Sodium aluminate
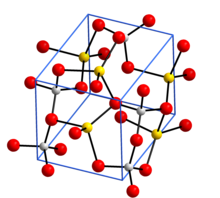 Crystal structure with sodium in yellow, aluminium in grey, and oxygen in red[1]
| |
 Sodium metaaluminate sample
| |
| Names | |
|---|---|
| IUPAC name
Sodium aluminate
| |
| Other names
Sodium aluminium oxide,
Sodium metaaluminate Aluminate, ((AlO2)1−), sodium | |
| Identifiers | |
3D model (
JSmol ) |
|
ECHA InfoCard
|
100.013.728 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| NaAlO2 | |
| Molar mass | 81.97 g/mol |
| Appearance | white powder (sometimes light-yellowish) hygroscopic/ when dissolved in water a colloidal black solution is formed |
| Odor | odorless |
| Density | 1.5 g/cm3 |
| Melting point | 1,650 °C (3,000 °F; 1,920 K) |
| highly soluble | |
| Solubility | Insoluble in alcohol[2] |
Refractive index (nD)
|
1.566 |
| Structure | |
| orthorhombic | |
| Thermochemistry | |
Heat capacity (C)
|
73.6 J/mol K |
Std molar
entropy (S⦵298) |
70.4 J/mol K |
Std enthalpy of (ΔfH⦵298)formation |
-1133.2 kJ/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium aluminate is an
Other related compounds, sometimes called sodium aluminate, prepared by reaction of Na2O and Al2O3 are Na5AlO4 which contains discrete AlO45− anions, Na7Al3O8 and Na17Al5O16 which contain complex polymeric anions, and NaAl11O17, once mistakenly believed to be β-alumina, a phase of aluminium oxide.[4][5]
Structure
Anhydrous sodium aluminate, NaAlO2, contains a three-dimensional framework of corner linked AlO4 tetrahedra. The hydrated form NaAlO2·5/4H2O has layers of AlO4 tetrahedra joined into rings and the layers are held together by sodium ions and water molecules that hydrogen bond to O atoms in the AlO4 tetrahedra.[6]
Manufacturing
Sodium aluminate is manufactured by the dissolution of aluminium hydroxide (Al(OH)3) in a
Reaction of aluminium and alkali
Sodium aluminate is also formed by the action of sodium hydroxide on elemental aluminium which is an
- 2Al + 2NaOH + 2H2O → 2NaAlO2 + 3H2
However, the species produced in solution is likely to contain the [Al(OH)4]− ion or perhaps the [Al(H2O)2(OH)4]− ion.[7]
Uses
In water treatment it is used as an adjunct to water softening systems, as a coagulant aid to improve
In construction technology, sodium aluminate is employed to accelerate the solidification of concrete, mainly when working during frost.
Sodium aluminate is also used in the paper industry, for fire brick production, alumina production and so forth.
Sodium aluminate solutions are intermediates in the production of
References
- .
- ^ The Merck Index. 10th ed. Rahway, New Jersey: Merck Co., Inc., 1983., p. 1229
- ^ "Aluminium". chemguide.co.uk.
- ISBN 0-12-352651-5
- ISBN 978-0-08-037941-8.
- S2CID 213214375.
- ISBN 0-471-93620-0
