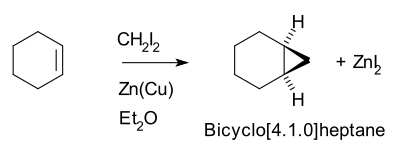
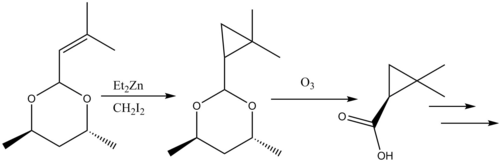
Simmons–Smith reaction
| Simmons-Smith reaction | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Named after | Howard Ensign Simmons, Jr.
Ronald D. Smith | ||||||||
| Reaction type | Ring forming reaction | ||||||||
| Reaction | |||||||||
| |||||||||
| Conditions | |||||||||
| Identifiers | |||||||||
| Organic Chemistry Portal | simmons-smith-reaction | ||||||||
| RSC ontology ID | RXNO:0000258 | ||||||||
The Simmons–Smith reaction is an

Mechanism
 Simmons-Smith reaction in progress |
Examples
Thus,
The Simmons–Smith reaction is generally preferred over other methods of cyclopropanation,[7] however it can be expensive due to the high cost of diiodomethane. Modifications involving cheaper alternatives have been developed, such as dibromomethane[8] or diazomethane and zinc iodide.[9] The reactivity of the system can also be increased by using the Furukawa modification, exchanging the zinc‑copper couple for diethylzinc.[10]
The Simmons–Smith reaction is generally subject to steric effects, and thus cyclopropanation usually takes place on the less hindered face.[11][12] However, when a hydroxy substituent is present in the substrate in proximity to the double bond, the zinc coordinates with the hydroxy substituent, directing cyclopropanation cis to the hydroxyl group (which may not correspond to cyclopropanation of the sterically most accessible face of the double bond):[13] An interactive 3D model of this reaction can be seen at ChemTube3D.
Asymmetric Simmons–Smith reaction
Although
The
Scope and limitations
Achiral alkenes
The Simmons–Smith reaction can be used to cyclopropanate simple

Despite the electron-withdrawing nature of

The cyclopropanation of N-substituted alkenes is made complicated by N-alkylation as a competing pathway. This can be circumvented by adding a protecting group to nitrogen, however the addition of electron-withdrawing groups decreases the nucleophilicity of the alkene, lowering yield. The use of highly electrophilic reagents such as CHFI2, in place of CH2I2, has been shown to increase yield in these cases.[21]
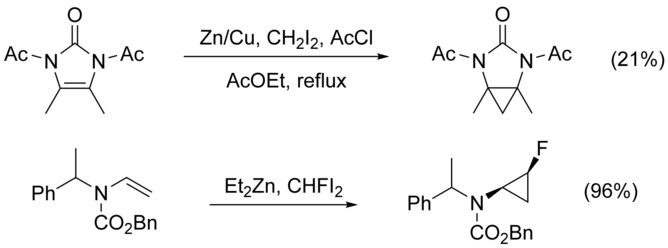
Polyenes
Without the presence of a directing group on the olefin, very little chemoselectivity is observed.[22] However, an alkene which is significantly more nucleophilic than any others will be highly favored. For example, cyclopropanation occurs highly selectively at enol ethers.[23]

Functional group compatibility
An important aspect of the Simmons–Smith reaction that contributes to its wide usage is its ability to be used in the presence of many functional groups. Among others, the haloalkylzinc-mediated reaction is compatible with
Most side reactions occur due to the Lewis-acidity of the byproduct, ZnI2. In reactions that produce acid-sensitive products, excess Et2Zn can be added to scavenge the ZnI2 that is formed, forming the less acidic EtZnI. The reaction can also be quenched with pyridine, which will scavenge ZnI2 and excess reagents.[24]

Modifications
While the Simmons–Smith reaction is often discussed in its basic form, a number of modifications to both the zinc catalyst and the added carbon have been proposed.
Furukawa modification
The Furukawa modification involves the replacement of the
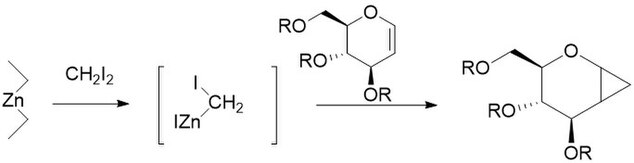
Charette modification
The Charette modification replaces the CH2I2 normally found in the Simmons–Smith reaction with aryldiazo compounds, such as
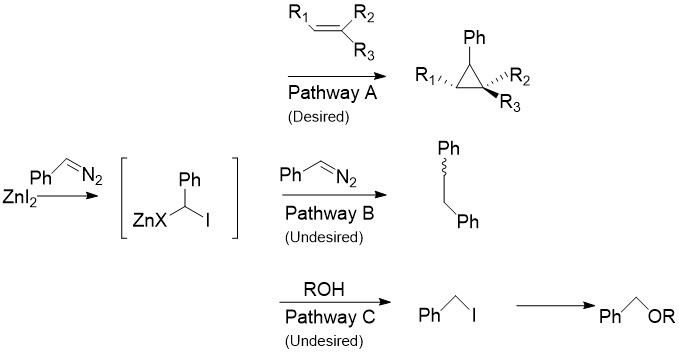
Non-zinc reagents
Although not commonly used, Simmons-Smith reagents that display similar reactive properties to those of zinc have been prepared from aluminum and samarium compounds in the presence of CH2IX. However, both reactions require near stoichiometric amounts of the starting metal compound, and Sm/Hg must be activated with the highly toxic HgCl2.
Most modern applications of the Simmons–Smith reaction use the Furukawa modification. Especially relevant and reliable applications are listed below.
A Furukawa-modified Simmons-Smith generated cyclopropane intermediate is formed in the synthesis of γ-keto esters from β-keto esters. The Simmons-Smith reagent binds first to the carbonyl group and subsequently to the α-carbon of the pseudo-enol that the first reaction forms. This second reagent forms the cyclopropyl intermediate which rapidly fragments into the product.[34][35]
A Furukawa-modified Simmons–Smith reaction cyclopropanates both double bonds in an allenamide to form amido-spiro [2.2] cyclopentanes, featuring two cyclopropyl rings which share one carbon. The product of monocyclopropanation is also formed.[36][37]
Cyclopropanation reactions in 
Uses in synthesis
Insertion to form γ-keto esters

Formation of amido-spiro [2.2] pentanes from allenamides

Natural product synthesis
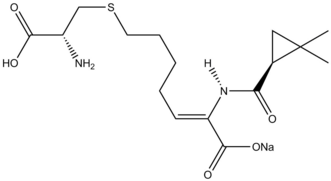
Drugs synthesis
The Simmons–Smith reaction is used in the syntheses of GSK1360707F and ropanicant.[39]
References
- .
- .
- .
- ISBN 978-0-471-26418-7.
- ^ Smith, R. D.; Simmons, H. E. "Norcarane". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 855. - ^ Ito, Y.; Fujii, S.; Nakatuska, M.; Kawamoto, F.; Saegusa, T. (1988). "One-Carbon Ring Expansion Of Cycloalkanones To Conjugated Cycloalkenones: 2-Cyclohepten-1-one". Organic Syntheses
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 327. - ISBN 978-0-19-850346-0.Page 1067
- .
- .
- .
- ISBN 978-0-471-26418-7, retrieved 2022-02-28
- ^ Girard, C.; Conia, J. M. (1978). Journal of Chemical Research, Synopses (Review): 182–.
{{cite journal}}: Missing or empty|title=(help) - .
- doi:10.1016/S0040-4039(00)92246-9.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 21627114.
- PMID 18288666.
- .
- .
- ^ Morikawa, T.; Sasaki, H.; Mori, K.; Shiro, M.; Taguchi, T.; Morikawa, T.; Sasaki, H.; Mori, K.; Shiro, M.; Taguchi, T. Simmons-Smith Reactions of Fluoroallyl Alcohol Derivatives. Chem. Pharm. Bull. (Tokyo) 1992, 40 (12), 3189.
- ^ Piers, E.; Coish, P. D. Preparation and Cyclopropanation of 2- and 3-Iodoalk-2-En-1-Ols: Synthesis of Functionalized, Stereodefined Iodocyclopropanes. Synthesis 1995, 1995 (1), 47–55.
- ^ Gagnon, J. L.; Jr, W. W. Z. Synthesis of Cis-1,5-Dimethyl-2,4-Dinitro-2,4-diazabicyclo[3.1.0]hexan-3-One and Cis-1,5-Dimethyl-2,4-Dinitro-2,4-diazabicyclo[3.2.0]heptan-3-One. Synth. Commun. 1996, 26 (4), 837–845.
- .
- .
- ^ Denis, J. M.; Girard, C.; Conia, J. M. Improved Simmons-Smith Reactions. Synthesis 1972, 1972 (10), 549–551.
- .
- .
- .
- ISBN 978-0-7623-0631-2.
- ^ "Diethyl Zinc MSDS" (PDF). Retrieved 10 May 2017.
- PMID 24555697.
- ISBN 978-0-471-10590-9.
- .
- .
- .
- .
- .
- .
- .
- S2CID 219767881.
External links