Silicate
4
In
For diverse manufacturing, technological, and artistic needs, silicates are versatile materials, both natural (such as
Structural principles
In most silicates, silicon atom occupies the center of an idealized tetrahedron whose corners are four oxygen atoms, connected to it by single covalent bonds according to the octet rule.[1] The oxygen atoms, which bears some negative charge, link to other cations (Mn+). This Si-O-M-O-Si linkage is strong and rigid, which properties are manifested in the rock-like silicates. The silicates can be classified according to the length and crosslinking of the silicate anions.
Isolated silicates
Isolated orthosilicate anions have the formula SiO4−
4. A common mineral in this group is olivine ((Mg,Fe)2SiO4).
Two or more silicon atoms can share oxygen atoms in various ways, to form more complex anions, such as pyrosilicate Si
2O6−
7.
Chains

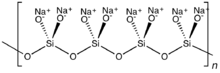
With two shared oxides bound to each silicon, cyclic or polymeric structures can result. The cyclic
In single-chain silicates, which are a type of

Double-chain silicates, the other category of inosilicates, occur when tetrahedra form a double chain (not always but mostly) by sharing two or three oxygen atoms each. Common minerals for this group are
Sheets

In this group, known as
have very weak layers that can be peeled off in sheets.Framework
In a framework silicate, known as a
Silicates with non-tetrahedral silicon
Although the tetrahedron is a common coordination geometry for silicon(IV) compounds, silicon may also occur with higher coordination numbers. For example, in the anion
At very high pressure, such as exists in the majority of the Earth's crust, even SiO2 adopts the six-coordinated octahedral geometry in the mineral
Chemical properties
Silicates with
Reactions
| Part of a series related to |
| Biomineralization |
|---|
 |
Silicates are generally inert chemically. Hence they are common minerals. Their resiliency also recommends their use as building materials.
When treated with calcium oxides and water, silicate minerals form Portland cement.
Equilibria involving hydrolysis of silicate minerals are difficult to study. The chief challenge is the very low solubility of SiO44- and its various protonated forms. Such equilibria are relevant to the processes occurring on geological time scales.[7][8] Some plants excrete ligands that dissolve silicates, a step in biomineralization.
Detection
Silicate anions in solution react with
Zeolite formation and geopolymers polymerisation
The nature of soluble silicates is relevant to understanding
See also
- Alkali-silica reaction
- Carbon cycle
- Carbonate-silicate cycle
- Ocean acidification
References
- ^ ISBN 978-0-08-037941-8.
- ISBN 978-0-87031-652-4.
- ISSN 0958-9465.
- ISSN 0958-9465.
- ISSN 0958-9465.
- ISSN 0958-9465.
- ^ PMID 17886822.
- ^
