Slow-wave sleep
Slow-wave sleep (SWS), often referred to as deep sleep, is the third stage of non-rapid eye movement sleep (NREM), where electroencephalography activity is characterised by slow delta waves.[2]
Slow-wave sleep usually lasts between 70 and 90 minutes, taking place during the first hours of the night.[3] SWS is characterised by moderate muscle tone, slow or absent eye movement, and lack of genital activity. Slow-wave sleep is considered important for memory consolidation, declarative memory, and the recovery of the brain from daily activities.
Prior to 2007, the term SWS referred to both the third and fourth stages of NREM. However, after both stages were combined into stage three, SWS refers only to the third stage.[4]: 291
Overview
Terminology
This period of sleep is called slow-wave sleep because the EEG activity is synchronized, and characterised by slow waves with a frequency range of 0.5–4.5 Hz and a relatively high amplitude power with peak-to-peak amplitude greater than 75µV. The first section of the wave signifies a "down state", an inhibition or hyperpolarizing phase in which the neurons in the neocortex are silent. This is the period when the neocortical neurons are able to rest. The second section of the wave signifies an "up state", an excitation or depolarizing phase in which the neurons fire briefly at a high rate. The principal characteristics during slow-wave sleep that contrast with REM sleep are moderate muscle tone, slow or absent eye movement, and lack of genital activity.[4]: 291, 293
Prior to 2007, the American Academy of Sleep Medicine (AASM) divided slow-wave sleep into stages 3 and 4.[5][6][7] The two stages are now combined as Stage three or N3. An epoch (30 seconds of sleep) which consists of 20% or more slow-wave (delta) sleep is now considered to be in slow-wave sleep.
Importance
Slow-wave sleep is considered important for
Specifically, SWS presents a role in spatial
Affective representations are generally better remembered during sleep compared to neutral ones. Emotions with negative salience presented as a cue during SWS show better reactivation, and therefore an enhanced consolidation in comparison to neutral memories. The former was predicted by
Sleep deprivation studies with humans suggest that the primary function of slow-wave sleep may be to allow the brain to recover from its daily activities.
Electroencephalographic characteristics
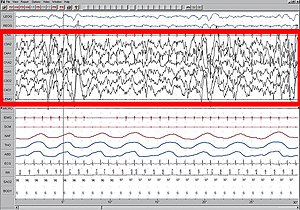
High amplitude EEG is highlighted in red.
Large 75-microvolt (0.5–2.0 Hz)
Longer periods of SWS occur in the first part of the night, primarily in the first two sleep cycles (roughly three hours). Children and young adults will have more total SWS in a night than older adults. The elderly may not go into SWS at all during many nights of sleep.[citation needed]
NREM sleep, as observed on the electroencephalogram (EEG), is distinguished by certain characteristic features.
Slow-wave sleep is an active phenomenon probably brought about by the activation of serotonergic neurons of the raphe system.[23]
The slow-wave seen in the cortical EEG is generated through recurrent connections within the cerebral cortex, where cortical pyramidal cells excite one another in a positive feedback loop. This recurrent excitation is balanced by inhibition, resulting in the active state of the slow oscillation of slow wave sleep. Failure of this mechanism results in a silencing of activity for a brief period of time. The recurrence of active and silent periods occurs at a rate of 0.5–4 Hz, giving rise to the slow waves of the EEG seen during slow wave sleep.[24]
Functions
Hemispheric asymmetries in the human sleep
Slow-wave sleep is necessary for survival. Some animals, such as dolphins and birds, have the ability to sleep with only one hemisphere of the brain, leaving the other hemisphere awake to carry out normal functions and to remain alert. This kind of sleep is called unihemispheric slow-wave sleep, and is also partially observable in human beings. Indeed, a study reported a unilateral activation of the somatosensorial cortex when a vibrating stimulus was put on the hand of human subjects. The recordings show an important inter-hemispheric change during the first hour of non-REM sleep and consequently the presence of a local and use-dependent aspect of sleep.[25] Another experiment detected a greater number of delta waves in the frontal and central regions of the right hemisphere.[26]
Considering that SWS is the only sleep stage that reports human deep sleep as well as being used in studies with mammals and birds, it is also adopted in experiments revealing the role of
The left hemisphere is shown to be more sensitive to deviant stimuli during the first night—compared to the following nights of an experiment. This asymmetry explains further the reduced sleep of half the brain during SWS. Indeed, in comparison to the right one, the left hemisphere plays a vigilant role during SWS.[27]
Furthermore, a faster behavioral reactivity is detected in the left hemisphere during SWS of the first night. The rapid awakening is correlated to the regional asymmetry in the activities of SWS. These findings show that the hemispheric asymmetry in SWS plays a role as a protective mechanism. SWS is therefore sensitive to danger and non-familiar environment, creating a need for vigilance and reactivity during sleep.[27]
Neural control of slow-wave sleep
Several neurotransmitters are involved in sleep and waking patterns: acetylcholine, norepinephrine, serotonin, histamine, and orexin.[4]: 305–307 Neocortical neurons fire spontaneously during slow-wave sleep, thus they seem to play a role during this period of sleep. Also, these neurons appear to have some sort of internal dialogue, which accounts for the mental activity during this state where there is no information from external signals (because of the synaptic inhibition at the thalamic level). The rate of recall of dreams during this state of sleep is relatively high compared to the other levels of the sleep cycle. This indicates that mental activity is closer to real life events.[14]
Physical healing and growth
Slow-wave sleep is the constructive phase of sleep for recuperation of the mind-body system in which it rebuilds itself after each day. Substances that have been ingested into the body while an organism is awake are synthesized into complex proteins of living tissue. Growth hormone is also secreted during this stage, which leads some scientists to hypothesize that a function of slow wave sleep is to facilitate the healing of muscles as well as repair damage to tissues.
Learning and synaptic homeostasis
Learning and memory formation occurs during wakefulness by the process of long-term potentiation; SWS is associated with the regulation of synapses thus potentiated. SWS has been found to be involved in the downscaling of synapses, in which strongly stimulated or potentiated synapses are kept while weakly potentiated synapses either diminish or are removed.[31] This may be helpful for recalibrating synapses for the next potentiation during wakefulness and for maintaining synaptic plasticity. Notably, new evidence is showing that reactivation and rescaling may be co-occurring during sleep.[32]
Problems associated with slow-wave sleep
Effects of sleep deprivation
J. A. Horne (1978) reviewed several experiments with humans and concluded that sleep deprivation has no effects on people's physiological stress response or ability to perform physical exercise. It did, however, have an effect on cognitive functions. Some people reported distorted perceptions or hallucinations and lack of concentration on mental tasks. Thus, the major role of sleep does not appear to be rest for the body, but rest for the brain.
When sleep-deprived humans sleep normally again, the recovery percentage for each stage of sleep is not the same. Only seven percent of stages one and two are regained, but 68 percent of stage-four slow-wave sleep and 53 percent of REM sleep are regained. This suggests that stage-four sleep (known today as the deepest part of stage-three sleep) is more important than the other stages.
During slow-wave sleep, there is a significant decline in cerebral metabolic rate and
According to J. Siegel (2005), sleep deprivation results in the build-up of
Amyloid beta pathology
Results from a number of research have shown how sleep affects Aβ dynamics.[34] A good candidate for slow wave activity (SWA), which occurs during deep non-REM sleep, is amyloid-b modulation. The researchers also highlighted a strong relationship between amyloid-b and SWA, pointing out that increased disruption in SWA is correlated with elevated levels of amyloid-b.[35] Hence, Slow waves of non-rapid eye movement sleep, or NREM sleep, are disrupted or decrease when amyloid beta (Aβ) builds up in the prefrontal cortex. As a result, this may hinder older people' capacity for memory consolidation.[36]
Moreover, the onset of
Individual differences
Though SWS is fairly consistent within the individual, it can vary across individuals.[39] To some degree, individual variations seem to be influenced by demographic factors such as gender and age.[21] Age and sex have been noted as two of the biggest factors that affect this period of sleep.[39]
Slow-wave sleep (SWS) and slow-wave activity (SWA) undergo significant transformations throughout one's lifespan, with aging serving as a particularly influential factor in predicting individual variations.[21][40] Aging is inversely proportional to the amount of SWS beginning by midlife, so SWS declines with age.[39] Moreover, recent findings indicate that older individuals exhibit a decreased inclination for daytime sleep compared to younger counterparts, and this decline persists even when accounting for variations in habitual sleep duration. This age-related decrease in daytime sleep propensity is evident in middle-aged individuals and coincides with statistically significant reductions in total sleep time, slow-wave sleep (SWS), and slow-wave activity (SWA).[21]
Sex differences have also been found, such that females tend to have higher levels of SWS compared to males, at least up until menopause.[39] Older individuals exhibit gender-based variations in non-rapid eye movement (NREM) sleep, where women demonstrate increased slow-wave sleep (SWS) during both regular and recuperative sleep, along with higher occurrences of stage 3 and 4 which are considered as NREM sleep.[21] There have also been studies that have shown differences between races. The results showed that there was a lower percentage of SWS in African Americans compared to Caucasians, but since there are many influencing factors (e.g., body mass index, sleep-disordered breathing, obesity, diabetes, and hypertension), this potential difference must be investigated further.[39]
Mental disorders play a role in individual differences in the quality and quantity of SWS: subjects with depression show a lower amplitude of slow-wave activity (SWA) compared to healthy participants. Sex differences also persist in the former group: depressed men present significantly lower SWA amplitude. This sex divergence is twice as large as the one observed in healthy subjects. However, no age-related difference concerning SWS can be observed in the depressed group.[41]
Brain regions
During sleep, the distribution of slow-wave activity (SWA) typically exhibits a prevalence in the
Some of the brain regions implicated in the induction of slow-wave sleep include:
- the parafacial zone (GABAergic neurons),[43][44][45] located within the medulla oblongata
- the
- the
- the lateral hypothalamus (melanin-concentrating hormone-releasing neurons),[44][45][50][51] located within the hypothalamus
Drugs
Some drugs influence sleep architecture by encroaching upon or prolonging deep sleep.[52] Many drugs known to increase deep sleep in humans are of the GABAergic, dopaminergic, and anti-serotonergic classes.[53]
The administration of the GABAa agonist gabaxadol enhances both deep sleep while also positively impacting various indicators of insomnia.[52]
Levodopa is a drug commonly used to treat Parkinson's disease which acts to increases the brain's dopamine availability. Nocturnal single doses of levodopa have been shown to increase SWS by 10.6% in elderly.[59]
Antagonists of certain
See also
- Delta sleep-inducing peptide
- Gaboxadol
- Large irregular activity
- Non-rapid eye movement sleep (NREM)
- Preconscious
- Sharp wave–ripple complexes
- Sleep and learning
- Subconscious
- Unconscious mind
- Unihemispheric slow-wave sleep
References
- PMID 21887239.
- ^ Rechtschaffen A, Kales A (1968). A Manual of Standardized Terminology, Techniques and Scoring System for Sleep Stages of Human Subjects. US Dept of Health, Education, and Welfare; National Institutes of Health.
- PMID 10554397.
- ^ ISBN 978-0-205-23939-9.)
{{cite book}}: CS1 maint: location missing publisher (link - PMID 18468306.
Although the sequence of non-REM (NREM) sleep stages one to four (R&K classification) or N1 to N3 (AASM classification) fulfills the criteria...
- ^ "Glossary. A resource from the Division of Sleep Medicine at Harvard Medical School, in partnership with WG Education Foundation". Harvard University. 2008. Archived from the original on 2018-10-04. Retrieved 2009-03-11.
The 1968 categorization of the combined Sleep Stages 3 – 4 was reclassified in 2007 as Stage N3.
- ^ a b Iber, C; Ancoli-Israel, S; Chesson, A; Quan, SF. for the American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications. Westchester: American Academy of Sleep Medicine; 2007.
- ^ Carey, Benedict (2013-01-27). "Aging in Brain Found to Hurt Sleep Needed for Memory". The New York Times. Archived from the original on 2017-03-17. Retrieved 2017-04-17.
- PMID 18803104.
- ^ a b Walker, Matthew P. "The Role of Slow Wave Sleep in Memory Processing" (PDF). Journal of Clinical Sleep Medicine. Supplement to Vol.5, No. 2, 2009. Archived (PDF) from the original on 2013-05-09. Retrieved 2014-05-06.
- PMID 19998871.
- S2CID 15470045. Archived from the original(PDF) on 18 April 2017.
- PMID 12151563.
- ^ PMID 15493541. Archived from the originalon 2014-05-06. Retrieved 2014-05-06.
- ^ S2CID 19788434.
- S2CID 1424898.
- ^ PMID 24688163.
- PMID 14766981.
- ^ ISBN 978-0-9561387-1-2.
- PMID 32332806.
- ^ PMID 19998869.
- S2CID 42683788.
- PMID 12700104.
- S2CID 509469.
- S2CID 26078900.
- S2CID 44808363.
- ^ PMID 27112296.
- ^ "What Happens When You Sleep: The Science of Sleep". Sleep Foundation. 2009-12-22. Archived from the original on 2021-06-21. Retrieved 2021-06-25.
- CiteSeerX 10.1.1.723.1235. Archivedfrom the original on 2021-06-25. Retrieved 2021-06-25 – via CiteSeerX.
- ^ "The importance of sleep & why we need it". Human Givens Institute. 2015-10-23. Archived from the original on July 11, 2019.
- S2CID 16129740.
- PMID 28692062.
- ISBN 0205239390.
- ^ PMID 27568802.
- ^ PMID 28899014.
- PMID 26030850.
- PMID 32714142.
- PMID 15978517.
- ^ PMID 21955220.
- PMID 20175405.
- S2CID 1903649.
- ^ S2CID 206682.
- (PDF) from the original on 2018-11-04. Retrieved 2018-11-04.
In the present study we show, for the first time, that activation of a delimited node of GABAergic neurons located in the medullary PZ can potently initiate SWS and cortical SWA in behaving animals. ... For now however it remains unclear if the PZ is interconnected with other sleep– and wake–promoting nodes beyond the wake–promoting PB. ... The intensity of cortical slow–wave–activity (SWA: 0.5–4Hz) during SWS is also widely accepted as a reliable indicator of sleep need ... In conclusion, in the present study we demonstrated that all polygraphic and neurobehavioral manifestation of SWS, including SWA, can be initiated in behaving animals by the selective activation of a delimited node of GABAergic medullary neurons.
- ^ PMID 26600100.
More recently, the medullary parafacial zone (PZ) adjacent to the facial nerve was identified as a sleep-promoting center on the basis of anatomical, electrophysiological and chemo- and optogenetic studies.23, 24 GABAergic PZ neurons inhibit glutamatergic parabrachial (PB) neurons that project to the BF,25 thereby promoting NREM sleep at the expense of wakefulness and REM sleep. ... Sleep is regulated by GABAergic populations in both the preoptic area and the brainstem; increasing evidence suggests a role for the melanin-concentrating hormone cells of the lateral hypothalamus and the parafacial zone of the brainstem
- ^ PMID 26124745.
The sleep-promoting action of GABAergic neurons located in the preoptic hypothalamus (6–8) is now well-known and accepted (9). More recently, other groups of sleep-promoting GABAergic neurons in the lateral hypothalamus (melanin-concentrating hormone neurons) and brainstem [parafacial zone; (10)] have been identified.
- PMID 29283174.
The nucleus accumbens comprises a contingent of neurons specifically expressing the post-synaptic A2A-receptor (A2AR) subtype making them excitable by adenosine, its natural agonist endowed with powerful sleep-promoting properties[4]. ... In both cases, large activation of A2AR-expressing neurons in NAc promotes slow wave sleep (SWS) by increasing the number and duration of episodes. ... After optogenetic activation of the core, a similar promotion of SWS was observed, whereas no significant effects were induced when activating A2AR-expressing neurons within the shell.
- PMID 28963505.
Here, we show that chemogenetic or optogenetic activation of excitatory adenosine A2A receptor-expressing indirect pathway neurons in the core region of the NAc strongly induces slow-wave sleep. Chemogenetic inhibition of the NAc indirect pathway neurons prevents the sleep induction, but does not affect the homoeostatic sleep rebound.
- PMID 29022877.
- PMID 26156991.
- PMID 23477948.
MCHergic neurons are silent during wakefulness (W), increase their firing during slow wave sleep (SWS) and still more during REM sleep (REMS). Studies in knockout mice for MCH (MCH(-/-)) have shown a reduction in SWS and an increase of W during the light and the dark phase of the light-dark cycle.
- PMID 21516258.
Neurons containing the neuropeptide melanin-concentrating hormone (MCH) are mainly located in the lateral hypothalamus and the incerto-hypothalamic area, and have widespread projections throughout the brain. ... Intraventricular microinjection of MCH increases both slow wave sleep (SWS) and REM sleep; however, the increment in REM sleep is more pronounced. ... Although both SWS and REM sleep are facilitated by MCH, REM sleep seems to be more sensitive to MCH modulation.
- PMID 19998872.
- PMID 192353.
- PMID 21344068.
- ^ "Xyrem - European Drugs Reference Encyclopedia". Archived from the original on 2013-08-21. Retrieved 2013-07-16.
- PMID 16260179.
- PMID 17557435.
- PMID 37091594.
- PMID 12187365.
- S2CID 46587971.
- S2CID 37749231.
Further reading
- Massimini M, Ferrarelli F, Huber R, Esser SK, Singh H, Tononi G (September 2005). "Breakdown of cortical effective connectivity during sleep". Science. 309 (5744): 2228–32. S2CID 38498750.
- Cicogna P, Natale V, Occhionero M, Bosinelli M (2000). "Slow wave and REM sleep mentation". Sleep Research Online. 3 (2): 67–72. PMID 11382903.
- Vogel G, Foulkes D, Trosman H (March 1966). "Ego functions and dreaming during sleep onset". Archives of General Psychiatry. 14 (3): 238–48. PMID 5903415.
- Rock A (2004). The Mind at Night.
- Warren, Jeff (2007). "The Slow Wave". The Head Trip: Adventures on the Wheel of Consciousness. Random House. ISBN 978-0-679-31408-0.
