Sex ratio
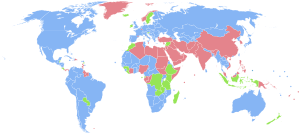
| Countries with more males than females. Countries with the same number of males and females (accounting that the ratio has 3 significant figures, i.e., 1.00 males to 1.00 females). Countries with more females than males. No data |
A sex ratio is the
The human sex ratio is of particular interest to anthropologists and demographers. In human societies, sex ratios at birth may be considerably skewed by factors such as the age of mother at birth[5] and by sex-selective abortion and infanticide. Exposure to pesticides and other environmental contaminants may be a significant contributing factor as well.[6] As of 2024, the global sex ratio at birth is estimated at 107 boys to 100 girls (1,000 boys per 934 girls).[7] By old age, the sex ratio reverses, with 81 older men for every 100 older women; across all ages, the global population is nearly balanced, with 101 males for every 100 females.[7]
Types
In most species, the sex ratio varies according to the age profile of the population.[8]
It is generally divided into four subdivisions:
- primary sex ratio — ratio at fertilization
- secondary sex ratio — ratio at birth
- tertiary sex ratio — ratio in sexually mature organisms
- The tertiary sex ratio is equivalent to the adult sex ratio (ASR), which is defined as the ratio of adult males to females in a population.[9][10]
- The operational sex ratio (OSR) is the ratio of sexually active males to females in a population, and is therefore derived from a subset of the individuals included when calculating the ASR.[10] Although conceptually distinct, researchers have sometimes equated the ASR with the OSR, particularly in experimental studies of animals where the difference between the two values may not always be readily apparent.[11]
- quaternary sex ratio — ratio in post-reproductive organisms
These definitions can be somewhat subjective since they lack clear boundaries.
Sex ratio theory
Sex ratio theory is a field of academic study which seeks to understand the sex ratios observed in nature from an evolutionary perspective. It continues to be heavily influenced by the work of Eric Charnov.[12] He defines five major questions, both for his book and the field in general (slightly abbreviated here):
- For a dioeciousspecies, what is the equilibrium sex ratio maintained by natural selection?
- For a sequential hermaphrodite, what is the equilibrium sex order and time of sex change?
- For a simultaneous hermaphrodite, what is the equilibrium allocation of resources to male versus female function in each breeding season?
- Under what conditions are the various states of hermaphroditism or dioecy evolutionarily stable? When is a mixture of sexual types stable?
- When does selection favour the ability of an individual to alter its allocation to male versus female function, in response to particular environmental or life history situations?
Biological research mostly concerns itself with sex allocation rather than sex ratio, sex allocation denoting the allocation of energy to either sex. Common research themes are the effects of local mate and resource competition (often abbreviated LMC and LRC, respectively).
Fisher's principle
Fisher's principle (1930)[2] explains why in most species, the sex ratio is approximately 1:1. His argument was summarised by W. D. Hamilton (1967)[3] as follows, assuming that parents invest the same whether raising male or female offspring:
- Suppose male births are less common than female.
- A newborn male then has better mating prospects than a newborn female, and therefore can expect to have more offspring.
- Therefore parents genetically disposed to produce males tend to have more than average numbers of grandchildren born to them.
- Therefore the genes for male-producing tendencies spread, and male births become more common.
- As the 1:1 sex ratio is approached, the advantage associated with producing males dies away.
- The same reasoning holds if females are substituted for males throughout. Therefore 1:1 is the equilibrium ratio.
In modern language, the 1:1 ratio is the evolutionarily stable strategy (ESS).[13] This ratio has been observed in many species, including the bee Macrotera portalis. A study performed by Danforth observed no significant difference in the number of males and females from the 1:1 sex ratio.[14]
Examples in non-human species
Environmental and individual control
Spending equal amounts of resources to produce offspring of either sex is an evolutionarily stable strategy: if the general population deviates from this equilibrium by favoring one sex, one can obtain higher reproductive success with less effort by producing more of the other. For species where the cost of successfully raising one offspring is roughly the same regardless of its sex, this translates to an approximately equal sex ratio.
Several species of reptiles have temperature-dependent sex determination, where incubation temperature of eggs determines the sex of the individual. In the American alligator, for example, females are hatched from eggs incubated between 27.7 to 30 °C (81.9 to 86.0 °F), whereas males are hatched from eggs 32.2 to 33.8 °C (90.0 to 92.8 °F). In this method, however, all eggs in a clutch (20–50) will be of the same sex. In fact, the natural sex ratio of this species is five females to one male.[16]
In birds, mothers can influence the sex of their chicks. In peafowl, maternal body condition can influence the proportion of daughters in the range from 25% to 87%.[17]
Domesticated animals
Traditionally, farmers have discovered that the most economically efficient community of animals will have a large number of females and a very small number of males. A herd of cows with a few bulls or a flock of hens with one rooster are the most economical sex ratios for domesticated livestock.
Dioecious plants secondary sex ratio and amount of pollen
It was found that the amount of fertilizing pollen can influence secondary sex ratio in dioecious plants. Increase in pollen amount leads to decrease in number of male plants in the progeny. This relationship was confirmed on four plant species from three families –
Polyandrous and cooperatively breeding homeotherms
In
Male-biased adult sex ratios have also been shown to correlate with cooperative breeding in mammals such as alpine marmots and wild canids.[26] This correlation may also apply to cooperatively breeding birds,[27] though the evidence is less clear.[24] It is known, however, that both male-biased adult sex ratios[28] and cooperative breeding tend to evolve where caring for offspring is extremely difficult due to low secondary productivity, as in Australia[29] and Southern Africa. It is also known that in cooperative breeders where both sexes are philopatric like the varied sittella,[30] adult sex ratios are equally or more male-biased than in those cooperative species, such as fairy-wrens, treecreepers and the noisy miner[31] where females always disperse.
See also
- Evolution of sex
- Operational sex ratio
- Sex allocation
- Trivers–Willard hypothesis
- XY sex-determination system
Humans:
- Human sex ratio
- List of countries by sex ratio
- Bride kidnapping
- Groom kidnapping
- Demographic transition
- Sex selection
- Sex-selective abortion and infanticide
- Youth bulge
Institutions:
Notes
- CIA World Factbook[1]. Map compiled in 2021, data from 2020.
- ^ a b Fisher, R. A. (1930). The Genetical Theory of Natural Selection. Oxford: Clarendon Press. pp. 141–143 – via Internet Archive.
- ^ PMID 6021675.
- PMID 23807025.
- ^ "Trend Analysis of the sex Ratio at Birth in the United States" (PDF). U.S. Department of Health and Human Services, National Center for Health Statistics.
- Journal of the American Medical Association; April 1, 1998, volume 279(13); pp. 1018-1023
- ^ a b "Field Listing—Sex ratio". CIA Factbook. The Central Intelligence Agency of the United States. Retrieved 2024-04-18.
{{cite web}}: CS1 maint: url-status (link) (2023 estimates) - .
- .
- ^ ISBN 9780521818964. Retrieved 24 December 2022.
- S2CID 8350737.
- ISBN 9780691083124.
- S2CID 4224989.
- .
- S2CID 13412222.
- S2CID 4307265.
- S2CID 53185717.
- ^ Correns С. (1922). "Geschlechtsbestimmung und Zahlenverhaltnis der Geschlechter beim Sauerampfer (Rumex acetosa)". Biologisches Zentralblatt. 42: 465–80.
- ^ Rychlewski J.; Kazlmierez Z. (1975). "Sex ratio in seeds of Rumex acetosa L. as a result of sparse or abundant pollination". Acta Biol Crac Ser Bot. 18: 101–14.
- ^ Correns C. (1928). "Bestimmung, Vererbung und Verteilung des Geschlechter bei den hoheren Pflanzen". Handb. Vererbungswiss. 2: 1–138.
- .
- ^ Riede W. (1925) Beitrage zum Geschlechts- und Anpassungs-problem. "Flora" 18/19
- ^ Kihara H., Hirayoshi J. (1932) Die Geschlechtschromosomen von Humulus japonicus. Sieb. et. Zuce. In: 8th Congr. Jap. Ass. Adv. Sci., p. 363—367 (cit.: Plant Breeding Abstr., 1934, 5, № 3, p. 248, ref. № 768).
- ^ PMID 23481395.
- S2CID 6954828.
- ^ Allainé, Dominique; Brondex, Francine; Graziani, Laurent; Coulon, Jacques and Till-Bottraud, Irène; "Male-biased sex ratio in litters of alpine marmots supports the helper repayment hypothesis"
- S2CID 53165151.
- S2CID 14624385.
- S2CID 39566226.
- .
- .
References
- S2CID 24724998.
- Nishimura K, Jahn GC (1996). "Sex allocation of three solitary ectoparasitic wasp species on bean weevil larvae: sex ratio change with host quality and local mate competition". Journal of Ethology. 14 (1): 27–34. S2CID 10590797.
- S2CID 29326420.
- Rath, R.M., and Mishra A.K. (2005). Techniques for Sex Ratio Analysis. Association of Professional Geographers.
