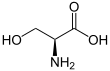
Serine
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Serine
| |||
| Other names
2-Amino-3-hydroxypropanoic acid
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider | |||
| DrugBank |
| ||
ECHA InfoCard
|
100.000.250 | ||
| EC Number |
| ||
IUPHAR/BPS |
| ||
| KEGG | |||
PubChem CID
|
|||
| UNII |
| ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties[2] | |||
| C3H7NO3 | |||
| Molar mass | 105.093 g·mol−1 | ||
| Appearance | white crystals or powder | ||
| Density | 1.603 g/cm3 (22 °C) | ||
| Melting point | 246 °C (475 °F; 519 K) decomposes | ||
| soluble | |||
| Acidity (pKa) | 2.21 (carboxyl), 9.15 (amino)[1] | ||
| Supplementary data page | |||
| Serine (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Serine (symbol Ser or S)
Occurrence

This compound is one of the
Biosynthesis
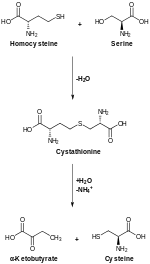
The biosynthesis of serine starts with the
In bacteria such as E. coli these enzymes are encoded by the genes serA (EC 1.1.1.95), serC (EC 2.6.1.52), and serB (EC 3.1.3.3).[10]

Glycine biosynthesis:
4, and mTHF in a reaction catalyzed by glycine synthase.[8]
Synthesis and reactions
Industrially, L-serine is produced from glycine and methanol catalyzed by
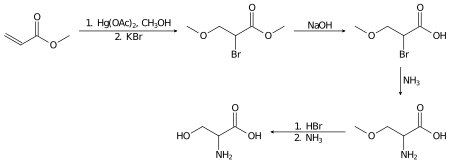
Racemic serine can be prepared in the laboratory from methyl acrylate in several steps:[13]
Hydrogenation of serine gives the diol serinol:
- HOCH2CH(NH2)CO2H + 2 H2 → HOCH2CH(NH2)CH2OH + 2 H2O
Biological function
Metabolic
Serine is important in
Signaling
D-Serine, synthesized in neurons by
Ligands
D-serine was thought to exist only in bacteria until relatively recently; it was the second D amino acid discovered to naturally exist in humans, present as a signaling molecule in the brain, soon after the discovery of
Gustatory sensation
Pure D-serine is an off-white crystalline powder with a very faint musty aroma. D-Serine is sweet with an additional minor sour taste at medium and high concentrations.[23]
Clinical significance
Serine deficiency disorders are rare defects in the biosynthesis of the amino acid L-serine. At present three disorders have been reported:
- 3-phosphoglycerate dehydrogenase deficiency
- 3-phosphoserine phosphatase deficiency
- Phosphoserine aminotransferase deficiency
These enzyme defects lead to severe neurological symptoms such as congenital microcephaly and severe psychomotor retardation and in addition, in patients with 3-phosphoglycerate dehydrogenase deficiency to intractable seizures. These symptoms respond to a variable degree to treatment with L-serine, sometimes combined with glycine.[24][25] Response to treatment is variable and the long-term and functional outcome is unknown. To provide a basis for improving the understanding of the epidemiology, genotype/phenotype correlation and outcome of these diseases their impact on the quality of life of patients, as well as for evaluating diagnostic and therapeutic strategies a patient registry was established by the noncommercial International Working Group on Neurotransmitter Related Disorders (iNTD).[26]
Besides disruption of serine biosynthesis, its transport may also become disrupted. One example is
Research for therapeutic use
The classification of L-serine as a non-essential amino acid has come to be considered as conditional, since vertebrates such as humans cannot always synthesize optimal quantities over entire lifespans.[27] Safety of L-serine has been demonstrated in an FDA-approved human phase I clinical trial with Amyotrophic Lateral Sclerosis, ALS, patients (ClinicalTrials.gov identifier: NCT01835782),[28][29] but treatment of ALS symptoms has yet to be shown. A 2011 meta-analysis found adjunctive sarcosine to have a medium effect size for negative and total symptoms of schizophrenia.[30] There also is evidence that L‐serine could acquire a therapeutic role in diabetes.[31]
D-Serine is being studied in rodents as a potential treatment for schizophrenia.[32] D-Serine also has been described as a potential biomarker for early Alzheimer's disease (AD) diagnosis, due to a relatively high concentration of it in the cerebrospinal fluid of probable AD patients.[33] D-serine, which is made in the brain, has been shown to work as an antagonist/inverse co-agonist of t-NMDA receptors mitigating neuron loss in an animal model of temporal lobe epilepsy.[34]
D-Serine has been theorized as a potential treatment for sensorineural hearing disorders such as hearing loss and tinnitus.[35]
See also
- Isoserine
- Homoserine (isothreonine)
- Serine octamer cluster
References
- ^ Dawson, R.M.C., et al., Data for Biochemical Research, Oxford, Clarendon Press, 1959.
- ISBN 0-8493-0462-8.
- ^ "Nomenclature and Symbolism for Amino Acids and Peptides". IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. Archived from the original on 9 October 2008. Retrieved 5 March 2018.
- .
- ^ Cramer E (1865). "Ueber die Bestandtheile der Seide" [On the constituents of silk]. Journal für praktische Chemie (in German). 96: 76–98. Serine is named on p. 93: "Ich werde den in Frage stehenden Körper unter dem Namen Serin beschreiben." (I will describe the body [i.e., substance] in question by the name "serine".)
- .
- ^ "Serine". The Columbia Encyclopedia 6th ed. encyclopedia.com. Retrieved 22 October 2012.
- ^ ISBN 978-0-7167-1843-7.
- ^ KEGG EC 3.1.3.3 etc.
- ^ Uniprot: serB
- ISBN 1-57259-153-6.
- ISBN 978-3527306732.)
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link - .
- PMID 11292060.
- PMID 30787885.
D-Serine is more potent than glycine as a coagonist at the NMDA receptor, has a regional distribution in the brain that is similar to that of NMDA receptors and appears to be more closely associated with synaptic NMDA receptors than glycine (which is more closely associated with non-synaptic NMDA receptors).
- PMID 32518273.
D-Serine is functionally a more potent activator of synaptic NMDARs than glycine, and mounting evidence suggests that it serves as the major NMDAR co-agonist in limbic brain regions implicated in neuropsychiatric disorders.
- S2CID 23158971.
- PMID 37437688.
- PMID 10781100.
- S2CID 45669104.
- PMID 18809793.
- PMID 20170643.
- S2CID 17671611.
- S2CID 25013468.
- PMID 19963421.
- ^ "Patient registry".
- S2CID 20271849.
- PMID 24086518.
- S2CID 4584977.
- S2CID 207299820.
- PMID 31344283.
- PMID 23729812.
- PMID 25942042.
- PMID 33009404.
- PMID 34975405.