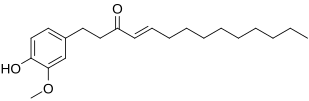
Shogaol

| |
| Names | |
|---|---|
| Preferred IUPAC name
(4E)-1-(4-Hydroxy-3-methoxyphenyl)dec-4-en-3-one | |
Other names
| |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C17H24O3 | |
| Molar mass | 276.376 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
| Shogaol | |
|---|---|
| Heat | Very hot (chemical) |
| Scoville scale | 160,000[1] SHU |
Shogaols are
The name shogaol is derived from the Japanese name for ginger (生姜、shōga).
Shogaol is rated 160,000 SHU on the Scoville scale.[1] When compared to other pungent compounds, shogaol is moderately more pungent than piperine, but less than capsaicin.
| Compound | Scoville Heat Units (SHU) |
|---|---|
| Capsaicin | 16,000,000[3] |
| [6]-Shogaol | 160,000 |
| Piperine | 100,000 |
| [6]-Gingerol | 60,000 |
Shogaols group
[4]-Shogaol, [8]-shogaol, [10]-shogaol, and [12]-shogaol (all found in ginger) together constitute the group shogaols. There also exist in ginger cultivars methylated shogaols: methyl [6]-shogaol and methyl [8]-shogaol, respectively.[4]
Shogaols are artifacts formed during storage or through excess heat, probably created by a dehydration reaction of the gingerols. The ratio of shogaols to gingerols sometimes is taken as an indication of product quality.[5]
Synthesis
A possible synthesis starts with a

References
- ^ S2CID 40241982.
- ^ Harold McGee (2004). On Food and Cooking: The Science and Lore of the Kitchen (2nd ed.). New York: Scribner. pp. 425–426.
- PMID 2039598.
- ^ "Analysis of Chemical Properties of Edible and Medicinal Ginger by Metabolomics Approach : Table 1". Retrieved 3 December 2016.
- ^ NSF International Determination of Gingerols and Shogaols in Zingiber officinale rhizome and powdered extract by High-Performance Liquid Chromatography [full citation needed]
- PMID 24599082.