this technique is to remove the SAM molecules in a process called shaving, where the atomic force microscope can also remove SAM molecules by local oxidation nanolithography.[33]
- Ultraviolet irradiation
- In this process, UV light is projected onto the surface with a SAM through a pattern of apertures in a chromium film. This leads to photo oxidation of the SAM molecules. These can then be washed away in a polar solvent. This process has 100 nm resolutions and requires exposure time of 15–20 minutes.[1]
3. Modify tail groups
The final strategy focuses not on the deposition or removal of SAMS, but the modification of terminal groups. In the first case the terminal group can be modified to remove functionality so that SAM molecule will be inert. In the same regards the terminal group can be modified to add functionality[35] so it can accept different materials or have different properties than the original SAM terminal group. The major techniques that use this strategy are:[33]
- Focused electron beam and ultraviolet irradiation
- Exposure to electron beams and UV light changes the terminal group chemistry. Some of the changes that can occur include the cleavage of bonds, the forming of double carbon bonds, cross-linking of adjacent molecules, fragmentation of molecules, and confromational disorder.[1]
- Atomic force microscope
- A conductive AFM tip can create an electrochemical reaction that can change the terminal group.[33]
Applications
Thin-film SAMs
SAMs are an inexpensive and versatile surface coating for applications including control of wetting and adhesion,
Thin film SAMs can also be placed on
Patterned SAMs
Photolithographic methods are useful in patterning SAMs.
Metal organic superlattices
There has been considerable interest in use of SAMs for new materials e.g. via formation of two- or three-dimensional metal organic superlattices by assembly of SAM capped nanoparticles[43] or layer by layer SAM-nanoparticle arrays using dithiols.[44] A detailed review on this subject using dithiols is given by Hamoudi and Esaulov[45]
References
- ^ PMID 15826011.
- .
- PMID 19746490.
- .
- PMID 12792937.
- S2CID 97346507.
- ^ PMID 11326061.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ .
- ^ Wnek, Gary, Gary L. Bowlin (2004). Encyclopedia of Biomaterials and Biomedical Engineering. Informa Healthcare. pp. 1331–1333.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b c d e f g h i j Vos, Johannes G., Robert J. Forster, Tia E. Keyes (2003). Interfacial Supramolecular Assemblies. Wiley. pp. 88–94.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b Madou, Marc (2002). Fundamentals of Microfabrication: The Science of Miniaturization. CRC. pp. 62–63.
- ^ a b c Kaifer, Angel (2001). Supramolecular Electrochemistry. Coral Gables. Wiley VCH. pp. 191–193.
- ^ Saliterman, Steven (2006). Self-assembled monolayers (SAMs). Fundamentals of BioMEMS and Medical Microdevices. SPIE Press. pp. 94–96.
- S2CID 24880913.
- PMID 17274618.
- ^ PMID 17451727.
- doi:10.1021/la00016a001.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ doi:10.1021/ja00155a016.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ Wysocki. "Self-Assembled Monolayers (SAMs) as Collision Surfaces for Ion Activation" (PDF). Archived from the original (PDF) on 22 June 2010.
- ^ .
- ^ from the original on 23 September 2017.
- ^ PMID 20199099.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ PMID 19015788.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ doi:10.1021/jp1044157.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- .
- .
- ^ .
- PMID 15875853.
- PMID 18980356.
- PMID 20058870.
- PMID 21214202.
- ^ PMID 21794185.
- S2CID 27011581.
- PMID 20845943.
- ISSN 0002-7863.
- PMID 21167979.
- PMID 17177439.
- .
- PMID 19370233.
- PMID 16519501.
- PMID 16365640.
- S2CID 4420426.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - doi:10.1021/jp983836l.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - S2CID 124513678.
Further reading
- Sagiv, J.; Polymeropoulos, E.E. (1978). "Adsorbed monolayers - molecular-organization and electrical-properties". Berichte der Bunsengesellschaft für Physikalische Chemie. 82 (9): 883. .
- I. Rubinstein, E. Sabatani, R. Maoz and J. Sagiv, Organized Monolayers on Gold Electrodes, in Electrochemical Sensors for Biomedical Applications, C.K.N. Li (Ed.), The Electrochemical Society 1986: 175.
- Faucheux, N.; Schweiss, R.; Lützow, K.; Werner, C.; Groth, T. (2004). "Self-assembled monolayers with different terminating groups as model substrates for cell adhesion studies". Biomaterials. 25 (14): 2721–2730. PMID 14962551.
- Wasserman, S. R.; Tao, Y. T.; Whitesides, G. M. (1989). "Structure and Reactivity of Alkylsiloxane Monolayers Formed by Reaction of Alkyltrichlorosilanes on Silicon Substrates". Langmuir. 5 (4): 1074–1087. .
- Hoster, H.E.; Roos, M.; Breitruck, A.; Meier, C.; Tonigold, K.; Waldmann, T.; Ziener, U.; PMID 17914848.
- Sigma-Aldrich "Material Matters", Molecular Self-Assembly

